Strategic Synthesis of Cefpodoxime Dippivoxil: Enhancing Impurity Control for Global Cephalosporin Supply Chains
The pharmaceutical industry's relentless pursuit of medication safety has placed impurity profiling at the forefront of quality control strategies, particularly for complex beta-lactam antibiotics like cephalosporins. Patent CN113999252A introduces a groundbreaking methodology for the directional synthesis of Cefpodoxime dippivoxil, a critical process-related impurity of the widely used third-generation oral cephalosporin, Cefpodoxime proxetil. This innovation addresses a significant gap in the current regulatory landscape, where the identification and quantification of unknown single impurities exceeding 0.1% are strictly mandated by major pharmacopoeias. The ability to synthetically produce this specific impurity with high fidelity allows manufacturers to establish robust analytical methods, ensuring that final drug products meet the rigorous safety standards required for global market approval. 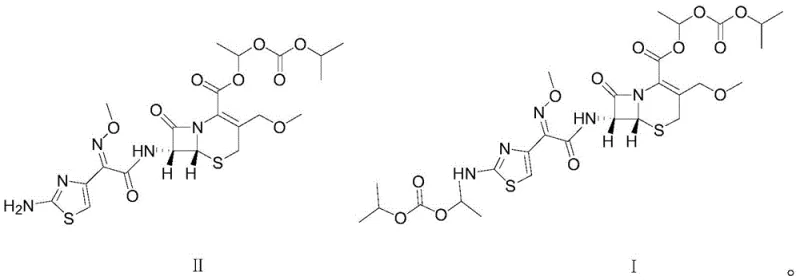 By providing a reliable source of this reference standard, the technology empowers quality assurance teams to detect trace contaminants that could otherwise compromise the therapeutic efficacy or safety profile of the active pharmaceutical ingredient.
By providing a reliable source of this reference standard, the technology empowers quality assurance teams to detect trace contaminants that could otherwise compromise the therapeutic efficacy or safety profile of the active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of specific cephalosporin impurities like Cefpodoxime dippivoxil has been fraught with significant technical and logistical challenges. In many legacy processes, these impurities were merely observed as minor byproducts during the bulk synthesis of the API, making their isolation extremely difficult and inefficient. Attempting to isolate such compounds from complex reaction mixtures often requires preparative high-performance liquid chromatography (HPLC), a technique that is notoriously expensive, time-consuming, and difficult to scale beyond milligram quantities. Furthermore, relying on random degradation or forced stress testing to generate impurities often results in a mixture of multiple degradation products, necessitating arduous separation steps that drastically reduce overall yield. This lack of a dedicated synthetic route means that supply is often inconsistent, leading to bottlenecks in method validation and stability studies for pharmaceutical developers who require gram-to-kilogram quantities of reference standards.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by offering a direct, one-step condensation route to synthesize Cefpodoxime dippivoxil with exceptional selectivity. Instead of relying on chance formation during API manufacturing, this approach utilizes Cefpodoxime proxetil as a starting material and reacts it with 1-iodoethyl isopropyl carbonate under precisely controlled conditions. 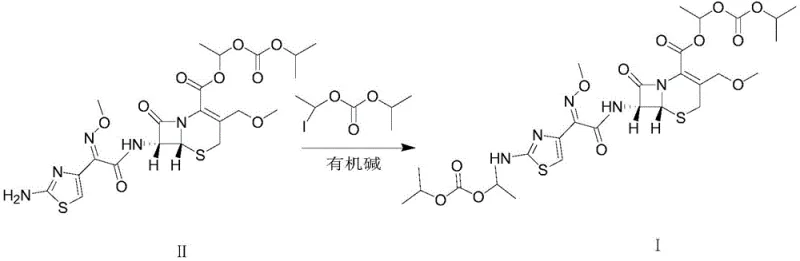 The key innovation lies in the modulation of the reaction environment, specifically maintaining a pH between 8.0 and 10.0 and a temperature range of 20-45°C. This delicate balance activates the nucleophilic amino group on the thiazole ring without compromising the integrity of the sensitive beta-lactam moiety. The result is a streamlined process that bypasses the need for complex purification, delivering the target impurity with a purity exceeding 93% directly after crystallization. This deterministic synthesis ensures a consistent, high-quality supply of the impurity standard, fundamentally changing how manufacturers approach impurity control for this class of antibiotics.
The key innovation lies in the modulation of the reaction environment, specifically maintaining a pH between 8.0 and 10.0 and a temperature range of 20-45°C. This delicate balance activates the nucleophilic amino group on the thiazole ring without compromising the integrity of the sensitive beta-lactam moiety. The result is a streamlined process that bypasses the need for complex purification, delivering the target impurity with a purity exceeding 93% directly after crystallization. This deterministic synthesis ensures a consistent, high-quality supply of the impurity standard, fundamentally changing how manufacturers approach impurity control for this class of antibiotics.
Mechanistic Insights into Organic Base-Catalyzed N-Alkylation
The core chemical transformation driving this synthesis is a nucleophilic substitution reaction where the primary amino group on the 2-aminothiazole ring of Cefpodoxime proxetil acts as the nucleophile. In the presence of an organic base, such as triethylamine or diisopropylamine, the amino group is effectively deprotonated or activated, increasing its electron density and nucleophilicity. This activated amine then attacks the electrophilic carbon of the 1-iodoethyl isopropyl carbonate, displacing the iodide ion. The choice of the alkylating agent is crucial; the 1-iodoethyl isopropyl carbonate moiety mimics the ester side chain found in the parent drug but attaches to the nitrogen rather than the carboxylic acid, creating the unique 'dippivoxil' structure. The reaction mechanism is highly sensitive to the basicity of the medium; if the pH is too low, the amine remains protonated and unreactive, while a pH that is too high risks hydrolyzing the beta-lactam ring or the existing ester groups, leading to unwanted degradation products.
Furthermore, the process incorporates a sophisticated workup strategy designed to maximize purity through selective solubility differences. Post-reaction, the mixture is treated with dilute acid, typically hydrochloric or sulfuric acid, to adjust the pH to an acidic range of 3.0-5.0. This step is mechanistically vital because it protonates any unreacted Cefpodoxime proxetil, converting it into a water-soluble salt that can be easily removed via aqueous extraction. In contrast, the target impurity, Cefpodoxime dippivoxil, remains in the organic phase due to its modified lipophilicity. Following the removal of the organic solvent, recrystallization using poor solvents like isopropyl ether or cyclohexane further refines the product lattice, excluding structurally similar byproducts. This multi-stage purification logic, embedded within the synthesis design, ensures that the final material meets the stringent purity requirements necessary for use as an analytical reference standard in regulated environments.
How to Synthesize Cefpodoxime Dippivoxil Efficiently
The synthesis of this critical impurity standard is designed for operational simplicity and robustness, making it suitable for both laboratory-scale reference material production and larger-scale manufacturing support. The process begins by dissolving the Cefpodoxime proxetil starting material in a benign, water-insoluble organic solvent such as dichloromethane or toluene, ensuring a homogeneous reaction medium. The subsequent addition of the alkylating agent and organic base must be carefully managed to maintain the optimal pH window, which drives the reaction forward while suppressing side reactions. For a comprehensive understanding of the precise stoichiometric ratios, temperature ramping profiles, and crystallization parameters required to replicate this high-yield process, please refer to the detailed technical guide below.
- Dissolve Cefpodoxime proxetil in a non-water-soluble organic solvent such as dichloromethane or toluene.
- Add 1-iodoethyl isopropyl carbonate and an organic base to maintain system pH between 8.0 and 10.0.
- Control reaction temperature at 20-45°C, followed by acid wash workup and recrystallization to achieve >93% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the production workflow, which translates directly into cost efficiency and supply stability. By eliminating the need for resource-intensive chromatographic separations and reducing the number of unit operations, the overall manufacturing footprint is significantly minimized. This leaner process not only lowers the cost of goods sold but also reduces the dependency on specialized equipment and consumables, making the supply of this critical impurity standard more resilient to market fluctuations. Furthermore, the use of common, commercially available solvents and reagents ensures that raw material sourcing remains straightforward and unaffected by geopolitical or logistical disruptions that often plague the supply of exotic catalysts or proprietary intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the elimination of expensive purification steps. Traditional methods often rely on preparative HPLC or multiple recrystallizations from exotic solvent systems, which incur high operational costs and solvent waste disposal fees. In contrast, this novel method achieves high purity through a simple acid-base extraction followed by a single crystallization step. The removal of unreacted starting material via aqueous washing is a low-cost operation that significantly enriches the product stream before the final isolation. Additionally, the reaction proceeds with high conversion efficiency under mild conditions, reducing energy consumption associated with heating or cooling. These factors collectively contribute to a substantially reduced cost structure, allowing for more competitive pricing of the reference standard without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical manufacturers who cannot afford delays in their quality control workflows. This synthesis route enhances reliability by utilizing Cefpodoxime proxetil, a widely produced bulk intermediate, as the starting material. Unlike custom-synthesized precursors that may have long lead times, Cefpodoxime proxetil is readily available from multiple global suppliers, mitigating the risk of raw material shortages. The robustness of the reaction conditions—operating effectively at ambient to moderately elevated temperatures—further ensures that the process can be executed consistently across different manufacturing sites without requiring highly specialized infrastructure. This flexibility allows for rapid scale-up from gram to kilogram quantities, ensuring that demand spikes during regulatory filing periods or stability testing phases can be met without delay.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with modern green chemistry principles. The solvents employed, such as ethyl acetate, toluene, and dichloromethane, are standard industrial solvents with well-established recovery and recycling protocols. The absence of heavy metal catalysts or toxic reagents simplifies waste treatment and reduces the environmental burden of the manufacturing process. The workup procedure, which relies on phase separation and crystallization rather than column chromatography, generates significantly less solid waste and solvent volume per kilogram of product. This makes the process inherently easier to scale to multi-kilogram or tonne levels, as the engineering challenges associated with heat transfer and mixing in large reactors are minimal. Consequently, manufacturers can confidently plan for long-term supply contracts, knowing that the production capacity can be expanded to meet growing regulatory demands for impurity standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Cefpodoxime dippivoxil. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages of this new synthesis method for stakeholders involved in pharmaceutical quality control and supply chain management.
Q: What is the primary chemical challenge in synthesizing Cefpodoxime dippivoxil?
A: The main challenge lies in selectively alkylating the amino group on the thiazole ring without degrading the sensitive beta-lactam core or the ester side chains. Precise pH control (8.0-10.0) and mild temperatures (20-45°C) are critical to prevent hydrolysis while ensuring high conversion.
Q: Why is high-purity Cefpodoxime dippivoxil essential for pharmaceutical manufacturing?
A: As a potential process-related impurity, Cefpodoxime dippivoxil must be strictly monitored to meet pharmacopoeial standards (e.g., EP, ChP). Having a certified high-purity reference standard allows manufacturers to accurately quantify and control this impurity during the production of Cefpodoxime proxetil, ensuring patient safety and regulatory compliance.
Q: How does this novel synthesis method improve supply chain reliability?
A: The method utilizes readily available starting materials and avoids complex chromatographic purification. By employing a simple acid-base workup and crystallization strategy, the process is highly scalable and reduces dependency on specialized separation technologies, thereby stabilizing supply lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpodoxime Dippivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the highest quality reference standards and intermediates. Our team of expert chemists has thoroughly analyzed the synthetic pathway described in patent CN113999252A and is fully prepared to implement this advanced methodology for our global clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams for method validation or kilograms for stability batches, our supply is uninterrupted. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Cefpodoxime dippivoxil meets the exacting standards required by international pharmacopoeias.
We invite you to collaborate with us to optimize your impurity control strategy. By leveraging our technical expertise, you can secure a stable supply of this critical compound while potentially reducing your overall procurement costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring that your path to regulatory approval is smooth and efficient.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
