Advanced One-Pot Synthesis of p-Ferrocenyl Benzaldehyde for Commercial Scale-Up
Advanced One-Pot Synthesis of p-Ferrocenyl Benzaldehyde for Commercial Scale-Up
The landscape of organometallic chemistry has long sought efficient pathways to functionalize ferrocene derivatives, particularly for their applications in bioactive pharmaceutical intermediates and advanced materials. Patent CN103145767A introduces a transformative methodology for the preparation of p-ferrocenyl benzaldehyde, addressing critical bottlenecks in yield and operational complexity that have historically plagued this synthesis. By leveraging a modified electrophilic substitution strategy using phosphorus oxychloride in a dichloromethane medium, this innovation shifts the paradigm from laborious, low-efficiency reflux protocols to a streamlined, one-pot operation. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-value ferrocene-based building blocks, ensuring consistent quality and reduced manufacturing overheads through a robust, scientifically validated process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ferrocenyl benzaldehyde derivatives relied heavily on traditional Friedel-Crafts acylation or formylation techniques that often necessitated harsh reaction conditions and complex workup procedures. As detailed in the background art of the patent, prior methods involved refluxing ferrocene with phosphorus oxychloride followed by the addition of p-dimethylaminobenzaldehyde in anhydrous ether, a process that is not only time-consuming but also inherently dangerous due to the volatility and peroxide formation risks associated with ether solvents. Furthermore, the isolation of the target molecule required prolonged extraction periods spanning up to 48 hours and multiple washing steps, yet the final isolated yield remained dismally low at approximately 15.32%. Such inefficiencies translate directly into exorbitant production costs, excessive solvent waste, and unreliable batch-to-batch consistency, making conventional routes economically unviable for large-scale commercial adoption in the competitive fine chemicals market.
The Novel Approach
In stark contrast, the novel approach disclosed in CN103145767A utilizes a "one-pot cooking" strategy that dramatically simplifies the reaction engineering while simultaneously boosting productivity. By employing dichloromethane as the primary solvent and carefully controlling the addition sequence of reagents, the reaction proceeds smoothly at mild temperatures ranging from 10°C to 40°C, eliminating the need for energy-intensive reflux conditions. The process achieves a remarkable improvement in efficiency, with embodiment data demonstrating isolated yields soaring to between 76% and 85%, a five-fold increase over traditional methods. This leap in performance is achieved without compromising on purity, as the simplified post-treatment involving pH adjustment and recrystallization effectively removes unreacted starting materials and by-products, delivering a high-quality crystalline product suitable for downstream pharmaceutical applications with minimal purification burden.
Mechanistic Insights into POCl3-Mediated Electrophilic Substitution
The core of this technological breakthrough lies in the precise activation of the aromatic aldehyde via phosphorus oxychloride (POCl3), which acts as a potent dehydrating agent and electrophile generator in situ. In this mechanism, POCl3 reacts with the carbonyl oxygen of p-dimethylaminobenzaldehyde to form a highly reactive iminium salt intermediate, which subsequently serves as the electrophile attacking the electron-rich ferrocene ring. This electrophilic aromatic substitution is highly regioselective, favoring the para-position relative to the dimethylamino group due to steric and electronic factors, ensuring the formation of the desired p-ferrocenyl benzaldehyde isomer with high specificity. The use of dichloromethane as a solvent is critical here, as it provides excellent solubility for both the organometallic ferrocene and the polar intermediates, facilitating homogeneous reaction kinetics that are essential for maintaining high conversion rates and minimizing the formation of poly-substituted impurities.
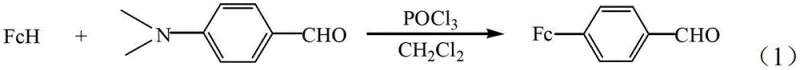
Furthermore, the patent emphasizes the importance of stoichiometric control in managing the impurity profile of the final product. By maintaining a molar ratio of p-dimethylaminobenzaldehyde to ferrocene between 1.5:1 and 3.5:1, the process ensures that ferrocene is the limiting reagent, thereby driving the reaction to completion and preventing the accumulation of unreacted ferrocene which can be difficult to separate due to similar solubility properties. The reaction temperature window of 10°C to 40°C is also mechanistically significant; it is sufficiently warm to overcome the activation energy barrier for the substitution but cool enough to suppress thermal degradation of the sensitive iminium intermediate or unwanted side reactions such as polymerization. This delicate balance of thermodynamic and kinetic parameters results in a clean reaction mixture that requires only standard aqueous workup and recrystallization to achieve pharmaceutical-grade purity, validating the robustness of the catalytic system for industrial deployment.
How to Synthesize p-Ferrocenyl Benzaldehyde Efficiently
To implement this high-yielding synthesis in a laboratory or pilot plant setting, operators must adhere to a strict sequence of reagent addition and temperature control to maximize the formation of the active electrophilic species. The process begins with the dissolution of the aromatic aldehyde in dry dichloromethane, followed by the controlled dropwise addition of phosphorus oxychloride to generate the reactive complex before introducing the ferrocene substrate. Maintaining the reaction mixture within the specified temperature range of 10°C to 40°C for a duration of 1.5 to 3 hours is crucial for ensuring complete conversion while avoiding thermal stress on the organometallic framework. For detailed operational parameters, including specific stirring rates, quenching protocols, and recrystallization solvent ratios, please refer to the standardized synthesis guide below which outlines the step-by-step execution of this patented methodology.
- Dissolve p-dimethylaminobenzaldehyde in dichloromethane and add phosphorus oxychloride dropwise with stirring.
- Introduce ferrocene to the mixture and maintain reaction temperature between 10°C and 40°C for 1.5 to 3 hours.
- Quench with water, adjust pH to neutral, extract with dichloromethane, and recrystallize the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound advantages in terms of cost structure and supply chain resilience for buyers of organometallic intermediates. The drastic increase in reaction yield from roughly 15% to over 80% fundamentally alters the cost of goods sold (COGS) by significantly reducing the amount of raw ferrocene and aldehyde precursors required to produce a single kilogram of the final active ingredient. This efficiency gain translates directly into substantial cost savings in fine chemical manufacturing, allowing procurement managers to negotiate more competitive pricing structures while maintaining healthy margins. Additionally, the elimination of hazardous ether solvents and the reduction of reaction time from days to mere hours streamline the production schedule, enabling faster turnaround times and improved responsiveness to fluctuating market demands for specialized pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The transition to a high-yield, one-pot process eliminates the need for expensive, time-consuming purification steps such as 48-hour extractions and sublimation, which are characteristic of the legacy ether-based method. By removing these labor-intensive unit operations, manufacturers can significantly lower utility consumption and labor costs, passing these efficiencies down the supply chain. The use of common, recyclable solvents like dichloromethane further enhances the economic viability of the process, reducing the total solvent inventory costs and waste disposal fees associated with volatile ether handling, thereby creating a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The mild reaction conditions (10-40°C) and the use of stable, commercially available reagents reduce the risk of batch failures due to thermal runaway or reagent instability, ensuring a consistent and reliable supply of high-purity intermediates. Unlike processes requiring cryogenic conditions or strictly anhydrous environments that are prone to disruption, this method is robust against minor variations in ambient conditions, making it ideal for continuous manufacturing campaigns. This reliability minimizes the risk of stockouts for downstream drug manufacturers, securing the continuity of supply for critical therapeutic pipelines that depend on ferrocene-derived scaffolds for their biological activity.
- Scalability and Environmental Compliance: The simplified workup procedure, which involves straightforward filtration and pH adjustment rather than complex distillation or chromatography, makes this process exceptionally easy to scale from gram-scale R&D to multi-ton commercial production. The reduction in solvent volume and the avoidance of persistent organic pollutants associated with long-duration ether extractions align with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This scalability ensures that suppliers can rapidly ramp up production capacity to meet surging demand without the need for massive capital investment in specialized reactor infrastructure, offering a flexible and sustainable solution for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios or for quality assurance personnel establishing specification limits for incoming raw materials. The answers provided reflect the specific operational parameters and beneficial effects observed during the validation of this method, ensuring that stakeholders have accurate information for decision-making.
Q: What is the primary advantage of this synthesis method over conventional ether reflux techniques?
A: The patented method utilizes a one-pot procedure in dichloromethane at mild temperatures (10-40°C), achieving yields up to 85% compared to the traditional 15.32% yield obtained via ether reflux, significantly reducing raw material waste and energy consumption.
Q: How does the stoichiometry affect the purity of the final organometallic product?
A: Maintaining a molar ratio of p-dimethylaminobenzaldehyde to ferrocene between 1.5:1 and 3.5:1 ensures complete conversion of the ferrocene while minimizing side reactions, resulting in high-purity crystals after simple recrystallization.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process avoids extreme temperatures and hazardous solvents like anhydrous ether, utilizing standard dichloromethane workups that are easily scalable from laboratory to multi-ton industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Ferrocenyl Benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organometallic intermediates play in the development of next-generation therapeutics and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering p-ferrocenyl benzaldehyde with stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our capability to replicate and optimize the patented one-pot synthesis allows us to offer a product that combines superior quality with the economic benefits of a streamlined manufacturing process, positioning us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can specifically benefit your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to our optimized supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique application needs, ensuring that you secure a reliable source of high-purity intermediates that drives innovation and profitability in your organization.
