Scalable Production of Ferrocenecarboxaldehyde Using Advanced Solvent-Free Vilsmeier Chemistry
The chemical landscape for organometallic intermediates has evolved significantly, driven by the demand for greener, more efficient synthetic routes that align with modern regulatory and economic standards. Patent CN103145768A introduces a transformative methodology for the preparation of ferrocenecarboxaldehyde, a critical building block widely utilized in the synthesis of bioactive ferrocene derivatives for pharmaceutical and agrochemical applications. This innovation addresses long-standing inefficiencies in traditional formylation processes by leveraging a solvent-free Vilsmeier-Haack reaction system. By utilizing dimethylformamide (DMF) not merely as a reagent but effectively as the reaction medium, the process achieves a remarkable convergence of operational simplicity and chemical efficiency. The technical breakthrough lies in the precise control of stoichiometry and temperature, allowing for the direct conversion of ferrocene to its aldehyde derivative without the need for auxiliary organic solvents like chloroform. This approach not only streamlines the workflow but also fundamentally alters the economic and environmental footprint of producing this high-value intermediate, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks seeking sustainable optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ferrocenecarboxaldehyde has been plagued by procedural complexities and safety concerns inherent to older methodologies, such as the method reported by Masaru Sato in 1968. This conventional route typically necessitates the use of chloroform as a bulk solvent, which introduces significant hazards regarding toxicity, volatility, and environmental disposal costs. Furthermore, the traditional process requires rigorous inert atmosphere protection, often involving nitrogen purging to prevent oxidation or moisture interference, which adds layers of operational complexity and equipment requirements. Perhaps most critically, the reaction kinetics in these older systems are sluggish, often demanding extended heating periods of up to 20 hours at elevated temperatures around 55-60°C to drive the conversion to completion. Such prolonged exposure to heat increases the risk of thermal decomposition and the formation of polymeric byproducts, thereby compromising the purity of the final product and necessitating extensive downstream purification efforts. The combination of hazardous solvents, long cycle times, and difficult workup procedures renders these legacy methods economically unviable for modern, high-volume commercial scale-up of complex organometallic intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data revolutionizes this transformation by adopting a 'one-pot' solvent-free strategy that drastically reduces both time and resource consumption. By eliminating chloroform entirely and relying on the dual functionality of DMF, the process creates a highly concentrated reaction environment that accelerates the formation of the active Vilsmeier reagent. The reaction conditions are notably mild, proceeding efficiently at temperatures ranging from 20°C to 60°C, which significantly lowers energy consumption and mitigates safety risks associated with high-temperature operations. The reaction time is compressed dramatically from nearly a day to a mere 1 to 2.5 hours, representing a massive improvement in throughput capacity for manufacturing facilities. Additionally, the workup procedure is simplified to a straightforward aqueous quench and extraction, avoiding the tedious distillation of bulk solvents required in previous methods. This streamlined approach not only enhances the overall yield, which can reach impressive levels up to 85%, but also ensures a cleaner crude product profile, facilitating easier recrystallization and final purification.
Mechanistic Insights into Solvent-Free Vilsmeier-Haack Formylation
The core of this technological advancement relies on the precise generation and utilization of the Vilsmeier-Haack reagent within a neat reaction matrix. The mechanism initiates with the interaction between phosphorus oxychloride (POCl3) and DMF at low temperatures, specifically between 8°C and 10°C, to form the highly electrophilic chloroiminium salt intermediate. In traditional dilute solutions, the collision frequency between reactants is lower, but in this solvent-free system, the high concentration of DMF ensures rapid and quantitative formation of the active species. Once formed, this electrophile attacks the electron-rich cyclopentadienyl rings of the ferrocene substrate. Ferrocene is known for its exceptional nucleophilicity due to the iron center donating electron density to the ligands, making it highly susceptible to electrophilic aromatic substitution. The absence of competing solvent molecules allows the Vilsmeier reagent to interact directly and efficiently with the ferrocene surface, driving the substitution reaction forward with high regioselectivity for the mono-formylated product.
Following the electrophilic attack, the resulting iminium complex undergoes hydrolysis upon the addition of water during the workup phase. This step is critical for releasing the free aldehyde group from the nitrogen-containing intermediate. The patent specifies a careful pH adjustment to neutrality (pH 6-7) using sodium or potassium hydroxide solutions, which is essential to prevent acid-catalyzed degradation of the sensitive ferrocenyl aldehyde or the formation of resinous tars. The subsequent extraction with dichloromethane effectively separates the organic product from inorganic phosphate salts generated during the reaction. The final recrystallization from a dichloromethane and n-hexane mixture further refines the crystal lattice, removing trace impurities and ensuring the high melting point consistency observed in the embodiments. This mechanistic clarity provides R&D teams with the confidence to adapt the process for various substituted ferrocene derivatives, knowing that the fundamental electronic drivers remain consistent.
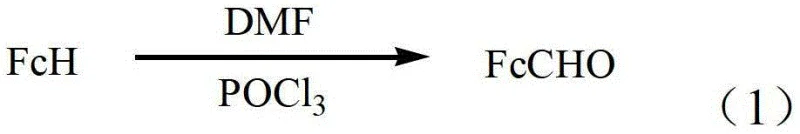
How to Synthesize Ferrocenecarboxaldehyde Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters outlined in the patent to maximize yield and safety. The process begins with the controlled addition of phosphorus oxychloride to DMF under cooling, followed by the introduction of ferrocene and a gradual warm-up phase. Operators must monitor the exotherm carefully during the initial mixing to maintain the specified 8-10°C range, preventing premature decomposition of the Vilsmeier reagent. Once the ferrocene is added, the temperature is ramped to the 20-60°C window, where the bulk of the conversion occurs over a short 1 to 2.5-hour period. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational sequence.
- Charge DMF into a reactor and cool to 8-10°C, then slowly add phosphorus oxychloride (POCl3) while maintaining the molar ratio of DMF to POCl3 at 1: 1.
- After stirring for 0.5 hours, add ferrocene gradually and raise the temperature to 20-60°C, maintaining reaction for 1 to 2.5 hours to complete the formylation.
- Quench the mixture with water, adjust pH to 6-7 using NaOH or KOH, extract with dichloromethane, dry, and recrystallize from a DCM/n-hexane mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this solvent-free methodology represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of chloroform, a regulated and increasingly expensive solvent, removes a major line item from the raw material budget and simplifies regulatory compliance regarding volatile organic compound (VOC) emissions. Furthermore, the drastic reduction in reaction time from 20 hours to roughly 2 hours means that existing reactor assets can be turned over much more frequently, effectively increasing production capacity without the need for capital expenditure on new equipment. This intensification of the process leads to substantial cost savings by lowering utility consumption, particularly steam for heating and electricity for agitation over extended periods. The simplified workup also reduces the demand for drying agents and solvent recovery infrastructure, streamlining the entire production workflow and minimizing waste generation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of auxiliary solvents and the compression of cycle times. By operating without chloroform, the facility avoids the costs associated with solvent purchase, storage, and hazardous waste disposal, which are significant overheads in fine chemical production. Additionally, the high yield achieved through this optimized pathway means that less starting material is wasted, improving the overall atom economy of the synthesis. The reduction in processing time allows for better utilization of labor and equipment, effectively lowering the fixed cost allocation per kilogram of product. These factors combine to create a more competitive cost structure, enabling suppliers to offer better pricing while maintaining healthy margins in a volatile market.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this one-pot method ensures greater consistency in delivery schedules. Traditional methods prone to long reaction times and complex workups are more susceptible to batch failures or delays due to equipment bottlenecks. In contrast, the shortened cycle time of this novel approach allows for more flexible scheduling and faster response to urgent customer demands. The use of common, readily available reagents like DMF and POCl3 reduces the risk of raw material shortages that might occur with specialized solvents. This reliability is crucial for downstream pharmaceutical manufacturers who depend on a steady flow of high-quality intermediates to maintain their own production timelines and avoid costly stoppages in API synthesis.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions and the absence of hazardous bulk solvents. The lower operating temperatures reduce the thermal load on cooling systems, making it easier to manage heat transfer in large-scale reactors. Environmentally, the process aligns with green chemistry principles by minimizing solvent use and waste generation, which simplifies permitting and reduces the environmental footprint of the manufacturing site. The straightforward aqueous workup generates less hazardous waste compared to solvent-intensive protocols, easing the burden on wastewater treatment facilities. These attributes make the technology highly attractive for companies aiming to meet stringent sustainability goals while expanding their production capabilities for complex organometallic compounds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. They are derived from the specific operational parameters and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of adoption. Understanding these nuances is essential for ensuring successful technology transfer and optimizing the process for specific facility constraints.
Q: What are the primary advantages of this solvent-free method over traditional chloroform-based synthesis?
A: The solvent-free approach eliminates the need for hazardous chloroform, significantly reducing reaction time from 20 hours to under 2.5 hours, and simplifies post-reaction workup by avoiding complex solvent removal steps.
Q: How does the temperature control impact the purity of ferrocenecarboxaldehyde?
A: Maintaining the reaction temperature between 20°C and 60°C prevents thermal degradation and side reactions, ensuring a cleaner impurity profile and higher isolated yields compared to high-temperature conventional methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the one-pot nature, mild conditions, and absence of exotic solvents make this protocol highly scalable for industrial production, offering robust supply chain continuity for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenecarboxaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity organometallic intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering ferrocenecarboxaldehyde with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to implement solvent-free and green chemistry protocols allows us to offer a sustainable supply chain solution that meets the evolving needs of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for ferrocene derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project milestones. By leveraging our advanced manufacturing technologies, you can secure a reliable source of high-quality intermediates that drive innovation and efficiency in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
