Scalable Synthesis of Proton Pump Inhibitor Zinc Salts for Global Pharmaceutical Supply Chains
Scalable Synthesis of Proton Pump Inhibitor Zinc Salts for Global Pharmaceutical Supply Chains
The global demand for effective gastric acid secretion inhibitors continues to drive innovation in the synthesis of proton pump inhibitor (PPI) intermediates. Patent CN100384839C introduces a robust and industrially viable method for preparing [(substituted pyridyl)methyl]sulfinyl-1H-benzimidazole compound zinc salts, which serve as critical active pharmaceutical ingredients or intermediates for drugs like omeprazole, pantoprazole, and lansoprazole. This technology addresses the longstanding need for stable, high-purity salt forms that offer improved solubility and bioavailability compared to traditional alkali metal salts. By utilizing organozinc reagents in an organic solvent medium, this novel approach simplifies the downstream processing typically associated with PPI salt formation, offering a reliable pharmaceutical intermediates supplier pathway for generic and branded drug manufacturers alike.
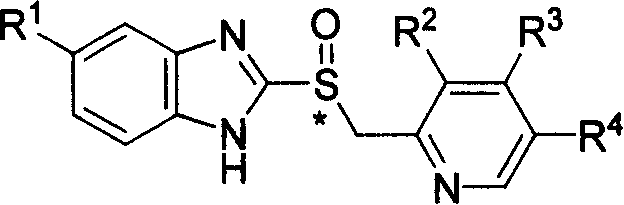
The core chemical architecture involves a benzimidazole ring linked via a sulfinyl methyl bridge to a substituted pyridine ring, as illustrated in the general structural formula. The versatility of this scaffold allows for the synthesis of various analogues by modifying the R groups, covering a wide spectrum of therapeutic agents within the PPI class. The transition from free base to zinc salt is not merely a formulation change but a strategic enhancement of the drug's physicochemical profile. Zinc salts are increasingly preferred in modern pharmacopeia due to their enhanced stability profiles and favorable toxicological properties, making this synthetic methodology a cornerstone for cost reduction in API manufacturing where salt selection impacts the entire lifecycle of the drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of PPI salts has relied heavily on the reaction of the free base with inorganic zinc salts in aqueous or mixed aqueous-organic media. These conventional pathways often suffer from significant drawbacks, including the introduction of water which can promote hydrolysis of the sensitive sulfinyl linkage, leading to sulfide or sulfone impurities that are difficult to remove. Furthermore, the use of inorganic salts frequently results in the co-precipitation of inorganic byproducts, necessitating extensive washing and purification steps that reduce overall yield and increase waste generation. The control of particle size and morphology is also challenging in aqueous systems, often resulting in crystalline forms with inferior dissolution rates compared to their amorphous counterparts, thereby limiting the bioavailability of the final dosage form.
The Novel Approach
In stark contrast, the methodology disclosed in CN100384839C employs an organozinc reagent, specifically diethylzinc, in an anhydrous organic solvent environment such as tetrahydrofuran. This non-aqueous approach fundamentally mitigates the risk of hydrolytic degradation during the salt formation step, preserving the integrity of the chiral sulfinyl center which is crucial for the drug's potency. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling cycles. The resulting zinc salt precipitates efficiently upon quenching with a protic solvent like water or alcohol, allowing for straightforward isolation via filtration. This streamlined process not only enhances the chemical purity of the product but also facilitates the formation of the desired amorphous state, which is recommended for optimal solubility.
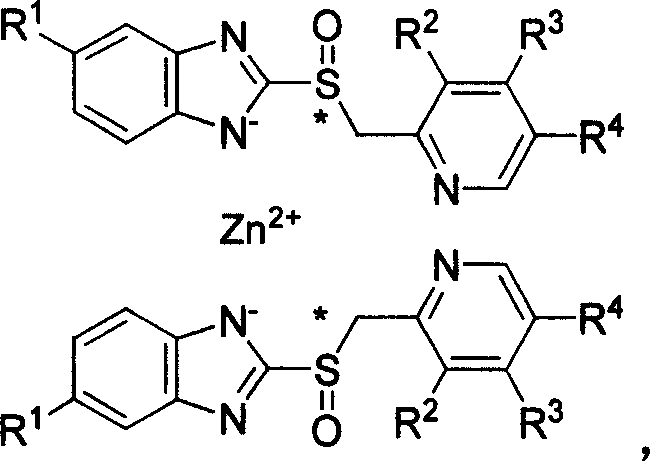
Mechanistic Insights into Organozinc-Mediated Salt Formation
The reaction mechanism involves the coordination of the organozinc species with the nitrogen atoms of the benzimidazole ring system. When the free base or its (S)-enantiomer is introduced to the organozinc reagent, a proton transfer occurs wherein the acidic proton on the benzimidazole nitrogen is abstracted by the alkyl group of the zinc reagent, releasing an alkane gas and forming a zinc-nitrogen bond. This direct metallation strategy is highly efficient and avoids the equilibrium issues often seen with inorganic salt metathesis reactions. The use of diethylzinc is particularly advantageous due to its high reactivity and solubility in common organic solvents, ensuring homogeneous reaction conditions that promote uniform nucleation of the zinc salt particles. This homogeneity is critical for achieving consistent batch-to-batch quality in a commercial setting.
Furthermore, the preservation of stereochemistry is a paramount concern for single-enantiomer PPIs like (S)-omeprazole. The mild conditions of this organozinc-mediated process ensure that the chiral center at the sulfur atom remains untouched, preventing racemization which would otherwise dilute the therapeutic efficacy of the drug. The patent data indicates that starting materials with high enantiomeric excess (ee) values translate directly to products with similarly high chiral purity, demonstrating the robustness of the method against epimerization. Additionally, the ability to tune the molar ratio of the organozinc reagent allows for precise control over the stoichiometry of the final salt, accommodating both 1:1 and 2:1 drug-to-metal ratios depending on the specific stability requirements of the target molecule.
How to Synthesize [(Substituted Pyridyl) Methyl] Sulfinyl-1H-Benzimidazole Zinc Salts Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating laboratory success to pilot and commercial production. The process begins with the dissolution of the PPI free base in a suitable organic solvent, followed by the controlled addition of the organozinc reagent under inert atmosphere conditions to manage the pyrophoric nature of dialkylzinc compounds. After a brief stirring period at ambient temperature, the reaction is quenched to induce precipitation of the zinc salt. This simplicity belies the sophistication of the chemistry, which balances reactivity with selectivity to deliver high-purity intermediates suitable for direct tableting or further formulation. For detailed operational parameters and safety guidelines regarding the handling of organometallic reagents, please refer to the standardized synthesis steps provided below.
- Dissolve the [(substituted pyridyl)methyl]sulfinyl-1H-benzimidazole free base or its (S)-enantiomer in an organic solvent such as tetrahydrofuran at room temperature.
- Slowly add an organozinc reagent, preferably diethylzinc, to the solution while maintaining stirring for 0.5 to 3 hours to facilitate the salt formation reaction.
- Quench the reaction mixture with water or a protic solvent, filter the resulting white solid precipitate, wash with methanol, and dry to obtain the high-purity zinc salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organozinc-based synthesis route offers compelling economic and logistical benefits. The elimination of aqueous reaction media significantly reduces the burden on wastewater treatment facilities, aligning with increasingly stringent environmental regulations and reducing the overall environmental footprint of the manufacturing site. The simplicity of the workup procedure, which relies primarily on filtration rather than complex extraction or chromatography, translates to shorter cycle times and higher throughput in multipurpose reactors. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a steady supply of critical PPI intermediates without the bottlenecks associated with traditional salt formation technologies.
- Cost Reduction in Manufacturing: The use of diethylzinc, while a specialized reagent, is offset by the drastic simplification of the downstream processing. By avoiding the need for extensive purification to remove inorganic salts and by minimizing solvent usage through efficient precipitation, the overall cost of goods sold is significantly optimized. The high yields reported in the patent examples suggest that raw material utilization is maximized, reducing waste disposal costs and improving the atom economy of the process. Furthermore, the ability to produce the amorphous form directly eliminates the need for additional milling or micronization steps often required to enhance the dissolution of crystalline salts, providing substantial cost savings in the finishing stages of production.
- Enhanced Supply Chain Reliability: The reliance on readily available organic solvents like tetrahydrofuran and commercially sourced organozinc reagents ensures a stable supply chain that is less susceptible to the geopolitical volatility often associated with rare earth metals or specialized inorganic catalysts. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require exotic equipment, means that production can be easily transferred between different manufacturing sites globally. This flexibility is crucial for maintaining business continuity and mitigating the risks of supply disruptions, ensuring that pharmaceutical customers receive their orders on time and to specification regardless of regional manufacturing constraints.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the exothermic nature of the reaction which is easily manageable at room temperature, removing the need for complex thermal control systems that often limit reactor capacity. The reduction in aqueous waste streams simplifies compliance with environmental discharge permits, a critical factor for long-term operational sustainability. Additionally, the high purity of the resulting zinc salt reduces the risk of batch failures due to out-of-specification impurities, thereby enhancing the overall reliability of the supply chain and reducing the inventory buffers that companies typically hold to guard against quality issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc salt preparation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding the nuances of chirality retention, physical form, and scalability is essential for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. We encourage technical teams to review these points closely to assess the fit for their specific product pipelines.
Q: Does this organozinc method preserve the chiral integrity of (S)-PPI enantiomers?
A: Yes, the method described in patent CN100384839C specifically demonstrates that reacting (S)-enantiomers with organozinc reagents retains high optical purity, with chiral HPLC purity reaching up to 99.5% in optimized examples, ensuring the pharmacological efficacy of the single enantiomer drug substance.
Q: What are the advantages of the amorphous zinc salt form over crystalline forms?
A: The patent recommends the amorphous form for these zinc salts because the finer powder structure inherent to the amorphous state offers potentially superior solubility characteristics compared to rigid crystalline lattices, which is critical for the bioavailability of oral proton pump inhibitor formulations.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely, the process operates at room temperature with short reaction times of 0.5 to 3 hours and utilizes simple separation techniques like filtration, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Proton Pump Inhibitor Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the competitive landscape of generic and specialty pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to commercial reality is seamless and efficient. We are committed to delivering high-purity proton pump inhibitors that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling sensitive organometallic chemistries allows us to safely and effectively implement the advanced zinc salt formation processes described in CN100384839C, providing our partners with a distinct competitive advantage in the marketplace.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your operation. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your trusted partner in the supply of high-quality PPI zinc salts, ensuring your production lines run smoothly and your patients receive the best possible medication.
