Advanced Synthesis of 3,6-Dichloropyridazin-4-ol for High-Efficiency Herbicide Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical heterocyclic intermediates. A significant technological breakthrough in this sector is detailed in patent CN113277984A, which discloses a novel and robust method for preparing 3,6-dichloropyridazin-4-ol. This compound serves as a pivotal building block for the synthesis of cyclopyrimorate, a potent pyridazine herbicide widely used in paddy fields to control barnyard grass and broadleaf weeds. The disclosed methodology represents a paradigm shift from traditional, hazardous chlorination processes to a safer, two-step synthetic route that leverages 3-acetoxyl-2,5-furandione as a key starting material. By avoiding the use of elemental chlorine gas and high-pressure conditions, this invention addresses long-standing safety and environmental concerns while delivering superior chemical purity exceeding 98.0% by gas chromatography. For R&D directors and process chemists, this patent offers a compelling alternative that balances operational safety with high reaction efficiency, positioning it as a cornerstone technology for modern agrochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 3,6-dichloropyridazin-4-ol has relied on processes that pose significant safety and environmental challenges. One prevalent method involves the use of maleic anhydride as a raw material, which undergoes a series of transformations including high-temperature chlorination with virulent chlorine gas. This approach necessitates specialized equipment capable of withstanding corrosive environments and high pressures, leading to substantial capital expenditure and maintenance costs. Furthermore, the handling of elemental chlorine introduces severe occupational health risks and requires rigorous waste gas treatment systems to prevent environmental contamination. Another existing route utilizes 2,6-dichloropyridazine as a precursor; however, this pathway is often plagued by low reaction yields and poor atom economy, resulting in excessive waste generation and higher raw material consumption. These inherent limitations in conventional technologies create bottlenecks in supply chain reliability and increase the overall cost of goods sold for downstream herbicide manufacturers.
The Novel Approach
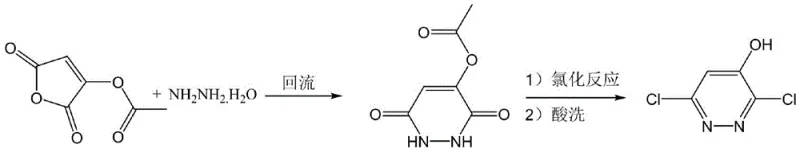
In stark contrast to these legacy methods, the novel approach described in the patent utilizes 3-acetoxyl-2,5-furandione, a readily available and cost-effective feedstock, to construct the pyridazine ring system. The process initiates with a cyclization reaction using hydrazine hydrate, forming a stable dihydropyridazine dione intermediate under mild thermal conditions. This is followed by a catalytic chlorination step that replaces hydroxyl groups with chlorine atoms efficiently. By substituting dangerous chlorine gas with liquid chlorinating agents such as phosphorus oxychloride or phosphorus trichloride, the process eliminates the need for high-pressure reactors and complex gas scrubbing infrastructure. The result is a streamlined workflow that not only enhances operator safety but also simplifies the purification process, yielding a product with exceptional purity profiles suitable for direct use in subsequent coupling reactions for herbicide synthesis.
Mechanistic Insights into Lewis Acid-Catalyzed Chlorination
The core innovation of this synthesis lies in the second step, where the intermediate 4-acetoxyl-1,2-dihydropyridazine-3,6-dione undergoes chlorination and deacetylation. The patent highlights the critical role of Lewis acid catalysts, such as anhydrous ferric trichloride (FeCl3), anhydrous aluminum trichloride (AlCl3), or triphenylboron, in driving this transformation. Mechanistically, the Lewis acid coordinates with the oxygen atoms of the carbonyl and acetoxy groups, increasing the electrophilicity of the carbon centers and facilitating the nucleophilic attack by the chlorinating agent. This catalytic activation lowers the activation energy required for the substitution reaction, allowing it to proceed at moderate temperatures ranging from 60°C to 100°C. Without the catalyst, comparative examples in the patent demonstrate that the reaction suffers from incomplete conversion and the formation of significant amounts of monochloro-substituted byproducts, which are difficult to separate and reduce the overall yield.
Furthermore, the reaction conditions are meticulously optimized to control impurity profiles and ensure high regioselectivity. The use of an acid-binding agent, such as triethylamine or pyridine, during the chlorination step neutralizes the hydrogen chloride generated in situ, preventing acid-catalyzed degradation of the sensitive pyridazine ring. Following the chlorination, the mixture is subjected to acidic hydrolysis in an aqueous solution, which simultaneously removes the acetyl protecting group and precipitates the final 3,6-dichloropyridazin-4-ol product. This tandem chlorination-deacetylation strategy minimizes the number of unit operations required, thereby reducing solvent usage and processing time. The precise control over molar ratios—specifically maintaining a catalyst-to-substrate ratio of approximately 0.01 to 0.03—ensures that the reaction proceeds to completion without excessive catalyst loading, which could complicate downstream metal removal processes.
How to Synthesize 3,6-Dichloropyridazin-4-ol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The procedure begins with the dissolution of 3-acetoxyl-2,5-furandione in a polar solvent such as ethanol or water, followed by the controlled addition of hydrazine hydrate to effect ring closure. Once the intermediate is isolated, it is subjected to the catalytic chlorination sequence described previously. For detailed operational parameters, including specific temperature ramps, stirring rates, and work-up procedures, please refer to the standardized synthesis guide below which encapsulates the critical process variables identified in the patent examples.
- Cyclization Reaction: React 3-acetoxyl-2,5-furandione with hydrazine hydrate in a solvent like ethanol at 40-100°C to form 4-acetoxyl-1,2-dihydropyridazine-3,6-dione.
- Chlorination and Deacetylation: Treat the intermediate with a chlorinating agent (e.g., phosphorus oxychloride) and a Lewis acid catalyst (e.g., FeCl3), followed by acid hydrolysis to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The transition away from hazardous gaseous chlorine fundamentally alters the risk profile of the manufacturing facility, potentially lowering insurance premiums and regulatory compliance costs associated with storing and handling toxic gases. Moreover, the reliance on liquid reagents like phosphorus oxychloride simplifies logistics and inventory management, as these materials are standard commodities in the fine chemical industry with stable supply chains. The robustness of the reaction conditions, which tolerate a range of solvents and temperatures, provides operational flexibility that can be leveraged to optimize energy consumption based on local utility costs. These factors collectively contribute to a more resilient and cost-effective supply chain for critical agrochemical intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and specialized gas handling infrastructure results in significant capital expenditure savings for new production lines. Additionally, the improved two-step yield, reaching up to 93% in optimized examples, directly translates to lower raw material consumption per kilogram of finished product. By minimizing the formation of hard-to-remove impurities, the process reduces the burden on purification steps such as recrystallization or chromatography, further driving down processing costs and solvent waste disposal fees.
- Enhanced Supply Chain Reliability: Sourcing 3-acetoxyl-2,5-furandione and hydrazine hydrate is generally more straightforward and less prone to geopolitical disruption compared to securing bulk quantities of elemental chlorine, which is often subject to strict transportation regulations. The modular nature of the batch process allows for scalable production that can be easily adjusted to meet fluctuating market demands for herbicide intermediates. This flexibility ensures that manufacturers can maintain consistent delivery schedules to downstream formulators, even during periods of raw material volatility.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts and avoids the release of toxic chlorine gas, aligning with increasingly stringent global environmental regulations. The use of common organic solvents and aqueous acid washes simplifies wastewater treatment protocols, reducing the environmental footprint of the manufacturing site. This green chemistry advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production facilities.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: Unlike conventional methods that utilize highly toxic chlorine gas under high temperature and pressure, this patented route employs safer chlorinating agents like phosphorus oxychloride at moderate temperatures, significantly reducing operational hazards and infrastructure costs.
Q: How does the use of Lewis acid catalysts impact the reaction yield?
A: The introduction of catalysts such as anhydrous ferric trichloride or aluminum trichloride facilitates the chlorination step, improving the two-step overall yield to between 78% and 93%, compared to significantly lower yields in non-catalytic processes.
Q: Is this process suitable for large-scale commercial production of herbicide intermediates?
A: Yes, the process utilizes cheap and easily obtained raw materials like 3-acetoxyl-2,5-furandione and operates under mild conditions, making it highly scalable and economically viable for industrial manufacturing of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Dichloropyridazin-4-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable intermediate supply in the fast-paced agrochemical sector. Our team of expert process chemists has extensively analyzed the route described in CN113277984A and possesses the technical capability to scale this synthesis from laboratory benchtop to full commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering 3,6-dichloropyridazin-4-ol with purity levels exceeding 98.0% as required for high-performance herbicide formulations.
We invite procurement directors and supply chain managers to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing processes and bulk sourcing capabilities, we can help you achieve significant reductions in your total cost of ownership for pyridazine intermediates. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing quotes that will empower your supply chain strategy for the upcoming growing season.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
