Scalable Synthesis of Broad-Spectrum Triazole Schiff Base Intermediates for Modern Drug Discovery
Introduction to Next-Generation Antibacterial Scaffolds
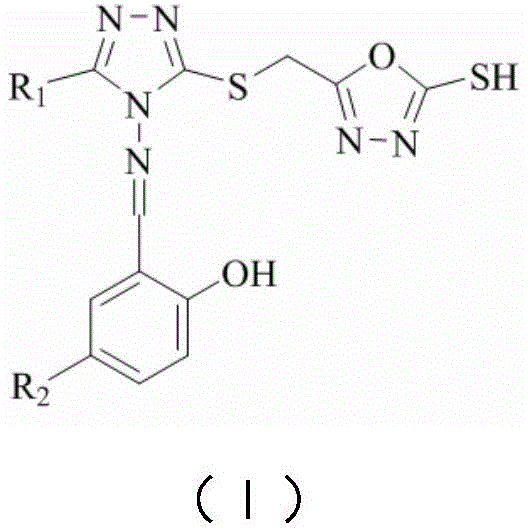
The escalating crisis of bacterial resistance to conventional antibiotics has necessitated the urgent development of novel therapeutic agents with distinct mechanisms of action. Patent CN107935950B addresses this critical challenge by disclosing a class of 1,3,4-oxadiazole modified 1,2,4-triazole Schiff base compounds. These molecules are engineered based on the principle of biological activity superposition, strategically assembling an 'o-hydroxyphenyl' molecular fragment with '1,2,4-triazole' and '1,3,4-oxadiazole' heterocycles. This rational design aims to introduce two or more heterocyclic active centers simultaneously into the drug molecule, thereby significantly enhancing biological potency. For R&D directors and procurement specialists in the pharmaceutical sector, this technology represents a promising avenue for developing high-purity pharmaceutical intermediates capable of combating resistant strains of Escherichia coli, Staphylococcus aureus, and Candida albicans.
The significance of this patent lies not only in its biological potential but also in its practical manufacturability. Unlike many complex heterocyclic syntheses that require exotic reagents or cryogenic conditions, this method employs relatively mild reaction parameters and accessible starting materials. The preparation process is described as simple and easy to implement, with yields reaching 40-60 percent, making it suitable for the requirement of large-scale industrial production. This balance between structural complexity and process simplicity is exactly what supply chain heads look for when evaluating new candidates for commercial scale-up of complex polymer additives or pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing bioactive heterocycles often suffer from significant drawbacks that hinder their commercial viability. Many conventional routes rely on harsh reaction conditions, such as strong acids or high temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, older methodologies frequently involve the use of toxic heavy metal catalysts or expensive coupling reagents, which not only drive up the cost of goods sold (COGS) but also create substantial environmental burdens regarding waste disposal and solvent recovery. In the context of antibacterial drug development, where purity is paramount, these traditional methods often require extensive and costly purification steps, such as preparative HPLC, to remove trace metal residues and side products, thereby extending lead times and reducing overall process efficiency.
The Novel Approach
The methodology outlined in CN107935950B offers a transformative alternative by utilizing a streamlined, three-step synthetic pathway that circumvents these traditional bottlenecks. The novel approach leverages a hydrazinolysis reaction followed by a cyclization with carbon disulfide and a final condensation step, all performed in common protic solvents like methanol and ethanol. This strategy eliminates the need for transition metal catalysts entirely, relying instead on basic reagents like potassium hydroxide (KOH) and hydrazine hydrate. By avoiding heavy metals, the process inherently simplifies the downstream purification workflow, as there is no need for specialized metal scavenging resins or rigorous testing for residual catalysts. This results in a cleaner crude product profile and a more robust process that is easier to control and scale, directly addressing the pain points of cost reduction in API manufacturing.
Mechanistic Insights into Heterocyclic Assembly and Bioactivity
The core innovation of this technology rests on the strategic fusion of pharmacophores known for their antimicrobial properties. The 1,2,4-triazole ring is a well-established motif in medicinal chemistry, recognized for its ability to interfere with fungal ergosterol synthesis and bacterial enzyme function. By modifying this core with a 1,3,4-oxadiazole ring via a sulfanyl linker, the molecule gains additional metabolic stability and hydrogen-bonding capabilities. The introduction of the Schiff base linkage (C=N) with an o-hydroxyphenyl group further enhances the molecule's ability to chelate metal ions within bacterial active sites, potentially disrupting essential metalloenzymes. This multi-target approach is designed to overcome single-point mutations that typically confer resistance, providing a broader spectrum of activity.
From a mechanistic synthesis perspective, the formation of the 1,3,4-oxadiazole ring is a critical step that dictates the overall success of the route. The reaction involves the nucleophilic attack of the hydrazide nitrogen on the electrophilic carbon of carbon disulfide, followed by intramolecular cyclization and dehydration. This transformation is highly sensitive to reaction conditions, particularly pH and temperature. The patent specifies a controlled addition of base and careful temperature management (40-80°C) to ensure complete conversion while minimizing the formation of polymeric byproducts. Understanding these mechanistic nuances is vital for process chemists aiming to optimize the reaction for kilogram-scale production, ensuring consistent quality and high-purity triazole Schiff base compounds.
How to Synthesize Triazole Schiff Base Compounds Efficiently
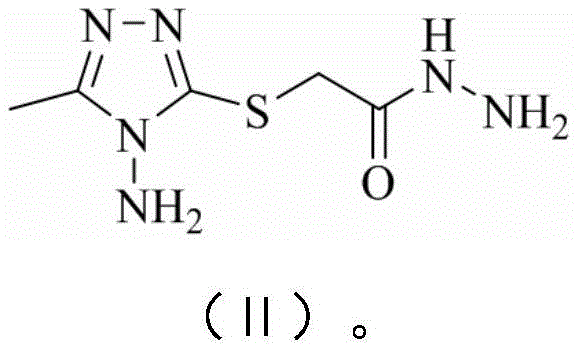
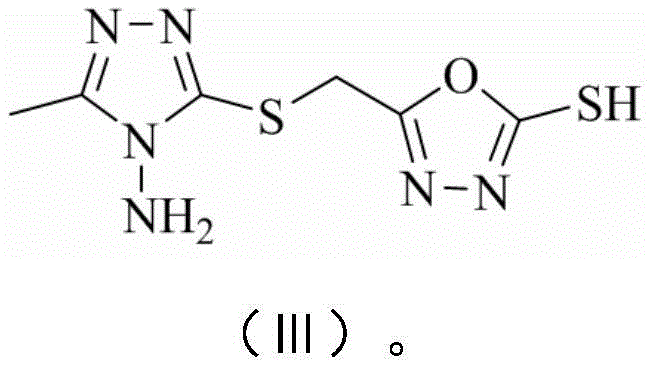
The synthesis of these valuable intermediates follows a logical progression that maximizes atom economy and operational simplicity. The process begins with the preparation of a key hydrazide intermediate, which serves as the nucleophilic precursor for the subsequent heterocycle formation. This is followed by the construction of the oxadiazole ring, creating the dual-heterocycle core. Finally, the diversity of the library is achieved through the condensation with various substituted salicylaldehydes, allowing for the fine-tuning of physicochemical properties such as lipophilicity and solubility. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, solvent volumes, and workup procedures required to achieve the reported yields.
- Perform hydrazinolysis of 3-methyl-4-amino-5-ethoxycarbonylmethylthio-1,2,4-triazole with hydrazine hydrate in methanol to obtain the acetic acid hydrazide intermediate.
- React the hydrazide intermediate with carbon disulfide (CS2) in the presence of a base like KOH in ethanol to form the 1,3,4-oxadiazole-2-thiol core.
- Condense the oxadiazole-thiol intermediate with various substituted salicylaldehydes in ethanol under acidic conditions to yield the final Schiff base compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The reliance on commodity chemicals and the absence of proprietary catalysts significantly de-risk the supply chain, ensuring continuity of supply even in volatile market conditions. The simplicity of the workup procedures—primarily involving filtration and recrystallization—reduces the demand for specialized equipment and skilled labor, translating into lower operational expenditures. Furthermore, the use of ethanol and methanol as primary solvents aligns with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, bulk-available solvents like ethanol and methanol drastically reduce the raw material costs. Additionally, the simplified purification process, which avoids complex chromatographic separations in favor of crystallization, lowers the processing time and energy consumption. This streamlined approach ensures that the cost of goods remains competitive, providing substantial cost savings compared to traditional multi-step heterocyclic syntheses that require rigorous purification.
- Enhanced Supply Chain Reliability: The starting materials, including hydrazine hydrate, carbon disulfide, and various salicylaldehydes, are widely produced commodities with stable global supply chains. This reduces the risk of bottlenecks associated with sourcing niche reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, further enhances reliability by minimizing batch failures and ensuring consistent output, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on multi-gram scales in the patent examples. The absence of heavy metals simplifies waste treatment, as the effluent does not require specialized heavy metal removal processes. This ease of waste management facilitates compliance with stringent environmental regulations, making the process suitable for commercial scale-up of complex heterocyclic intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial potential of this technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a transparent view of the compound's capabilities and the manufacturing process. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these intermediates into their drug discovery pipelines.
Q: What is the primary biological advantage of this triazole scaffold?
A: The compound integrates multiple heterocyclic active centers (1,2,4-triazole and 1,3,4-oxadiazole) with an o-hydroxyphenyl fragment, resulting in synergistic broad-spectrum antibacterial activity against Gram-negative, Gram-positive bacteria, and fungi.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like ethanol and methanol, avoids expensive transition metal catalysts, and involves straightforward unit operations such as filtration and recrystallization, making it highly amenable to scale-up.
Q: What are the typical yields for this preparation method?
A: The patent reports overall yields ranging from 40% to 60% across different derivatives, which is considered robust for multi-step heterocyclic synthesis involving sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains in the development of next-generation therapeutics. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, ensuring that every batch of Triazole Schiff Base compounds meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify identity and purity, guaranteeing that the materials you receive are ready for immediate use in biological screening or further synthesis.
We invite you to collaborate with us to leverage this innovative technology for your drug discovery programs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and efficiency.
