Advanced Arylamine Compounds for High-Efficiency OLED Capping Layers and Commercial Scale-Up
Introduction to Next-Generation OLED Capping Layer Technology
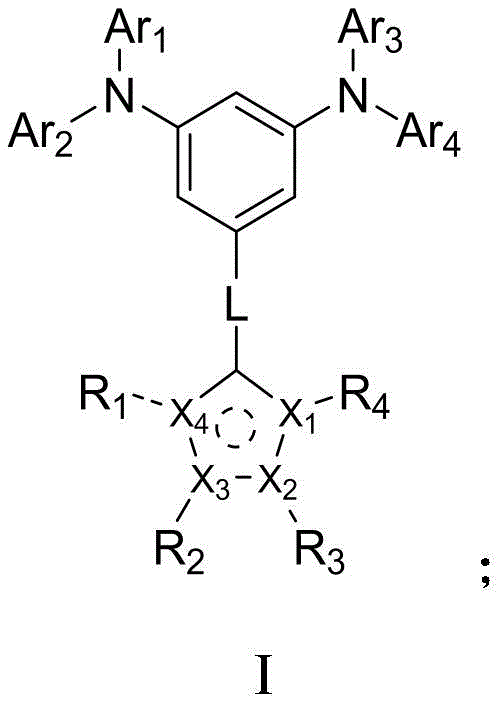
The rapid evolution of organic light-emitting diode (OLED) technology demands continuous innovation in material science, particularly for components that optimize light extraction and device longevity. Patent CN113121493B introduces a groundbreaking class of arylamine compounds specifically engineered to serve as high-performance capping layer materials in organic electroluminescent devices. These novel compounds are characterized by a unique structural integration of arylamine groups and five-membered heterocyclic rings, which collectively address critical limitations found in conventional capping layer materials. By leveraging this specific molecular architecture, the technology achieves a refractive index greater than 2.0 in the visible light spectrum, a crucial parameter for minimizing total internal reflection and maximizing photon output. Furthermore, the inherent steric hindrance within the molecular structure contributes to an exceptionally high glass transition temperature, ensuring that the thin films remain stable under the thermal stress of prolonged device operation. This patent represents a significant leap forward for manufacturers seeking to enhance the efficiency and lifespan of next-generation display panels and lighting solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional materials utilized for OLED capping layers often struggle to balance optical performance with thermal stability, creating a bottleneck for high-efficiency device manufacturing. Many existing aromatic amine derivatives or phosphoroxy-based compounds fail to achieve a sufficiently high refractive index, which directly limits the light extraction efficiency of the OLED device. When the refractive index is too low, a significant portion of the generated light is trapped within the device layers due to total internal reflection, resulting in wasted energy and reduced brightness. Additionally, conventional materials frequently exhibit lower glass transition temperatures, making them susceptible to crystallization or morphological changes when the device heats up during operation. This instability can lead to the formation of dark spots and a rapid decline in luminous efficiency over time. The reliance on these suboptimal materials forces manufacturers to compromise on either the brightness or the operational lifetime of their display products, hindering the development of premium consumer electronics.
The Novel Approach
The innovative approach detailed in the patent overcomes these historical challenges by synthesizing arylamine compounds that incorporate specific five-membered heterocyclic structures directly into the molecular backbone. This structural modification is not merely cosmetic; it fundamentally alters the electronic and physical properties of the material to suit the rigorous demands of modern OLED architectures. The presence of the heterocyclic ring significantly enhances the polarizability of the molecule, which directly correlates to a higher refractive index in the visible light range, effectively improving the light out-coupling efficiency. Simultaneously, the bulky arylamine substituents create a robust steric hindrance effect that inhibits molecular movement, thereby elevating the glass transition temperature and ensuring exceptional film stability. This dual improvement allows device engineers to design OLED stacks that are both brighter and more durable without necessitating complex changes to the existing deposition equipment or process flows.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling Synthesis
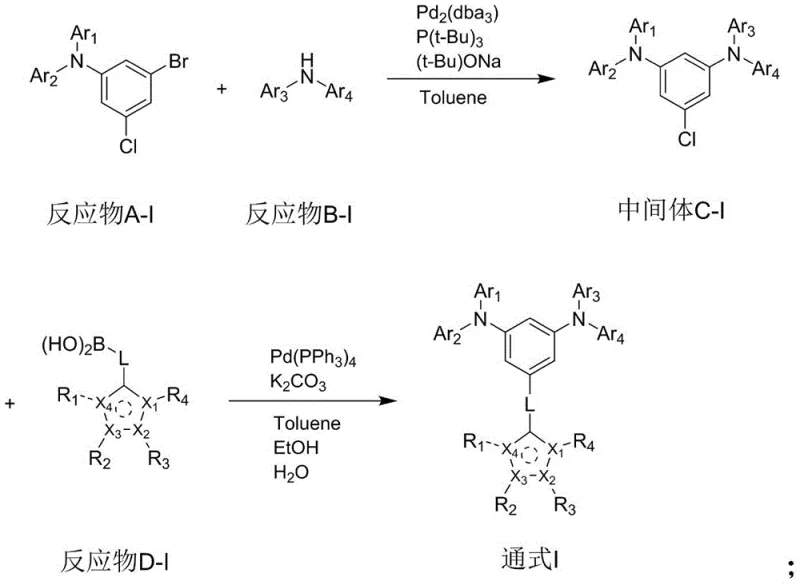
The synthesis of these high-performance arylamine compounds relies on a sophisticated yet robust two-step palladium-catalyzed cross-coupling strategy that ensures high purity and structural precision. The first stage typically involves a Buchwald-Hartwig amination reaction, where a halogenated arylamine precursor reacts with a secondary amine in the presence of a palladium catalyst and a specialized phosphine ligand. This step is critical for establishing the core triarylamine structure, which serves as the electron-donating backbone of the final molecule. The reaction conditions are carefully optimized, often utilizing toluene as a solvent and heating the mixture to temperatures between 100°C and 115°C to drive the coupling to completion. Following this, the intermediate undergoes rigorous purification, including hot filtration through diatomaceous earth to remove palladium residues, which is essential for preventing metal contamination that could quench excitons in the final OLED device.
The second stage employs a Suzuki-Miyaura coupling reaction to attach the crucial five-membered heterocyclic moiety to the arylamine intermediate. This step involves reacting the chlorinated intermediate with a heterocyclic boronic acid derivative using a palladium catalyst and a carbonate base in a mixed solvent system. The choice of the heterocyclic boronic acid is pivotal, as it determines the final refractive index and electronic properties of the capping layer material. The reaction proceeds under reflux conditions, typically around 110°C to 120°C, ensuring efficient bond formation between the aryl ring and the heterocycle. The resulting crude product is then subjected to recrystallization, often using 1,4-dioxane, to achieve the high purity levels required for electronic grade materials. This meticulous synthetic pathway guarantees that the final compound possesses the precise structural attributes needed to function effectively as a hole blocking and light extracting layer.
How to Synthesize High-Performance Arylamine Compounds Efficiently
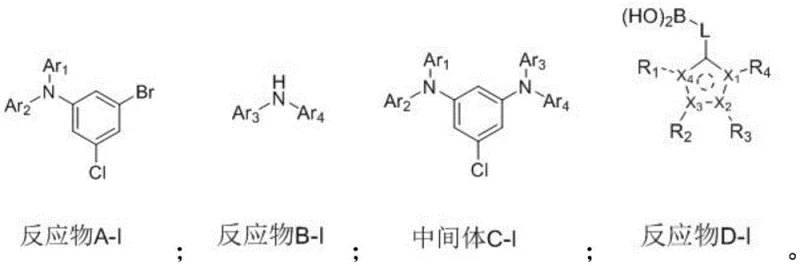
The preparation of these advanced OLED materials follows a standardized protocol designed to maximize yield while maintaining the stringent purity standards required for electronic applications. The process begins with the precise weighing and mixing of halogenated arylamine precursors and amine reactants under an inert nitrogen atmosphere to prevent oxidation. Catalysts and ligands are added in specific molar ratios to ensure complete conversion, and the reaction temperature is carefully ramped up to facilitate the coupling. After the initial reaction, the mixture undergoes a series of workup procedures including aqueous washing and organic extraction to remove inorganic salts and byproducts. The intermediate is then purified via column chromatography before proceeding to the second coupling step. For the full detailed standardized synthesis steps, please refer to the guide below.
- Perform Buchwald-Hartwig amination between a halogenated arylamine and a secondary amine using a palladium catalyst and phosphine ligand in toluene at elevated temperatures.
- Purify the resulting intermediate via hot filtration, aqueous washing, and column chromatography to remove catalyst residues and salts.
- Conduct a Suzuki coupling reaction between the purified intermediate and a heterocyclic boronic acid derivative using a palladium catalyst and carbonate base to finalize the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented arylamine technology offers substantial strategic advantages that extend beyond mere technical performance. The synthesis route utilizes widely available starting materials and standard palladium catalysis, which mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. This accessibility ensures a more stable and predictable supply of raw materials, allowing manufacturing partners to plan production schedules with greater confidence. Furthermore, the robustness of the chemical process means that it can be scaled from laboratory benchtop quantities to multi-ton annual production capacities without significant re-engineering of the process infrastructure. This scalability is crucial for meeting the fluctuating demands of the consumer electronics market, where production volumes can surge rapidly with the launch of new device generations.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize the number of purification steps required to achieve electronic-grade purity, which directly translates to lower processing costs. By eliminating the need for complex transition metal removal procedures often required in other catalytic systems, the overall cost of goods sold is significantly optimized. Additionally, the high yields reported in the patent examples suggest that raw material utilization is efficient, reducing waste and further driving down the unit cost of the final capping layer material. These efficiencies allow display manufacturers to incorporate high-performance materials without inflating the bill of materials for the final consumer device.
- Enhanced Supply Chain Reliability: The reliance on common solvents like toluene and ethanol, along with standard inorganic bases such as potassium carbonate, ensures that the supply chain is not vulnerable to shortages of specialized chemicals. This chemical commonality simplifies logistics and storage requirements, as these materials are handled routinely in fine chemical manufacturing facilities. Moreover, the stability of the intermediates allows for potential stockpiling or semi-finished goods inventory, providing a buffer against unexpected demand spikes. This reliability is a key factor for supply chain heads who prioritize continuity of supply to prevent production line stoppages in high-volume display fabrication plants.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale synthesis to kilogram and ton-scale production while maintaining consistent product quality. The use of recrystallization as a final purification step is particularly advantageous for large-scale operations, as it is more environmentally friendly and easier to manage than continuous chromatography. The ability to recover and recycle solvents further aligns with modern environmental compliance standards and sustainability goals. This combination of scalability and environmental stewardship makes the technology an attractive option for manufacturers looking to expand capacity while adhering to strict regulatory frameworks regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this arylamine technology in OLED manufacturing. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for R&D teams evaluating material compatibility and procurement officers assessing long-term supply viability. The information covers aspects ranging from molecular stability to process integration, providing a comprehensive overview for decision-makers.
Q: How does the five-membered heterocyclic structure improve OLED performance?
A: The incorporation of five-membered heterocyclic structures significantly increases the refractive index of the material in the visible light spectrum, which enhances light extraction efficiency when used as a capping layer.
Q: What are the thermal stability characteristics of these arylamine compounds?
A: These compounds exhibit high glass transition temperatures due to significant steric hindrance effects provided by the branched arylamine structure, ensuring superior film stability during device operation.
Q: Is the synthesis process scalable for industrial manufacturing?
A: Yes, the synthesis relies on standard palladium-catalyzed cross-coupling reactions using commercially available solvents and reagents, making it highly adaptable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylamine Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise and infrastructure necessary to bring complex synthetic routes like this arylamine technology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that the performance of OLED devices is critically dependent on the purity of the materials used; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards required for electronic applications. By partnering with us, you gain access to a supply chain that is not only robust but also deeply knowledgeable about the nuances of organic electroluminescent material synthesis.
We invite you to engage with our technical procurement team to discuss how we can support your specific material requirements and optimize your supply chain. Request a Customized Cost-Saving Analysis to understand how our manufacturing efficiencies can translate into tangible value for your organization. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality arylamine compounds reliably. Let us help you engineer the next generation of high-efficiency displays with materials that are proven, scalable, and commercially viable.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
