Advanced Manufacturing Of 3-Hydroxy-N-1-6-Trialkyl-4-Oxo-1-4-Dihydropyridine-2-Carboxamide Intermediates
Advanced Manufacturing Of 3-Hydroxy-N-1-6-Trialkyl-4-Oxo-1-4-Dihydropyridine-2-Carboxamide Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) intermediates, particularly for therapeutic classes addressing chronic conditions like iron overload disorders. Patent CN1578769A introduces a transformative methodology for the preparation of 3-hydroxy-N,1,6-trialkyl-4-oxo-1,4-dihydropyridine-2-carboxamides, which serve as potent oral iron chelators. This innovation addresses long-standing inefficiencies in prior art by replacing hazardous oxidation protocols with a catalytic TEMPO-based system and streamlining the amidation sequence. The core breakthrough lies in the direct conversion of protected hydroxymethyl pyranones to carboxylic acids without isolating aldehyde intermediates, significantly enhancing process safety and throughput.  . For procurement specialists and R&D leaders, this patent represents a pivotal shift towards greener chemistry that aligns with modern regulatory expectations while maintaining high purity standards essential for clinical applications.
. For procurement specialists and R&D leaders, this patent represents a pivotal shift towards greener chemistry that aligns with modern regulatory expectations while maintaining high purity standards essential for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these vital iron chelator intermediates relied on multi-step sequences fraught with operational hazards and inefficiencies. As illustrated in the prior art reaction schemes, traditional methods often employed Jones reagent (chromium trioxide in sulfuric acid) or dimethyl sulfoxide activated with sulfur trioxide-pyridine complexes to oxidize primary alcohols. 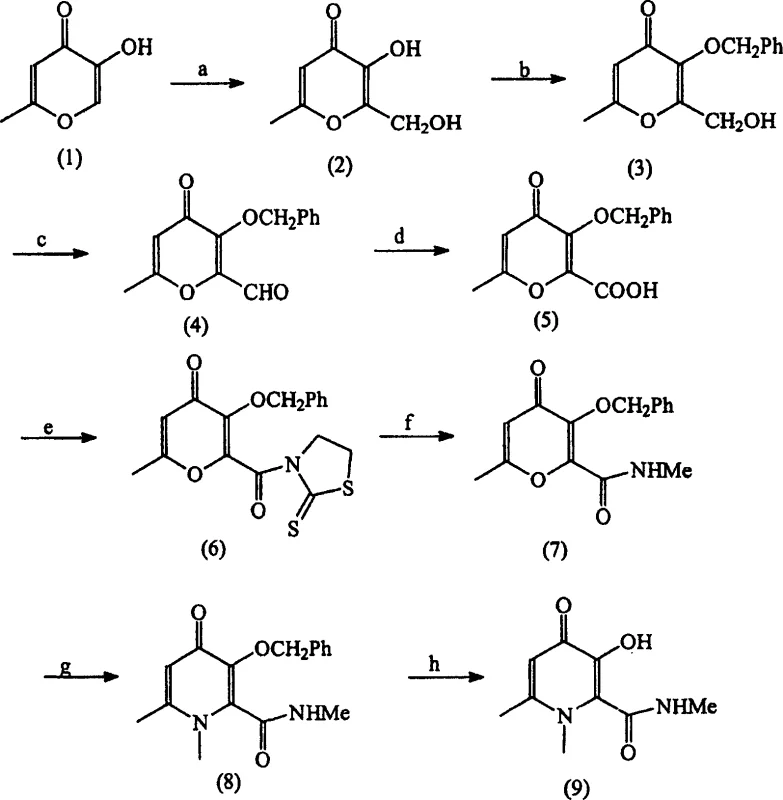 . These conventional approaches suffer from severe drawbacks, including the generation of large volumes of toxic heavy metal waste that require costly disposal procedures. Furthermore, the isolation of reactive aldehyde intermediates necessitates rigorous purification, often involving column chromatography, which is impractical for industrial scale-up. The use of coupling agents like dicyclohexylcarbodiimide (DCC) in subsequent amidation steps introduces additional complications, such as the formation of difficult-to-remove urea byproducts and potential skin sensitization risks for plant operators.
. These conventional approaches suffer from severe drawbacks, including the generation of large volumes of toxic heavy metal waste that require costly disposal procedures. Furthermore, the isolation of reactive aldehyde intermediates necessitates rigorous purification, often involving column chromatography, which is impractical for industrial scale-up. The use of coupling agents like dicyclohexylcarbodiimide (DCC) in subsequent amidation steps introduces additional complications, such as the formation of difficult-to-remove urea byproducts and potential skin sensitization risks for plant operators.
The Novel Approach
In stark contrast, the novel process described in CN1578769A utilizes a highly efficient one-step oxidation strategy employing TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) as a catalyst alongside sodium hypochlorite and bromide salts. This method allows for the direct transformation of the hydroxymethyl precursor into the corresponding carboxylic acid under mild biphasic conditions, typically maintaining temperatures below 10°C to ensure selectivity. By bypassing the isolation of the aldehyde intermediate, the process drastically reduces unit operations and solvent consumption. The subsequent amidation step utilizes 1,1'-carbonyldiimidazole (CDI) instead of DCC, facilitating a cleaner reaction profile that avoids the formation of insoluble urea precipitates. This streamlined approach not only improves overall yield but also simplifies the work-up procedure, enabling the production of high-purity intermediates suitable for downstream pharmaceutical manufacturing without extensive chromatographic purification.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and CDI Amidation
The mechanistic elegance of this process centers on the catalytic cycle of the nitroxyl radical TEMPO, which mediates the selective oxidation of primary alcohols to carboxylic acids via an aldehyde hydrate intermediate. In the presence of sodium hypochlorite and catalytic amounts of bromide ions, TEMPO is oxidized to an oxoammonium species, which acts as the active oxidant for the alcohol substrate. The reaction kinetics are carefully controlled by pH buffering with sodium bicarbonate, ensuring that the oxidation proceeds rapidly to the acid stage without over-oxidation or degradation of the sensitive pyranone ring system. This precise control is critical for minimizing impurities such as chlorinated byproducts or ring-opened species that could compromise the safety profile of the final API. The biphasic nature of the reaction mixture, often utilizing dichloromethane and water, facilitates efficient heat transfer and mass transport, which are essential parameters for maintaining consistency during large-batch production runs.
Following the oxidation, the conversion of the carboxylic acid to the amide is achieved through activation with CDI, forming a reactive acyl imidazole intermediate in situ. This activation method is superior to acid chloride formation or carbodiimide coupling because it generates imidazole as the only byproduct, which is generally soluble and easily removed during aqueous work-up. The nucleophilic attack by the alkylamine on the activated carbonyl center proceeds smoothly at elevated temperatures, typically between 60°C and 80°C, driving the reaction to completion. This mechanism ensures high atom economy and reduces the burden on downstream purification systems. For quality control teams, understanding this mechanism is vital for setting appropriate in-process controls, particularly regarding the stoichiometry of the activating agent and the removal of residual imidazole to meet stringent pharmacopeial limits.
How to Synthesize 3-Hydroxy-N-1-6-Trialkyl-4-Oxo-1-4-Dihydropyridine-2-Carboxamide Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to maximize the benefits of the patented technology. The process begins with the preparation of the protected hydroxymethyl pyranone, which is then subjected to the TEMPO oxidation protocol under strictly controlled thermal conditions. Operators must monitor the pH and temperature profiles closely to prevent exotherms that could lead to side reactions. Following the oxidation, the crude acid can often be used directly or with minimal purification for the subsequent amidation step, further compressing the manufacturing timeline.
- Oxidize 3-O-protected-2-hydroxymethyl-6-alkyl-4H-pyran-4-one using TEMPO, sodium hypochlorite, and bicarbonate buffer at low temperature.
- React the resulting carboxylic acid intermediate with 1,1'-carbonyldiimidazole (CDI) in an inert solvent.
- Add alkylamine to form the amide bond, followed by deprotection of the alcohol group to yield the final hydroxy-pyridone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers substantial strategic advantages for organizations managing the supply of complex pharmaceutical intermediates. The elimination of chromium-based oxidants and DCC coupling agents translates directly into reduced raw material costs and significantly lower waste management expenses. By simplifying the process flow and removing the need for intermediate isolation and chromatographic purification, manufacturers can achieve faster cycle times and higher throughput capacities. This efficiency gain is particularly valuable in a supply chain environment where agility and responsiveness to market demand are critical competitive differentiators. Furthermore, the use of readily available, commodity-grade reagents enhances supply security by reducing dependency on specialized or hazardous chemical vendors.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents like Jones reagent and DCC with cost-effective alternatives such as sodium hypochlorite and CDI results in a drastic reduction in direct material costs. Additionally, the simplified work-up procedures reduce labor hours and solvent usage, leading to substantial overall cost savings per kilogram of produced intermediate. The avoidance of chromatographic purification steps further lowers operational expenditures by minimizing silica gel consumption and solvent recovery loads.
- Enhanced Supply Chain Reliability: The robustness of the TEMPO oxidation method ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. The use of stable, non-hazardous reagents simplifies logistics and storage requirements, mitigating risks associated with the transportation of dangerous goods. This reliability allows supply chain managers to maintain leaner inventory levels while ensuring continuous availability of critical intermediates for API synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large reactors without requiring specialized equipment for handling toxic gases or cryogenic temperatures. The significant reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the carbon footprint of the manufacturing process. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the TEMPO oxidation method over Jones reagent?
A: The TEMPO-mediated oxidation avoids the use of toxic chromium(VI) reagents, eliminates heavy metal waste disposal issues, and operates under milder conditions with higher selectivity and yield compared to traditional Jones oxidation.
Q: How does this process improve supply chain reliability for iron chelator intermediates?
A: By reducing the number of synthetic steps and avoiding the isolation of unstable aldehyde intermediates, the process minimizes production lead times and reduces the risk of batch failures, ensuring more consistent supply continuity.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights the scalability of the method, noting that reagents are inexpensive and commercially available in metric ton quantities, and the work-up procedures avoid complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-N-1-6-Trialkyl-4-Oxo-1-4-Dihydropyridine-2-Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1578769A are fully realized in practical manufacturing settings. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the highest quality standards required for drug substance manufacturing. Our commitment to technical excellence ensures that complex chemical transformations are executed with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timelines, ensuring a seamless transition from laboratory scale to commercial supply.
