Advanced Co-immobilized Biocatalysis for Scalable Production of Chiral Statin Intermediates
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and cost-effective synthesis routes for high-value active pharmaceutical ingredients (APIs) has brought biocatalysis to the forefront of modern chemical manufacturing. Specifically, the production of chiral hydroxy esters, which serve as critical building blocks for statin-class hypolipidemic drugs, represents a significant challenge in process chemistry. A pivotal advancement in this domain is detailed in Chinese Patent CN111686809A, which discloses a novel carbonyl reductase and isopropanol dehydrogenase co-immobilized catalyst. This technology addresses the longstanding limitations of both traditional chemical reduction and earlier generation free-enzyme biocatalysis by offering a robust, reusable, and highly stereoselective solution for synthesizing (R)-3-hydroxy-5-hexenoate derivatives. The structural transformation involves the asymmetric reduction of a prochiral ketone substrate to a chiral alcohol with exceptional enantiomeric excess, a process that is fundamental to the quality and efficacy of downstream statin medications.

For R&D directors and process chemists evaluating reliable pharmaceutical intermediate suppliers, the implications of this patent are profound. The target molecule, (R)-3-hydroxy-5-hexenoate, acts as a versatile precursor in the total synthesis of various cholesterol-lowering agents. Achieving high optical purity at this early stage is non-negotiable, as impurities can propagate through the synthesis tree, complicating purification and jeopardizing regulatory approval. The disclosed technology leverages a synergistic dual-enzyme system embedded within a protective polymeric matrix, ensuring that the biocatalytic performance remains consistent even under industrial processing conditions. This approach not only guarantees the stringent purity specifications required for global markets but also simplifies the overall process flow by integrating cofactor regeneration directly into the catalyst structure, thereby reducing the complexity of reaction monitoring and control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydroxy esters like (R)-3-hydroxy-5-hexenoate has relied heavily on chemical asymmetric reduction methods, which often impose severe constraints on manufacturing feasibility. Traditional chemical routes frequently necessitate extreme reaction conditions, such as cryogenic temperatures ranging from -20°C to -50°C and the use of high-pressure hydrogen gas, as documented in prior art like US Patent 6355822 and EP1176135. These harsh parameters not only escalate energy consumption and equipment costs but also introduce significant safety hazards related to high-pressure operations and the handling of pyrophoric reducing agents. Furthermore, chemical catalysts based on transition metals often require rigorous downstream purification steps to remove trace metal residues, which is a critical quality attribute for pharmaceutical products. Even when shifting to earlier biocatalytic methods using free enzymes, manufacturers faced substantial hurdles regarding catalyst recovery; free enzymes exist in a soluble crude broth that is difficult to separate from the product, leading to complex work-up procedures involving extensive extraction and centrifugation, ultimately driving up production costs and waste generation.
The Novel Approach
The innovation presented in patent CN111686809A fundamentally reshapes this landscape by introducing a co-immobilized enzyme catalyst that combines the specificity of biocatalysis with the practicality of heterogeneous catalysis. By co-embedding carbonyl reductase and isopropanol dehydrogenase within a solid-phase carrier derived from polyvinyl alcohol and polyethylene glycol, the inventors have created a physical form of the catalyst that is easily separable from the reaction mixture via simple filtration. This solid-state configuration eliminates the need for complex protein precipitation or membrane filtration steps typically associated with free enzyme processes. Moreover, the immobilization matrix provides a microenvironment that stabilizes the enzyme structures against thermal and pH stress, allowing the reaction to proceed efficiently at mild temperatures between 15°C and 35°C and neutral pH levels. This shift from extreme chemical conditions to mild biological ones, coupled with the ease of catalyst recovery, represents a paradigm shift in cost reduction in API manufacturing, enabling safer, greener, and more economically viable production scales.
Mechanistic Insights into Dual-Enzyme Cofactor Regeneration
At the heart of this technology lies a sophisticated enzymatic cascade that elegantly solves the economic bottleneck of cofactor dependency. The primary reaction is driven by carbonyl reductase, which catalyzes the stereoselective reduction of the ketone substrate (II) to the chiral alcohol product (I). This reduction requires the reducing equivalent NADPH, which is stoichiometrically consumed in the process. In a standard free-enzyme setup, supplying sufficient NADPH would be prohibitively expensive. However, this patent integrates a second enzyme, isopropanol dehydrogenase, directly into the same immobilized particle. This secondary enzyme utilizes inexpensive isopropanol as a sacrificial electron donor to oxidize the spent cofactor NADP+ back into the active NADPH form. This creates a closed-loop catalytic cycle where the cofactor is continuously regenerated in situ, allowing a catalytic amount of NADP+ to drive the conversion of large quantities of substrate.
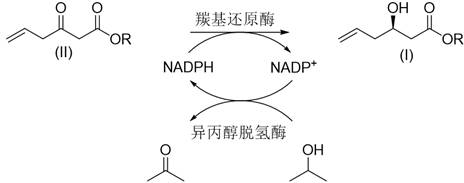
The physical proximity of the two enzymes within the PVA/PEG matrix likely enhances the efficiency of this cofactor shuttle, minimizing diffusion limitations and ensuring rapid turnover. From a mechanistic standpoint, the immobilization also restricts the conformational flexibility of the protein chains, which reduces the entropy of unfolding and thereby increases the activation energy required for denaturation. This explains the observed improvements in thermal stability, where the immobilized enzymes retain activity at temperatures up to 80°C, far exceeding the tolerance of their free counterparts. For technical teams focused on impurity control, this stability is crucial; it prevents the release of proteolytic fragments or denatured protein aggregates into the product stream, simplifying the purification profile and ensuring a cleaner final API. The result is a high-purity pharmaceutical intermediate produced with minimal byproduct formation, aligning perfectly with the rigorous quality standards of the global pharmaceutical supply chain.
How to Synthesize (R)-3-Hydroxy-5-Hexenoate Efficiently
Implementing this biocatalytic route requires precise control over the preparation of the immobilized matrix to ensure optimal enzyme loading and mass transfer characteristics. The process begins with the formulation of the polymer support, followed by the incorporation of the biocatalytic components under mild conditions that preserve enzyme activity. Once prepared, the solid catalyst can be deployed in standard stirred-tank reactors, offering a seamless transition from laboratory optimization to pilot and commercial production. The operational simplicity of this method allows for significant reductions in processing time and labor intensity compared to traditional batch processes. For detailed procedural specifics regarding buffer compositions, drying times, and reactor configurations, please refer to the standardized synthesis guide below.
- Prepare an aqueous solution of polyvinyl alcohol and polyethylene glycol, heating until clear and cooling below 50°C.
- Mix crude carbonyl reductase and isopropanol dehydrogenase enzyme solutions into the polymer matrix at optimized volume ratios.
- Drop-cast the mixture onto a film and dry at 35-40°C to form the solid co-immobilized catalyst beads.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this co-immobilized catalyst technology translates into tangible strategic advantages that extend beyond mere technical performance. The shift from single-use free enzymes to a reusable solid catalyst fundamentally alters the cost structure of the manufacturing process. By enabling the recovery and repeated use of the biocatalyst over multiple batches, the effective cost per kilogram of the enzyme contribution is drastically reduced. This reusability also mitigates the risk of supply disruptions associated with the frequent procurement of fresh enzyme batches, thereby enhancing supply chain reliability. Furthermore, the elimination of extreme reaction conditions removes the need for specialized cryogenic equipment or high-pressure vessels, allowing production to occur in standard glass-lined or stainless steel reactors available in most multipurpose facilities. This flexibility facilitates faster technology transfer and reduces capital expenditure requirements for scaling up production capacity.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive stoichiometric reagents and the maximization of catalyst utility. Since the cofactor NADPH is regenerated internally using low-cost isopropanol, there is no need for continuous dosing of expensive nucleotides, which traditionally represents a significant variable cost in bioreductions. Additionally, the ability to reuse the immobilized catalyst for numerous cycles means that the initial investment in enzyme production is amortized over a much larger volume of product output. The simplified downstream processing, which replaces complex extractions with straightforward filtration, further reduces solvent consumption and waste disposal costs, contributing to substantial overall cost savings without compromising yield or quality.
- Enhanced Supply Chain Reliability: Stability is a critical factor in maintaining a consistent supply of critical intermediates. The co-immobilized catalyst exhibits superior storage stability compared to liquid enzyme preparations, retaining high activity even after extended periods at ambient temperatures. This robustness simplifies logistics, as the catalyst does not require strict cold-chain transportation or frozen storage, reducing the risk of spoilage during transit. For supply chain planners, this means longer shelf lives and greater flexibility in inventory management. The consistent performance of the catalyst across different batches ensures predictable production schedules, minimizing the risk of delays caused by catalyst failure or variability, which is essential for meeting the just-in-time delivery demands of major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling biocatalytic processes often faces challenges related to oxygen transfer, mixing, and heat removal, but the solid nature of this catalyst simplifies these engineering constraints. The heterogeneous system allows for easy separation of the biocatalyst from the product stream, facilitating continuous flow processing options that are inherently more scalable than batch operations. From an environmental perspective, the process operates in aqueous buffers at neutral pH, generating significantly less hazardous waste compared to chemical reduction methods that produce heavy metal sludge or require harsh acidic/basic workups. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this co-immobilized system is essential for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding the operational parameters, stability profiles, and substrate compatibility of the catalyst. These insights are derived directly from the experimental data and embodiments described in the patent literature, providing a factual basis for decision-making. By clarifying these aspects, we aim to facilitate a smoother evaluation process for technical teams assessing the feasibility of this route for their specific portfolio of compounds.
Q: How does co-immobilization improve enzyme stability compared to free enzymes?
A: Co-immobilization within a PVA/PEG matrix restricts enzyme conformational mobility, protecting the active sites from thermal denaturation and pH fluctuations, thereby significantly extending operational lifespan.
Q: What is the primary advantage of the dual-enzyme system in this patent?
A: The system couples carbonyl reductase with isopropanol dehydrogenase to internally regenerate the expensive NADPH cofactor using cheap isopropanol, eliminating the need for external cofactor dosing.
Q: Can this catalyst be reused for multiple batches?
A: Yes, the solid nature of the catalyst allows for simple filtration and recovery, enabling reuse for over 18 cycles while maintaining high conversion rates above 85%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Hydroxy-5-Hexenoate Supplier
As the demand for high-quality statin intermediates continues to grow, partnering with a manufacturer that possesses deep expertise in advanced biocatalysis is crucial for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging proprietary immobilization techniques to deliver superior chiral building blocks. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required by global regulatory authorities. Our commitment to quality assurance ensures that our clients receive materials that are not only chemically pure but also consistent in their stereochemical profile, minimizing risks in downstream synthesis.
We invite pharmaceutical companies and contract manufacturing organizations to explore how this co-immobilized catalyst technology can optimize their supply chains and reduce overall production costs. By collaborating with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us help you engineer a more resilient and cost-effective supply chain for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
