Advanced One-Step Synthesis of Halogenated Terephthalonitriles for Commercial Scale-up
Introduction to Next-Generation Intermediate Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for safer, more efficient, and economically viable synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN1422843A, which introduces a novel methodology for the preparation of halogenated terephthalonitriles. These compounds, including chlorinated, fluorinated, and brominated variants, serve as critical building blocks in the synthesis of high-performance polymers, agrochemicals, and pharmaceutical active ingredients. The patent outlines a transformative approach that bypasses the severe engineering constraints of traditional industrial methods, utilizing a cascade reaction sequence that converts halogenated terephthaloyl chlorides directly into the corresponding nitriles. This technical breakthrough addresses long-standing challenges regarding equipment corrosion, energy consumption, and process safety, positioning it as a highly attractive alternative for modern chemical production facilities seeking to optimize their operational footprint.
For R&D directors and process engineers, the significance of this patent lies in its ability to simplify complex multi-step syntheses into a manageable, potentially one-pot operation. By leveraging the reactivity of acid chlorides with ammonia followed by in-situ dehydration, the method achieves high yields under relatively mild conditions compared to the extreme environments required by legacy technologies. This report delves deep into the mechanistic underpinnings, comparative advantages, and commercial implications of adopting this synthesis route, providing a comprehensive analysis for stakeholders responsible for sourcing reliable fine chemical intermediates and optimizing supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of halogenated terephthalonitriles has relied heavily on the ammoxidation of p-xylene, a process that, while capable of large-scale continuous production, imposes severe demands on infrastructure and operational safety. As illustrated in the reaction pathway below, the conventional route begins with the conversion of p-xylene to terephthalonitrile via ammoxidation, typically requiring fluidized bed reactors operating at elevated temperatures and pressures. Subsequent halogenation steps often necessitate specialized tube reactors to handle the corrosive nature of halogens and the exothermicity of the reactions. Furthermore, the synthesis of fluorinated derivatives traditionally requires a multi-step sequence involving chlorination followed by halogen exchange, compounding the complexity and cost of the overall process.
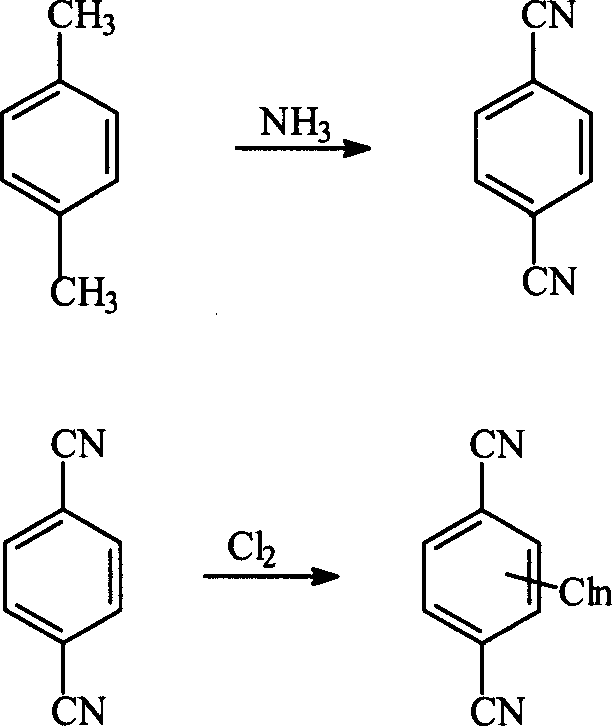
The reliance on fluidized beds and high-pressure tube reactors translates into substantial capital expenditure (CapEx) and rigorous maintenance schedules. The harsh reaction conditions not only accelerate equipment degradation but also pose significant safety risks related to thermal runaway and the handling of toxic gases at high pressures. Additionally, the multi-step nature of the traditional pathway, involving isolation and purification between the formation of the nitrile and subsequent halogenation, leads to cumulative yield losses and increased waste generation. For procurement managers, these factors manifest as higher unit costs and longer lead times, while supply chain heads face challenges in securing consistent quality due to the sensitivity of the process to minor operational fluctuations.
The Novel Approach
In stark contrast, the methodology described in CN1422843A offers a streamlined alternative that fundamentally alters the starting point of the synthesis. Instead of building the nitrile functionality from a methyl group via harsh oxidation, this novel approach starts with halogenated terephthaloyl chlorides, readily available precursors that already possess the necessary halogen substitution pattern. The process involves reacting these acid chlorides with liquid ammonia or ammonia gas to form the corresponding terephthalamides, followed immediately by dehydration to yield the target halogenated terephthalonitriles. Crucially, the patent highlights that these two distinct chemical transformations can be telescoped into a single synthetic operation, eliminating the need for intermediate isolation.
This strategic shift from oxidative construction to functional group interconversion allows the reaction to proceed under significantly milder conditions, typically ranging from -10°C to 100°C for the ammonolysis step and 80°C to reflux for the dehydration. The elimination of high-pressure fluidized beds and the ability to use standard glass-lined or stainless steel reactors for batch processing drastically reduces the barrier to entry for manufacturing. For organizations focused on cost reduction in fine chemical manufacturing, this represents a paradigm shift, replacing energy-intensive, high-risk continuous processes with flexible, safer batch operations that are easier to control and scale according to market demand.
Mechanistic Insights into Ammonolysis and Dehydration Cascade
The core of this innovative synthesis lies in the efficient execution of a nucleophilic acyl substitution followed by a dehydration event. In the first stage, the halogenated terephthaloyl chloride acts as a potent electrophile due to the electron-withdrawing nature of both the carbonyl group and the halogen substituents on the aromatic ring. When exposed to ammonia, a strong nucleophile, the chloride leaving groups are displaced to form the diamide intermediate. The patent specifies that this reaction can be conducted in various organic solvents, including ethers like ethylene glycol dimethyl ether or aromatic hydrocarbons like xylene and toluene. The molar ratio of ammonia to acid chloride is maintained in excess, typically between 1:2 and 1:20, ensuring complete conversion and driving the equilibrium towards the amide formation while neutralizing the generated hydrogen chloride as ammonium chloride.
Following the formation of the diamide, the system transitions seamlessly into the dehydration phase without the need for workup. The addition of a dehydrating agent such as phosphorus oxychloride (POCl3), phosphorus pentoxide (P2O5), or phosphorus trichloride (PCl3) activates the amide oxygen, facilitating the elimination of water to generate the nitrile triple bond. This step is thermally driven, with temperatures raised to between 80°C and the reflux temperature of the solvent, typically sustained for 4 to 10 hours. The choice of dehydrating agent is critical; phosphorus oxychloride, for instance, serves a dual role as both a dehydrating agent and a solvent medium in some variations, enhancing the solubility of the intermediate and promoting efficient mass transfer. This mechanistic elegance ensures that the final high-purity halogenated terephthalonitrile is obtained with minimal byproduct formation, as the reaction pathway avoids the radical mechanisms associated with direct halogenation of hydrocarbons which often lead to isomeric mixtures.
How to Synthesize Halogenated Terephthalonitrile Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity while maintaining safety standards. The process begins with the dissolution of the halogenated terephthaloyl chloride in a suitable anhydrous solvent, followed by the controlled introduction of ammonia gas at low temperatures to manage the exotherm of the amidation. Once the amide formation is complete, indicated by the cessation of ammonia absorption or monitored via TLC/HPLC, the dehydrating agent is introduced carefully to avoid violent reactions. The detailed standardized operating procedures, including specific stoichiometric ratios, agitation speeds, and quenching protocols necessary for GMP-compliant production, are outlined in the technical guide below.
- Dissolve halogenated terephthaloyl chloride in an organic solvent such as xylene or ethylene glycol dimethyl ether and introduce ammonia gas at controlled temperatures between -10°C and 100°C to form the amide intermediate.
- Without isolating the intermediate, add a dehydrating agent like phosphorus oxychloride or phosphorus pentoxide directly to the reaction mixture.
- Heat the mixture to reflux temperatures (80°C to 130°C) for 4 to 10 hours to complete the dehydration, then separate the solution and remove solvents under reduced pressure to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1422843A offers compelling strategic advantages that extend beyond simple chemistry. The shift away from high-pressure ammoxidation processes fundamentally changes the cost structure of producing these valuable intermediates. By utilizing standard batch reactors instead of specialized fluidized beds or tube reactors, manufacturers can significantly lower capital depreciation costs and reduce the frequency of expensive maintenance shutdowns. Furthermore, the ability to perform the synthesis in a telescoped manner reduces the number of unit operations, thereby lowering labor costs and minimizing the loss of material during transfer and isolation steps. This efficiency translates directly into a more competitive pricing structure for the end buyer, enabling cost reduction in pharmaceutical intermediates manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of extreme process conditions such as high-pressure ammoxidation removes the necessity for expensive, specialized reactor vessels and complex safety systems. By operating at atmospheric or near-atmospheric pressures and moderate temperatures, the energy consumption per kilogram of product is drastically reduced. Additionally, the use of common dehydrating agents and solvents avoids the need for exotic catalysts or reagents that drive up raw material costs. The telescoped nature of the reaction further enhances economic efficiency by reducing solvent usage and waste disposal fees associated with intermediate workups, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that starts from halogenated terephthaloyl chlorides diversifies the supply base, as these acid chlorides are commodity chemicals produced by multiple global suppliers. This contrasts with the traditional route which is often tied to specific large-scale petrochemical complexes capable of running continuous ammoxidation lines. The flexibility of the batch process allows for quicker response times to fluctuating market demands, reducing lead time for high-purity intermediates. Manufacturers can easily adjust batch sizes or switch between different halogenated variants (chloro, fluoro, bromo) using the same equipment setup, ensuring a steady and adaptable supply flow.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal catalysts or toxic gaseous reagents at high pressures make this process inherently safer and easier to scale from pilot plant to commercial production. The waste profile is significantly cleaner, primarily consisting of ammonium salts and phosphorus byproducts which are easier to treat than the complex mixed wastes from radical halogenation processes. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental compliance burden, ensuring long-term commercial scale-up of complex fine chemicals is sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of halogenated terephthalonitriles using this advanced methodology. These insights are derived directly from the experimental data and claims presented in the patent literature, aiming to clarify the feasibility and benefits for potential partners and technical evaluators.
Q: What are the primary advantages of the one-step method over traditional ammoxidation?
A: The one-step method starting from halogenated terephthaloyl chloride eliminates the need for high-pressure fluidized beds and tube reactors required in p-xylene ammoxidation, significantly reducing capital expenditure and operational complexity while allowing for milder reaction conditions.
Q: Which dehydrating agents are compatible with this synthesis route?
A: The patent specifies effective dehydration using phosphorus pentoxide (P2O5), phosphorus oxychloride (POCl3), or phosphorus trichloride (PCl3), which facilitate the conversion of the intermediate amide to the nitrile group efficiently.
Q: Is intermediate purification required between the ammonolysis and dehydration steps?
A: No, the process is designed as a telescoped one-pot reaction where the intermediate halogenated terephthalamide does not require purification or separation before the addition of the dehydrating agent, streamlining the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Terephthalonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your downstream applications, whether in crop protection, dye manufacturing, or drug discovery. Our technical team has extensively analyzed the pathway described in CN1422843A and possesses the expertise to implement this efficient, one-pot synthesis at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of halogenated terephthalonitrile meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this superior synthetic route for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your product competitiveness and secure your supply of these essential chemical building blocks.
