Advanced Synthesis of Perfluoro Unsaturated Nitrile Compounds for High-Performance Elastomer Cross-Linking
Advanced Synthesis of Perfluoro Unsaturated Nitrile Compounds for High-Performance Elastomer Cross-Linking
The development of high-performance fluorine-containing elastomers relies heavily on the availability of specialized cross-linking monomers that can withstand extreme chemical and thermal environments. Patent CN1078203C introduces a groundbreaking methodology for producing a novel class of perfluoro unsaturated nitrile compounds, specifically designed to serve as effective cross-linking site monomers. This technology addresses the critical need for monomers with flexible ether linkages, represented by the general formula CF2=CFO(CF2)nOCF(CF3)CN where n equals 2, 3, 4, or 5. By enabling precise control over the molecular architecture, this innovation allows manufacturers to tailor the curing characteristics and mechanical properties of fluoro-elastomers used in aerospace, automotive, and semiconductor applications. The patent details three distinct synthetic routes, offering flexibility in process design depending on available raw materials and desired throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the field of perfluoroalkyl vinyl ethers, such as the compounds disclosed in U.S. Patent No. 3,933,767 and U.S. Patent No. 4,281,092, often suffers from structural rigidity that limits their utility in specific elastomeric formulations. These conventional nitrile-containing monomers typically feature direct carbon-carbon linkages or shorter ether chains that do not provide sufficient flexibility or reactivity for advanced cross-linking requirements. Furthermore, traditional synthesis methods often struggle with side reactions at the vinyl group during functional group transformations, leading to lower yields and complex purification challenges. The inability to easily vary the chain length (n) in these older processes restricts the tunability of the final polymer properties, forcing formulators to compromise on performance metrics such as low-temperature flexibility or compression set resistance.
The Novel Approach
The novel approach presented in this patent overcomes these historical barriers by introducing a versatile synthetic strategy that incorporates a protected vinyl group during the critical functionalization steps. By initially reacting the perfluoro unsaturated carboxylic acid ester with halogens like bromine or chlorine, the reactive double bond is temporarily saturated, creating a stable halo-perfluoro intermediate. This strategic protection allows for subsequent harsh reactions, such as amidation and dehydration, to proceed without degrading the vinyl functionality. The result is a high-purity perfluoro unsaturated nitrile compound with a tailored ether chain length. This method ensures that the final monomer retains the essential CF2=CF- group required for copolymerization while introducing the nitrile group at the optimal position for cross-linking efficiency.
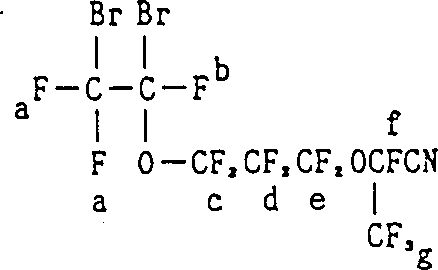
Mechanistic Insights into Halogenation and Dehydration Synthesis
The core mechanistic advantage of this process lies in the sequential manipulation of functional groups to preserve the integrity of the perfluorovinyl ether moiety. In the preferred Synthetic Route C, the reaction begins with the electrophilic addition of halogens across the carbon-carbon double bond of the starting ester. This step is crucial as it converts the electron-rich vinyl group into a chemically inert dibromo or dichloro segment, effectively shielding it from nucleophilic attack by ammonia in the next stage. The subsequent ammonolysis converts the ester moiety into a primary amide under mild conditions, typically below 30°C, minimizing thermal stress on the fluorinated backbone. Following this, a dehydration reaction using agents like Vanadium Pentoxide transforms the amide into a nitrile group. Finally, a dehalogenation step using zinc powder in a polar solvent restores the original double bond, yielding the target perfluoro unsaturated nitrile with high fidelity.
Impurity control is inherently built into this multi-step mechanism through the physical separation of intermediates. The halogenated intermediates possess distinct boiling points and solubility profiles compared to the starting esters and final nitriles, allowing for efficient purification via distillation or crystallization at each stage. For instance, the removal of excess halogen and hydrogen halide byproducts after the initial addition step prevents corrosion and side reactions in downstream units. Similarly, the dehydration step is conducted under vacuum to continuously remove water and drive the equilibrium towards the nitrile product, ensuring minimal residual amide content. This rigorous control over reaction pathways results in a final product with a clean impurity profile, which is essential for preventing premature curing or defects in the final fluoro-elastomer application.
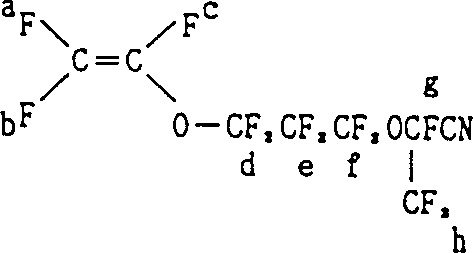
How to Synthesize Perfluoro Unsaturated Nitrile Efficiently
The synthesis of these specialized monomers requires precise adherence to reaction conditions to maximize yield and safety. The process generally involves handling reactive halogens and anhydrous ammonia, necessitating robust equipment and strict operational protocols. The patent outlines a clear progression from ester protection to nitrile formation and final deprotection. Operators must carefully monitor temperature gradients during the exothermic halogenation and ammonolysis steps to prevent runaway reactions. Detailed standardized operating procedures for each unit operation, including solvent recovery and waste neutralization, are essential for consistent production quality.
- React perfluoro unsaturated carboxylic acid ester with bromine or chlorine to form a halo-perfluoro saturated carboxylic acid ester, protecting the vinyl group.
- Convert the ester group to an amide group by reacting the halo-ester with ammonia under controlled temperature conditions.
- Perform dehydration of the amide to a nitrile using agents like Vanadium Pentoxide, followed by dehalogenation with Zinc to restore the vinyl double bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers significant strategic advantages in terms of cost stability and sourcing reliability. The ability to utilize standard fluorochemical feedstocks, such as perfluoro acyl fluorides and alcohols, reduces dependency on exotic or single-source precursors. The modular nature of the three synthetic routes (A, B, and C) allows manufacturing partners to select the most cost-effective pathway based on current raw material pricing and inventory availability. This flexibility acts as a hedge against market volatility, ensuring that production costs remain predictable even when specific reagent prices fluctuate. Furthermore, the high selectivity of the halogenation-protection strategy minimizes the formation of difficult-to-remove byproducts, reducing the burden on downstream purification units and lowering overall processing costs.
- Cost Reduction in Manufacturing: The elimination of complex cryogenic conditions for certain steps and the use of recyclable solvents like F-113 or perfluoro ethers contribute to substantial operational savings. By protecting the vinyl group, the process avoids the costly losses associated with polymerization or degradation that plague direct synthesis methods. This efficiency translates directly into a lower cost of goods sold, allowing for more competitive pricing in the high-value fluorine materials market without sacrificing margin.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including zinc powder, vanadium pentoxide, and common halogens, are widely available commodities with established global supply chains. This reduces the risk of supply disruptions that often accompany specialized catalysts or reagents. Additionally, the robustness of the intermediate compounds allows for safer storage and transport between production stages if a multi-site manufacturing strategy is employed, further enhancing supply continuity for critical customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as distillation and filtration that are easily expanded from pilot to commercial scale. The use of closed systems for ammonia and halogen handling ensures compliance with stringent environmental and safety regulations regarding toxic emissions. Waste streams, primarily consisting of zinc salts and aqueous washes, are manageable through standard industrial wastewater treatment protocols, facilitating easier permitting and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these perfluoro unsaturated nitrile compounds. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains and product portfolios. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What distinguishes this perfluoro unsaturated nitrile from prior art compounds?
A: Unlike previous compounds disclosed in U.S. Patents 3,933,767 and 4,281,092 which have rigid structures, this invention introduces a flexible ether linkage -(CF2)nO- where n=2-5, providing superior cross-linking sites for fluoro-elastomers.
Q: Why is the halogenation step critical in Synthetic Route C?
A: The halogenation protects the reactive vinyl double bond during the subsequent harsh amidation and dehydration steps, preventing polymerization or side reactions that would lower yield and purity.
Q: Can this process be scaled for industrial production of fluorine materials?
A: Yes, the process utilizes standard fluorochemical solvents like F-113 and common reagents like Zinc and Vanadium Pentoxide, making it highly adaptable for commercial scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoro Unsaturated Nitrile Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring this advanced synthesis technology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your demand for high-purity fluorine materials is met with consistency and precision. We understand that the quality of cross-linking monomers directly impacts the performance of the final elastomer, which is why we maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our commitment to quality assurance guarantees that every batch meets the exacting standards required for aerospace and semiconductor applications.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your next generation of high-performance fluoro-elastomers.
