Advanced Catalytic Synthesis of H Acid Monosodium Salt for Commercial Scale-up and Procurement Efficiency
The chemical industry constantly seeks methodologies that balance high-yield synthesis with environmental sustainability and equipment longevity, and the technical disclosures within patent CN103739524A offer a compelling solution for the production of H acid monosodium salt. This critical dye intermediate, chemically known as 1-amino-8-naphthol-3,6-disulfonate sodium, serves as a foundational building block for over ninety distinct dye varieties, including acid fuchsin and reactive brilliant reds, which are essential for the textile and leather processing sectors. The traditional manufacturing pathways for this compound have long been plagued by aggressive reaction conditions that accelerate equipment degradation and generate substantial hazardous waste, creating significant bottlenecks for procurement managers and supply chain directors aiming for stable long-term sourcing. The innovative approach detailed in this patent introduces a heteropolyacid-catalyzed sulfonation step that fundamentally alters the thermodynamic profile of the reaction, allowing for milder operating temperatures and reduced corrosion rates while maintaining exceptional conversion efficiency. By integrating this advanced catalytic system with continuous nitration and hydrogenation reduction technologies, the process achieves a level of operational sophistication that directly addresses the core concerns of R&D directors regarding purity and impurity profiles. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of H acid monosodium salt has relied on a multi-step batch process that begins with the sulfonation of naphthalene using concentrated sulfuric acid and oleum at excessively high temperatures. In these legacy protocols, the sulfonation reaction typically requires heating the mixture to ranges between 140°C and 160°C to drive the formation of the necessary trisulfonic acid intermediates, conditions that impose severe thermal stress on reactor vessels and piping systems. The aggressive nature of the reaction medium at these elevated temperatures leads to rapid corrosion of standard steel equipment, necessitating frequent maintenance shutdowns and the use of expensive corrosion-resistant alloys that drive up capital expenditure. Furthermore, the high thermal energy input often promotes the formation of undesirable by-products and isomers, complicating the downstream purification stages and resulting in a total recovery rate that historically hovers around a suboptimal 46% to 50%. The subsequent reduction step in conventional methods frequently employs iron powder, a technique that generates massive quantities of iron mud sludge, creating a heavy environmental burden and requiring complex waste treatment infrastructure that increases operational costs significantly. These cumulative inefficiencies create a fragile supply chain where yield fluctuations and equipment failures can lead to unpredictable lead times for downstream dye manufacturers.
The Novel Approach
The methodology presented in the patent data introduces a paradigm shift by incorporating a specific heteropolyacid catalyst into the initial sulfonation stage, which acts as a powerful promoter for the electrophilic substitution reaction on the naphthalene ring. This catalytic enhancement allows the sulfonation reaction to proceed effectively at a significantly reduced temperature range of 110°C to 125°C, thereby mitigating the corrosive impact on the reactor infrastructure and extending the operational lifespan of the production assets. By lowering the thermal threshold, the process also suppresses the formation of oxidative by-products, leading to a much cleaner reaction profile where the content of the desired 1,3,6-naphthalene trisulfonic acid can exceed 80% with total sulfonation recovery rates reaching up to 94%. 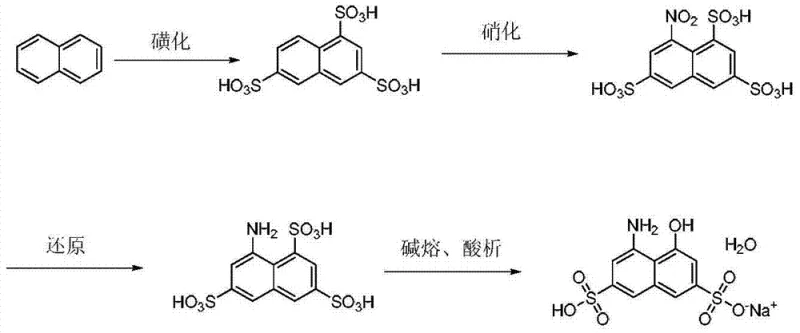
Furthermore, the transition from batch nitration to a continuous nitration process ensures that the exothermic reaction is managed with greater precision, maintaining the temperature strictly between 40°C and 60°C to prevent runaway reactions and ensure consistent quality. The replacement of iron powder reduction with catalytic hydrogenation using a nickel catalyst not only eliminates the generation of solid iron waste but also simplifies the workup procedure, as the catalyst can be filtered and potentially recycled. This holistic optimization of the synthetic route results in a final product purity exceeding 98%, providing a level of consistency that is critical for high-end dye applications where color strength and shade reproducibility are paramount for the end-user.
Mechanistic Insights into Heteropolyacid-Catalyzed Sulfonation
The core innovation driving the efficiency of this synthesis lies in the specific interaction between the heteropolyacid catalyst, such as molybdenum vanadium phosphoric acid (H4PMo11VO40), and the sulfuric acid medium during the sulfonation of refined naphthalene. Heteropolyacids are known for their strong Brønsted acidity and unique redox properties, which in this context serve to activate the sulfur trioxide species, making them more electrophilic and thus more reactive towards the aromatic naphthalene substrate at lower energy levels. This activation reduces the activation energy required for the sulfonation steps, allowing the first sulfonation to occur smoothly at 110°C to 125°C and the secondary sulfonation with sulfur trioxide to proceed efficiently at 135°C to 145°C, temperatures that are markedly lower than the 150°C to 160°C required in uncatalyzed systems. The catalyst loading is optimized to be between 0.5% and 5% of the naphthalene mass, a range that balances catalytic activity with economic feasibility, ensuring that the catalyst cost does not offset the savings gained from reduced energy consumption and equipment maintenance. The presence of the catalyst also appears to direct the regioselectivity of the sulfonation, favoring the formation of the 1,3,6-isomer over other potential isomers, which is crucial because only this specific isomer can be successfully converted into the target H acid structure through the subsequent nitration and reduction steps.
Impurity control is rigorously managed through the integration of continuous extraction and denitration steps immediately following the nitration reaction, which prevents the accumulation of unstable nitro compounds that could degrade product quality. The process utilizes an extraction agent, preferably N503, to separate the nitrated material from the acidic aqueous phase, allowing for the recovery of sulfuric acid which can be recycled back into the sulfonation stage, thereby closing the loop on acid consumption. The organic phase containing the nitro compound is then stripped with aqueous sodium hydroxide to isolate the nitro T acid, which is subsequently subjected to hydrogenation reduction under a controlled pressure of 25 to 35 atm. This hydrogenation step is critical for impurity management as it selectively reduces the nitro group to an amino group without affecting the sulfonic acid groups or the aromatic ring, a selectivity that is difficult to achieve with chemical reducing agents like iron or sulfides. The final alkali fusion step is conducted under pressure at 150°C to 190°C in the presence of methanol and sodium hydroxide, converting the amino T acid into the alkali melt which, upon acidification to pH 3-6, precipitates the H acid monosodium salt with high crystallinity and minimal inclusion of inorganic salts.
How to Synthesize H Acid Monosodium Salt Efficiently
Implementing this synthesis route requires precise control over reaction parameters and a robust understanding of the unit operations involved in handling corrosive acids and high-pressure hydrogenation systems. The process begins with the charging of refined naphthalene and the heteropolyacid catalyst into a reactor, followed by the gradual addition of sulfuric acid while maintaining the temperature within the narrow window of 110°C to 125°C to ensure complete melting and reaction without thermal degradation. Once the sulfonation is complete and verified via HPLC analysis to ensure high conversion to the trisulfonic acid, the material is transferred to the continuous nitration unit where it reacts with nitric acid at a controlled molar ratio of 1:1 to 1.2. The detailed standardized synthesis steps, including specific stirring rates, addition times, and safety protocols for handling sulfur trioxide and high-pressure hydrogen, are outlined in the technical guide below for engineering teams to review.
- Perform sulfonation of refined naphthalene with sulfuric acid and sulfur trioxide in the presence of a heteropolyacid catalyst at 110-125°C to obtain sulfonated material.
- Conduct continuous nitration of the sulfonated material with nitric acid at 40-60°C, followed by extraction and denitration to isolate nitro T acid.
- Execute hydrogenation reduction of nitro T acid using a nickel catalyst under pressure, followed by alkali fusion with methanol and sodium hydroxide, and final acidification to pH 3-6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational improvements that directly impact the bottom line and supply reliability. The reduction in reaction temperature and the elimination of iron powder sludge significantly lower the operational expenditure associated with energy consumption and waste disposal, allowing for a more competitive pricing structure without compromising on margin. The continuous nature of the nitration process enhances the throughput capacity of the manufacturing plant, enabling suppliers to respond more rapidly to fluctuating market demands and reducing the lead time for order fulfillment. Moreover, the reduced corrosion rate of the equipment means that production schedules are less likely to be interrupted by unplanned maintenance, ensuring a consistent and reliable flow of material to downstream dye manufacturers who depend on just-in-time inventory strategies.
- Cost Reduction in Manufacturing: The implementation of the heteropolyacid catalyst eliminates the need for excessive thermal energy input, resulting in substantial savings on steam and cooling utilities over the course of annual production cycles. By avoiding the use of iron powder for reduction, the facility removes the costly requirement for sludge treatment and disposal, which is a significant hidden cost in traditional dye intermediate manufacturing. The ability to recover and recycle sulfuric acid from the extraction phase further reduces the consumption of raw materials, driving down the variable cost per kilogram of the final product and enhancing overall profitability.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures high batch-to-batch consistency, reducing the risk of off-spec material that would otherwise require reprocessing or scrapping. Continuous processing capabilities allow for a steady output rate that smooths out supply volatility, making it easier for procurement teams to plan inventory levels and secure long-term contracts with confidence. The simplified purification steps reduce the overall cycle time from raw material input to finished goods, shortening the manufacturing lead time and improving the agility of the supply chain in response to urgent customer requests.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations that can be easily expanded from pilot scale to multi-ton commercial production without fundamental changes to the chemistry. The elimination of heavy metal waste and the reduction of acidic emissions align with increasingly stringent global environmental regulations, reducing the regulatory risk profile for manufacturing sites. This environmental compliance ensures long-term operational continuity, protecting the supply chain from potential shutdowns due to non-compliance penalties or environmental audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of H acid monosodium salt using this advanced catalytic methodology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide accurate guidance for technical evaluators. Understanding these details is crucial for assessing the feasibility of integrating this intermediate into your specific dye formulation or pharmaceutical synthesis pipeline.
Q: How does the heteropolyacid catalyst improve the sulfonation process compared to traditional methods?
A: The addition of a catalytic amount of heteropolyacid, such as molybdenum vanadium phosphoric acid, significantly enhances the activity of the sulfonation substrate. This allows the reaction to proceed at lower temperatures (110-125°C versus traditional 140-160°C), drastically reducing equipment corrosion and minimizing side reactions while improving the yield of naphthalene-1,3,6-trisulfonic acid.
Q: What are the environmental benefits of using hydrogenation reduction in this synthesis route?
A: By utilizing catalytic hydrogenation reduction with a nickel catalyst instead of traditional iron powder reduction, the process completely avoids the generation of iron mud. This elimination of solid waste significantly reduces the burden on wastewater treatment systems and aligns with stricter environmental compliance standards for modern chemical manufacturing facilities.
Q: Why is continuous nitration preferred for the commercial production of H acid intermediates?
A: Continuous nitration allows for precise control over reaction parameters such as temperature and molar ratios, leading to consistent product quality and higher safety standards. This approach reduces facility investment costs compared to batch processing and lowers the unit consumption of raw materials, thereby enhancing the overall economic efficiency of the supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable H Acid Monosodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a synthesis route is only as valuable as its ability to be executed reliably at scale, which is why we have invested heavily in mastering complex catalytic chemistries like the heteropolyacid-mediated sulfonation described in CN103739524A. Our engineering teams possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this low-temperature, low-corrosion process are fully realized in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of H acid monosodium salt meets the exacting standards required for high-performance dye applications, providing our partners with the consistency they need to maintain their own product quality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be leveraged to achieve a Customized Cost-Saving Analysis for your specific supply chain requirements. By collaborating with us, you can request specific COA data and route feasibility assessments that will demonstrate the tangible benefits of switching to a supplier who prioritizes advanced process chemistry and environmental stewardship. Let us help you secure a stable, high-quality supply of this critical intermediate while optimizing your overall procurement strategy for maximum efficiency and cost-effectiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
