Revolutionizing 3,3'-Bisindolylmethane Production with Green Ion-Pair Catalysis
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access privileged molecular scaffolds. Patent CN115073351A introduces a groundbreaking preparation method for 3,3'-bisindolylmethane (BIM) compounds, a class of molecules renowned for their significant biological activities including antioxidant, anti-inflammatory, and anti-tumor properties. This technology leverages a novel ion-pair catalyst system, specifically Ph3C+[B(C6F5)4]-, to drive the Friedel-Crafts alkylation between indoles and carbonyl compounds. Unlike traditional methods that rely heavily on transition metals or stoichiometric amounts of strong acids, this approach operates efficiently in an aqueous medium. For R&D directors and procurement managers alike, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity, addressing the critical need for reliable pharmaceutical intermediate supplier solutions that align with modern environmental standards.
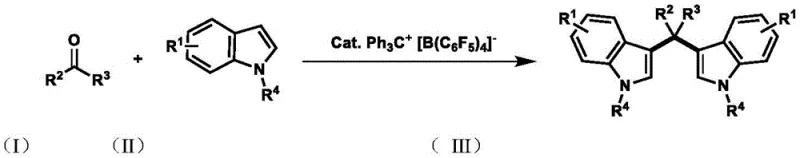
The limitations of conventional methods for synthesizing 3,3'-bisindolylmethanes have long plagued process chemists. Historically, these transformations were achieved using transition metal catalysts such as iron, copper, or gold complexes, or through the use of stoichiometric Brønsted acids. While effective in small-scale laboratory settings, these legacy processes introduce severe bottlenecks during scale-up. The primary concern is metal residue; even trace amounts of heavy metals in active pharmaceutical ingredients (APIs) can lead to regulatory rejection and necessitate expensive purification protocols involving chelating resins or repeated recrystallization. Furthermore, stoichiometric acid catalysis generates substantial quantities of chemical waste, creating significant environmental liabilities and increasing disposal costs. These factors collectively hinder cost reduction in pharmaceutical intermediate manufacturing and complicate the supply chain continuity for high-volume production.
In stark contrast, the novel approach detailed in the patent utilizes a metal-free ion-pair catalyst, Ph3C+[B(C6F5)4]-, which acts as a potent Lewis acid activator without introducing metallic contaminants. This catalyst system enables the reaction to proceed in pure water at a moderate temperature of 80°C, a condition that is both energy-efficient and operationally simple. The hydrophobic effect in water likely accelerates the reaction rate, while the stability of the catalyst ensures consistent performance over extended periods. By eliminating the need for anhydrous organic solvents and toxic metal reagents, this method drastically simplifies the downstream processing. The result is a streamlined workflow that enhances the commercial scale-up of complex pharmaceutical intermediates, offering a robust alternative for producing high-purity 3,3'-bisindolylmethanes with excellent atom economy.
Mechanistic Insights into Ion-Pair Catalyzed Friedel-Crafts Alkylation
The success of this methodology lies in the unique electronic properties of the trityl tetrakis(pentafluorophenyl)borate catalyst. The trityl cation (Ph3C+) serves as a strong electrophile activator, coordinating with the carbonyl oxygen of the aldehyde or ketone substrate to generate a highly reactive oxocarbenium ion intermediate. Simultaneously, the weakly coordinating [B(C6F5)4]- anion stabilizes this cationic species without nucleophilic interference, allowing the electron-rich C3 position of the indole ring to attack efficiently. This mechanism bypasses the need for harsh protonic acids that often lead to polymerization or decomposition of sensitive substrates. The compatibility with water is particularly noteworthy, as water typically deactivates many Lewis acids; however, this specific ion pair maintains its activity, likely due to the formation of a protected hydrophobic pocket around the active site. This deep mechanistic understanding assures R&D teams that the process is robust and predictable across a wide range of electronic variations in the substrate.
Impurity control is another critical aspect where this catalytic system excels. In metal-catalyzed reactions, side products often arise from metal-mediated oxidation or unintended C-H activation at non-target positions. With the Ph3C+[B(C6F5)4]- system, the reaction pathway is highly selective for the 3,3'-coupling, minimizing the formation of regioisomers or oligomeric byproducts. The mild reaction conditions further prevent the degradation of functional groups such as esters, nitriles, or halides, which are common in advanced intermediates. This high selectivity translates directly to a cleaner crude reaction profile, reducing the burden on purification teams and ensuring that the final product meets stringent purity specifications required for clinical applications. The ability to tolerate diverse functional groups expands the utility of this method for late-stage functionalization of complex drug candidates.
How to Synthesize Bis(1H-indol-3-yl)methane Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory equipment. The protocol involves charging a reaction vessel with the catalyst, indole, and carbonyl compound in specific molar ratios, followed by the addition of water. The mixture is then heated and stirred until conversion is complete. This simplicity lowers the barrier to entry for adoption in both academic and industrial settings. For detailed operational parameters, safety data, and specific workup procedures tailored to your facility, please refer to the standardized guide below.
- Charge a reaction vessel with the ion-pair catalyst Ph3C+[B(C6F5)4]- (0.08 mol%), substituted indole (2.2 equiv.), and carbonyl compound (1.0 equiv.).
- Add ultrapure water as the solvent (e.g., 2.5 mL for 0.5 mmol scale) and transfer the flask to a heating device.
- Heat the mixture to 80°C with rapid stirring for 8 hours, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free aqueous protocol offers transformative benefits for procurement and supply chain operations. The elimination of transition metals removes the necessity for costly metal scavenging steps and extensive analytical testing for residual metals, which are significant cost drivers in API manufacturing. This simplification of the purification train leads to substantial cost savings in manufacturing overheads. Additionally, the use of water as the primary solvent replaces expensive and hazardous organic solvents, reducing raw material costs and mitigating the risks associated with solvent storage and handling. These factors combine to create a more economically viable process that enhances the overall margin structure for high-value intermediates.
Supply chain reliability is significantly enhanced by the robustness of this catalytic system. The catalyst loading is remarkably low, down to 0.08 mol%, which means that a small quantity of catalyst can produce large volumes of product, reducing dependency on complex catalyst supply chains. The starting materials, primarily indoles and various aldehydes, are commodity chemicals with stable global availability, ensuring reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the reaction's tolerance to moisture and air simplifies logistics, as strict anhydrous conditions are not required. This resilience minimizes the risk of batch failures due to environmental fluctuations, providing supply chain heads with greater confidence in meeting delivery schedules and maintaining continuous production flows.
Scalability and environmental compliance are inherent strengths of this technology. The use of water aligns perfectly with green chemistry principles, drastically reducing the generation of volatile organic compounds (VOCs) and hazardous waste streams. This facilitates easier compliance with increasingly stringent environmental regulations across different jurisdictions. The exothermic nature of the reaction is manageable in water due to its high heat capacity, making the scale-up from gram to ton scale safer and more predictable. For organizations aiming to reduce their carbon footprint and achieve sustainability goals, this process offers a clear pathway to greener manufacturing without sacrificing efficiency or product quality, positioning companies as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new methodology is essential for stakeholders evaluating its potential impact on their projects. The following questions address common inquiries regarding catalyst performance, substrate compatibility, and process safety. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is the Ph3C+[B(C6F5)4]- catalyst preferred over transition metals?
A: Transition metal catalysts often leave toxic residues that require complex removal steps, which is critical for pharmaceutical intermediates. This non-metal ion-pair catalyst eliminates metal contamination risks entirely.
Q: Can this method be scaled up for industrial production?
A: Yes, the use of water as a solvent and mild reaction conditions (80°C) makes the process inherently safer and easier to scale compared to methods requiring anhydrous organic solvents or cryogenic temperatures.
Q: What is the substrate scope of this reaction?
A: The method demonstrates excellent functional group tolerance, successfully reacting various indoles with aldehydes, ketones, sugars, and even complex drug molecules like probenecid and pentoxifylline derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Bisindolylmethanes Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic methodologies like the one described in CN115073351A. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the absence of metal residues, which is critical for the pharmaceutical intermediates we produce. We are committed to leveraging this green catalytic technology to deliver high-quality BIM derivatives that meet the evolving needs of the global healthcare industry.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and purity targets. Please contact our technical procurement team to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and sustainability in the production of next-generation therapeutic agents.
