Advanced One-Step Cyclization for High-Purity 2-Aryl-5-(trifluoromethyl) Pyrrole Intermediates
Advanced One-Step Cyclization for High-Purity 2-Aryl-5-(trifluoromethyl) Pyrrole Intermediates
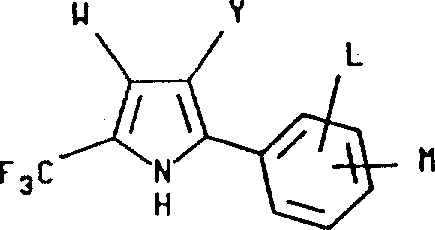
The development of efficient synthetic routes for fluorinated heterocycles remains a cornerstone of modern agrochemical and pharmaceutical innovation. Patent CN1030699C introduces a groundbreaking methodology for the preparation of 2-aryl-5-(trifluoromethyl) pyrrole compounds, which are highly valued for their insecticidal, acaricidal, and nematicidal properties. This technology addresses critical bottlenecks in traditional synthesis by enabling a direct, single-step cyclization between azlactone derivatives and alpha-halo-alpha,beta-unsaturated compounds. For R&D directors and process chemists, this represents a significant leap forward in constructing complex fluorinated scaffolds with high atom economy. The process not only simplifies the operational workflow but also ensures the production of high-purity intermediates essential for downstream biological activity testing and final formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted pyrrole rings, particularly those bearing electron-withdrawing trifluoromethyl groups, has been fraught with synthetic challenges. Traditional pathways often necessitate multi-step sequences involving harsh reaction conditions, expensive transition metal catalysts, or unstable intermediates that degrade rapidly. These conventional methods frequently suffer from poor regioselectivity, leading to difficult-to-separate isomeric mixtures that compromise the purity profile required for regulatory approval in agrochemical applications. Furthermore, the reliance on excessive reagents to drive reactions to completion generates substantial chemical waste, increasing both the environmental burden and the cost of goods sold. The inability to control the reaction kinetics precisely often results in prolonged processing times, creating bottlenecks in pilot plant operations and delaying time-to-market for new active ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in CN1030699C utilizes a highly efficient condensation strategy that merges an azlactone compound of Formula II with an alpha-halo-alpha,beta-unsaturated nitrile, ester, or nitro-compound of Formula III. This innovative route operates effectively in a single step within a polar solvent medium in the presence of a mild base. The reaction proceeds with remarkable speed and selectivity, often reaching completion within a single hour under reflux conditions. By employing stoichiometric amounts of reagents rather than large excesses, the process minimizes waste generation and simplifies the downstream purification protocol. The result is a robust, scalable synthesis that delivers the target 2-aryl-5-(trifluoromethyl) pyrrole structures in exceptional yields, often exceeding 90%, thereby setting a new standard for efficiency in fluorinated heterocycle manufacturing.
Mechanistic Insights into Base-Catalyzed Heterocyclic Cyclization
The success of this transformation hinges on the precise interplay between the nucleophilic azlactone species and the electrophilic unsaturated halide in a polar environment. Mechanistically, the base catalyst, such as triethylamine or pyridine, facilitates the deprotonation of the azlactone ring, generating a reactive nucleophilic center capable of attacking the beta-carbon of the alpha,beta-unsaturated system. This initial Michael-type addition is followed by an intramolecular cyclization and subsequent elimination of the halide leaving group, driven by the restoration of aromaticity in the newly formed pyrrole ring. The use of polar aprotic solvents like acetonitrile or dimethylformamide is critical, as they stabilize the charged transition states and intermediates involved in this cascade, ensuring rapid kinetics and high conversion rates that are unattainable in non-polar media.
Furthermore, the versatility of this mechanism allows for the incorporation of diverse functional groups at the 3-position of the pyrrole ring, including nitriles, esters, and nitro groups, simply by varying the starting unsaturated component. This modularity is invaluable for medicinal chemists seeking to optimize the structure-activity relationship (SAR) of potential agrochemical leads. The process also tolerates various substituents on the aryl ring attached to the 2-position, including halogens and alkyl groups, without significant loss in yield. Understanding these mechanistic nuances enables process engineers to fine-tune reaction parameters, such as temperature and base strength, to maximize throughput while maintaining strict control over impurity profiles, ensuring the final product meets the rigorous specifications demanded by global supply chains.
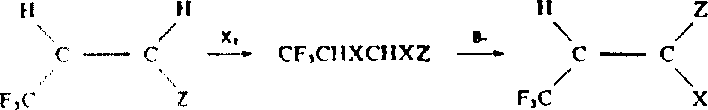
How to Synthesize 2-Aryl-5-(trifluoromethyl) Pyrrole Efficiently
Implementing this patented technology requires careful attention to solvent selection and reagent stoichiometry to replicate the high yields reported in the experimental data. The protocol involves dissolving the azlactone and the alpha-halo-unsaturated compound in a dry polar solvent, followed by the controlled addition of a tertiary amine base. The reaction mixture is then heated to reflux, where the cyclization occurs rapidly, often accompanied by the evolution of gas or precipitation of the product. Upon completion, the workup is straightforward, typically involving quenching with water and filtration or simple solvent extraction, avoiding the need for complex chromatographic purification on a commercial scale. This operational simplicity makes the route highly attractive for kilogram-to-tonne scale manufacturing.
- Combine the azlactone compound (Formula II) and the alpha-halo-alpha,beta-unsaturated nitrile, ester, or nitro-compound (Formula III) in a polar solvent such as acetonitrile or dimethylformamide.
- Add a stoichiometric amount of a base catalyst, such as triethylamine, pyridine, or sodium carbonate, to the reaction mixture while maintaining temperature control.
- Heat the mixture to reflux temperatures (approximately 65°C to 180°C) for a short duration (e.g., 1 hour), then cool, dilute with water, and isolate the solid product via filtration or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from multi-step, low-yielding processes to this streamlined one-pot reaction significantly reduces the consumption of raw materials and solvents, directly impacting the cost structure of the final intermediate. By eliminating the need for expensive transition metal catalysts and complex purification steps, manufacturers can achieve substantial cost savings in agrochemical manufacturing. The ability to use stoichiometric reagents rather than large excesses further optimizes material utilization, reducing waste disposal costs and aligning with increasingly stringent environmental regulations. This efficiency translates into a more competitive pricing model for the final active ingredient, providing a distinct market advantage.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of reaction steps drastically lower the operational expenditure associated with catalyst recovery and waste treatment. The use of common, commodity-grade solvents like acetonitrile and bases like triethylamine ensures that raw material costs remain stable and predictable. Additionally, the high yield achieved with stoichiometric reagents means less starting material is wasted, maximizing the return on investment for every batch produced. This lean manufacturing approach allows for significant margin improvement in the production of high-value fluorinated intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically azlactones and alpha-halo-unsaturated nitriles or esters, are readily available from established chemical suppliers, mitigating the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and minor variations in reagent quality, ensures consistent production output even in large-scale reactor environments. This reliability is crucial for maintaining continuous supply lines to downstream formulators and prevents costly production delays. Furthermore, the simplified workup procedure reduces the turnaround time between batches, enhancing overall plant throughput and responsiveness to market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram-scale laboratory experiments to larger pilot batches without loss of efficiency. The reduction in chemical waste and the avoidance of hazardous heavy metals simplify the environmental compliance landscape, reducing the regulatory burden on the manufacturing facility. The ability to isolate the product via simple filtration or extraction minimizes the volume of organic solvent waste generated, supporting sustainability goals. This green chemistry profile not only lowers disposal costs but also enhances the corporate social responsibility standing of the manufacturing entity in the eyes of global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl pyrrole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and optimization strategies. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production pipelines. The answers highlight the flexibility and robustness of the method across different substrate classes.
Q: Why is a polar solvent critical for this pyrrole cyclization?
A: According to the patent data, polar solvents like acetonitrile or DMF are essential because non-polar solvents (e.g., toluene) fail to produce the desired pyrrole ring even with base catalysis, likely due to insufficient stabilization of the ionic transition states required for ring closure.
Q: Can this reaction proceed without an added base catalyst?
A: While technically possible under extreme conditions (e.g., 52 hours reflux), omitting the base drastically reduces yield (down to 73% vs 97%) and significantly extends reaction time, making it commercially unviable compared to the base-catalyzed route.
Q: What are the primary applications of these trifluoromethyl pyrroles?
A: These compounds serve as potent active ingredients or key intermediates for insecticidal, acaricidal, and nematicidal agents, leveraging the metabolic stability provided by the trifluoromethyl group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl-5-(trifluoromethyl) Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-aryl-5-(trifluoromethyl) pyrrole meets the exacting standards required for agrochemical registration. Our commitment to process excellence allows us to deliver consistent quality while optimizing the cost-efficiency of the supply chain for our global partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. By partnering with us, you gain access to a reliable source of complex fluorinated building blocks that will accelerate your R&D timelines and strengthen your market position in the competitive agrochemical sector.
