Scalable Synthesis of 9-Aminoquinine Derivatives for High-Performance Organocatalysis
Scalable Synthesis of 9-Aminoquinine Derivatives for High-Performance Organocatalysis
The landscape of asymmetric organocatalysis has been significantly reshaped by the development of cinchona alkaloid-derived catalysts, particularly those based on the 9-aminoquinine and 9-aminocinchonidine scaffolds. Patent CN101857593A introduces a groundbreaking synthetic methodology that addresses critical bottlenecks in the production of these high-value pharmaceutical intermediates. This technology leverages a novel reductive amination strategy utilizing substituted salicylaldehydes, which not only simplifies the synthetic route but also imparts unique functional properties to the final catalyst structure. For R&D directors and procurement specialists in the fine chemical sector, understanding this innovation is paramount, as it offers a pathway to more efficient, cost-effective, and environmentally benign manufacturing processes for complex chiral amines used in drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of secondary amines from primary amines like 9-aminoquinine often involves multi-step sequences that require harsh reaction conditions or expensive coupling reagents. Conventional methods frequently necessitate the use of external acid catalysts to facilitate imine formation or subsequent activation steps, which introduces additional complexity to the downstream processing. The requirement for stoichiometric amounts of strong acids can lead to corrosion issues in large-scale reactors and generates significant acidic waste streams that require neutralization and disposal. Furthermore, traditional routes may lack the flexibility to introduce diverse functional groups at the benzylic position without compromising the integrity of the sensitive quinoline or quinuclidine moieties, limiting the structural diversity available for catalyst optimization.
The Novel Approach
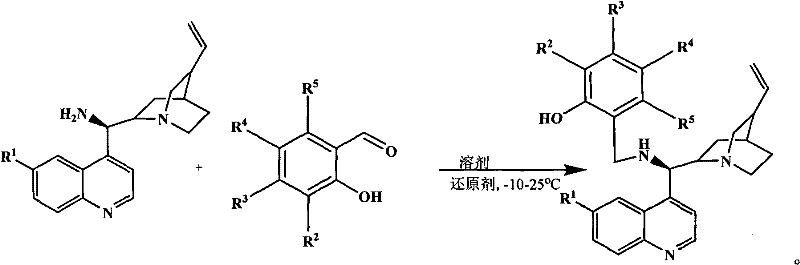
The methodology disclosed in the patent represents a paradigm shift by utilizing the intrinsic chemical properties of the starting materials to drive the reaction forward. By employing substituted or unsubstituted salicylaldehydes, the process capitalizes on the presence of the ortho-phenolic hydroxyl group. This structural feature allows for a seamless reductive amination under mild conditions, typically using methanol or ethanol as solvents and sodium borohydride as the reducing agent. The reaction proceeds smoothly at temperatures ranging from -10°C to 25°C, avoiding the thermal degradation often associated with more vigorous protocols. This approach not only streamlines the operation but also ensures that the resulting derivatives retain the crucial stereochemical information inherent in the cinchona backbone, which is essential for their function as chiral ligands.
Mechanistic Insights into Reductive Amination with Salicylaldehydes
The core mechanistic advantage of this synthesis lies in the dual functionality of the salicylaldehyde reactant. Initially, the primary amine of the 9-aminoquinine or 9-aminocinchonidine condenses with the aldehyde carbonyl group to form an imine intermediate. This step is facilitated by the solvent environment and the inherent nucleophilicity of the amine. Subsequently, the addition of sodium borohydride effects the reduction of the C=N double bond to a stable C-N single bond, yielding the secondary amine product. Crucially, the phenolic hydroxyl group remains intact throughout this transformation. In subsequent applications, such as the activation of alpha,beta-unsaturated aldehydes or ketones via iminium ion mechanisms, this phenolic proton serves as an internal Brønsted acid source. This eliminates the necessity for adding equimolar amounts of external acids, thereby simplifying the catalytic cycle and reducing the potential for side reactions caused by exogenous acidic species.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction conditions minimize the risk of epimerization at the chiral centers located at the 3, 4, 8, and 9 positions of the cinchona skeleton. Harsh acidic or basic conditions often promote racemization, which would render the final catalyst ineffective for asymmetric synthesis. By maintaining the pH within a neutral to slightly basic range during the reduction phase and utilizing the weak acidity of the phenol only when needed for catalysis, the process ensures high optical purity. The use of common solvents like methanol and ethanol further aids in the crystallization or chromatographic separation of the product from unreacted starting materials and borate byproducts, facilitating the attainment of stringent purity specifications required for pharmaceutical grade intermediates.
How to Synthesize 9-Aminoquinine Derivatives Efficiently
The practical implementation of this synthetic route is designed for operational simplicity and robustness, making it highly suitable for both laboratory scale optimization and industrial scale-up. The process begins with the dissolution of the amine and aldehyde components in a polar protic solvent under an inert atmosphere to prevent oxidation. Monitoring the reaction progress via thin-layer chromatography ensures complete conversion to the imine before the reducing agent is introduced, preventing the formation of over-reduced byproducts. The detailed standardized synthesis steps, including specific molar ratios, stirring times, and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety compliance.
- Dissolve 9-aminoquinine or 9-aminocinchonidine and substituted salicylaldehyde in methanol or ethanol under inert gas protection.
- Stir the mixture at temperatures between -10°C and 25°C until the primary amine is completely converted to the imine intermediate.
- Add sodium borohydride to reduce the imine, followed by aqueous quenching, solvent removal, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The reliance on commodity chemicals such as salicylaldehydes and sodium borohydride decouples the production from volatile markets for exotic reagents. Salicylaldehydes are widely produced industrial chemicals with stable pricing and abundant global supply chains, ensuring that raw material availability does not become a bottleneck for manufacturing schedules. Furthermore, the elimination of specialized acid additives and the use of common alcohol solvents reduce the overall bill of materials cost, contributing to a more competitive pricing model for the final active pharmaceutical ingredient or intermediate.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the atom economy of the reductive amination and the avoidance of expensive auxiliary reagents. By utilizing the phenolic hydroxyl group as an internal acid source for downstream catalysis, manufacturers save on the procurement costs of stoichiometric acid additives. Additionally, the simplified workup procedure, which involves standard aqueous quenching and solvent extraction, reduces the consumption of processing utilities and labor hours. The high yields reported, such as the 86% isolated yield in specific embodiments, mean that less raw material is wasted per unit of product, directly lowering the cost of goods sold and improving the overall margin profile for high-purity organocatalyst precursors.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Operating at near-ambient temperatures (-10°C to 25°C) reduces the energy demand for heating or deep cooling, making the process less susceptible to utility fluctuations. The flexibility in reaction time, ranging from 0.5 to 48 hours, allows production planners to optimize batch cycles according to facility capacity without compromising product quality. Since the starting materials are stable and commercially available in bulk quantities, the risk of supply disruption due to raw material shortages is minimized, ensuring consistent delivery timelines for reliable pharmaceutical intermediate supplier partners.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method aligns well with green chemistry principles. The use of ethanol or methanol as solvents is preferable to chlorinated solvents often used in traditional amine syntheses, reducing the environmental footprint and simplifying waste solvent recovery. The absence of heavy metal catalysts eliminates the need for costly and complex metal scavenging steps, which are often required to meet strict residual metal limits in drug substances. The process generates minimal hazardous waste, primarily consisting of aqueous borate salts which are easier to treat than organic sludge, facilitating regulatory compliance and reducing the costs associated with waste disposal and environmental remediation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and performance metrics of the synthesized derivatives. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these intermediates into their existing catalytic workflows.
Q: What is the key advantage of using salicylaldehyde in this synthesis?
A: The phenolic hydroxyl group in salicylaldehyde acts as an internal proton source, eliminating the need for additional acid additives in subsequent asymmetric catalytic reactions.
Q: What are the optimal reaction conditions for this reductive amination?
A: The reaction proceeds efficiently in methanol or ethanol at temperatures ranging from -10°C to 25°C, typically requiring 0.5 to 48 hours depending on the substrate.
Q: What yields can be expected from this synthetic method?
A: Experimental data indicates high yields, with specific examples achieving up to 86% isolated yield after column chromatography purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Aminoquinine Derivatives Supplier
As the demand for sophisticated chiral catalysts continues to grow in the pharmaceutical and agrochemical sectors, having a partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the structural integrity and enantiomeric excess of every batch, guaranteeing that our clients receive materials that perform consistently in sensitive asymmetric transformations.
We invite you to explore how our advanced manufacturing capabilities can support your R&D and production goals. By leveraging the efficiencies of the reductive amination process described in CN101857593A, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our supply chain solutions can enhance your operational efficiency and reduce time-to-market for your critical drug candidates.
