Advanced Clarithromycin Manufacturing: Technical Upgrades and Commercial Scalability Analysis
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for high-volume antibiotics like clarithromycin. Patent CN102268053A introduces a transformative preparation method that addresses critical bottlenecks in traditional macrolide synthesis. This technical disclosure outlines a novel two-step sequence starting from (2', 4''-O-bistrimethylsilyl)-erythromycin A-9[O-(1-ethoxy-1-methylethyl)] oxime. By shifting the methylation reagent from expensive and toxic methyl iodide to methyl bromide, and optimizing the deprotection conditions with acetic acid, the inventors have achieved a process that significantly lowers production costs while enhancing environmental compliance. For R&D directors and procurement strategists, this patent represents a viable pathway to optimize the supply chain for this essential respiratory infection treatment, offering a compelling alternative to legacy methods that suffer from harsh conditions and complex waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of clarithromycin has been plagued by several inherent chemical and economic challenges that impact overall manufacturing viability. Traditional protocols often rely on methyl iodide or dimethyl sulfate as methylating agents, both of which present severe drawbacks; methyl iodide is notoriously expensive and possesses high toxicity, posing significant occupational health risks and requiring stringent containment measures, while dimethyl sulfate is equally hazardous and difficult to handle safely on a large scale. Furthermore, conventional deprotection steps frequently utilize strong acids like formic acid under reflux conditions, which can lead to the degradation of the sensitive macrolide ring structure, generating unwanted dehydration by-products such as 8,9-dehydration-6,9-hemiketal derivatives. These side reactions not only reduce the final yield but also complicate the purification process, necessitating extensive chromatography or multiple recrystallization steps to meet pharmacopeial standards, thereby driving up both operational time and material costs significantly.
The Novel Approach
In stark contrast to these legacy issues, the methodology described in CN102268053A employs a strategic substitution of reagents and conditions to streamline the entire synthetic workflow. The core innovation lies in the utilization of methyl bromide for the critical C6-hydroxyl methylation step, conducted in a mixed solvent system of toluene and dimethyl sulfoxide (DMSO) at mild temperatures ranging from 0 to 15°C. This approach not only drastically reduces raw material costs but also improves regioselectivity, ensuring that methylation occurs predominantly at the desired position without affecting other sensitive functional groups on the erythromycin scaffold. Additionally, the subsequent deprotection and deoximation are combined into a single pot operation using acetic acid and sodium bisulfite, which eliminates the need for isolating intermediate compounds and avoids the harsh acidic environments that typically degrade product quality, resulting in a cleaner reaction profile and simplified downstream processing.
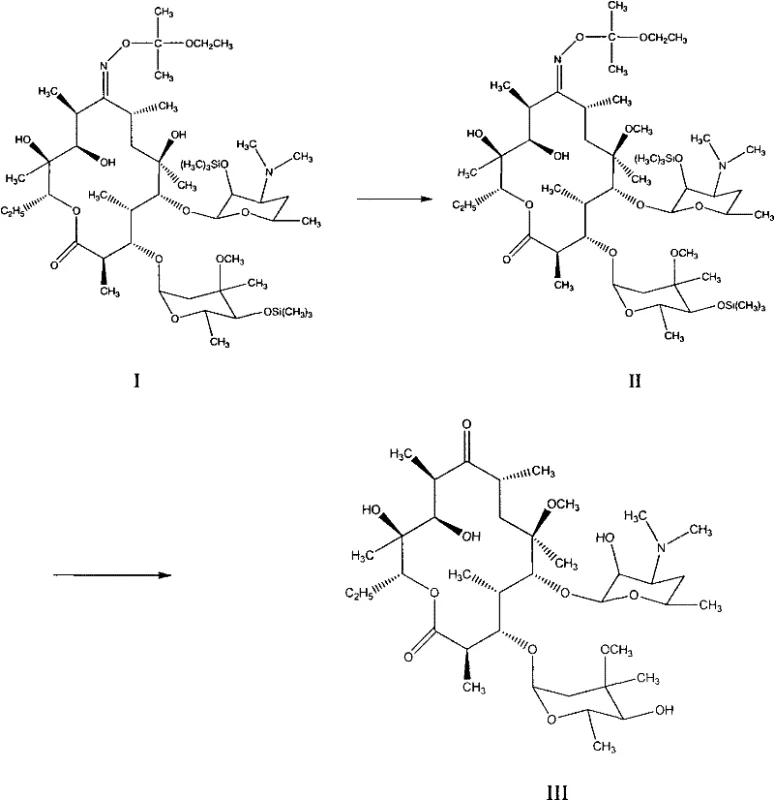
Mechanistic Insights into Selective Methylation and One-Pot Deprotection
The success of this synthetic route hinges on the precise orchestration of protecting group chemistry and nucleophilic substitution mechanisms. The starting material features trimethylsilyl groups at the 2' and 4'' positions, which effectively mask these hydroxyl groups to prevent them from participating in the methylation reaction, thereby directing the methyl bromide exclusively to the C6-hydroxyl group in the presence of a potassium hydroxide base. The use of a polar aprotic co-solvent like DMSO enhances the nucleophilicity of the C6-alkoxide ion, facilitating a rapid SN2 reaction with methyl bromide even at low temperatures, which kinetically suppresses potential elimination reactions or attack at other sites. This high degree of chemoselectivity is crucial for maintaining the integrity of the macrolide backbone, ensuring that the resulting methylated intermediate retains the stereochemical configuration required for biological activity without generating difficult-to-remove regioisomers.
Following methylation, the process leverages a synergistic deprotection mechanism where acetic acid serves a dual purpose as both a proton source for removing the silyl ethers and a catalyst for the hydrolysis of the C9-oxime group. The addition of sodium bisulfite acts as a reducing agent or scavenger that facilitates the cleavage of the oxime functionality, regenerating the C9-ketone essential for clarithromycin's antibiotic properties. By conducting this in an alcohol-water mixture at moderate temperatures (50-80°C), the system achieves a balance where the silyl groups are cleaved efficiently without inducing the acid-catalyzed dehydration of the C8 and C9 positions that is common with stronger mineral acids. This mechanistic finesse results in a crude product with a significantly improved impurity profile, reducing the burden on final purification steps and allowing for higher recovery rates of the active pharmaceutical ingredient.
How to Synthesize Clarithromycin Efficiently
Implementing this optimized synthesis requires careful attention to solvent ratios and temperature control to maximize the benefits of the methyl bromide reagent. The process begins with the dissolution of the protected oxime precursor in a specific ratio of toluene and DMSO, followed by the controlled addition of the methylating agent under basic conditions. Once the methylation is complete, the reaction mixture undergoes a straightforward aqueous workup to remove inorganic salts, after which the organic layer is concentrated to yield the methylated intermediate. This intermediate is then subjected to the one-pot deprotection sequence without further purification, utilizing acetic acid and sodium bisulfite in an alcoholic solvent to simultaneously remove the silyl protecting groups and the oxime moiety, ultimately yielding high-purity clarithromycin after crystallization. For detailed operational parameters and stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve (2', 4''-O-bistrimethylsilyl)-erythromycin A-9[O-(1-ethoxy-1-methylethyl)] oxime in a mixed solvent of toluene and DMSO, then cool to 0-15°C.
- Add methyl bromide and potassium hydroxide catalyst, stirring vigorously for 15-120 minutes to achieve selective C6-O-methylation.
- Perform a one-pot deprotection and deoximation reaction using acetic acid and sodium bisulfite in an alcohol-water solvent system at 50-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial strategic benefits for procurement managers and supply chain directors aiming to optimize the cost structure of antibiotic manufacturing. The primary driver of value creation is the substitution of methyl iodide with methyl bromide, a reagent that is not only significantly less expensive but also easier to source in bulk quantities, thereby stabilizing raw material costs and reducing exposure to volatile pricing markets associated with iodine-based chemicals. Moreover, the simplified workup procedure, which relies on basic extraction and crystallization rather than complex chromatographic separations, translates directly into reduced solvent consumption and lower energy requirements for concentration and drying, contributing to a leaner and more cost-effective production cycle that enhances overall margin potential.
- Cost Reduction in Manufacturing: The economic impact of switching to methyl bromide cannot be overstated, as it eliminates the premium costs associated with handling and disposing of highly toxic methyl iodide. By operating at lower temperatures and utilizing acetic acid instead of more aggressive acids, the process minimizes equipment corrosion and extends the lifespan of reactor vessels, leading to lower capital expenditure on maintenance and replacement. Furthermore, the reduction in by-product formation means that less material is lost to waste streams, effectively increasing the mass balance efficiency and lowering the cost per kilogram of the final active pharmaceutical ingredient produced.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like methyl bromide and acetic acid mitigates the risk of supply disruptions that can occur with specialized or heavily regulated chemicals. The robustness of the reaction conditions, which tolerate a broader range of operational parameters without compromising yield, ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance and meeting delivery commitments to downstream pharmaceutical customers. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for excessive safety stock and improving working capital efficiency across the organization.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, featuring simple unit operations that are easily transferred from pilot plants to multi-ton commercial reactors without significant engineering hurdles. The significant reduction in "three wastes"—specifically the minimization of hazardous organic waste and acidic effluent—aligns with increasingly stringent global environmental regulations, reducing the financial burden of waste treatment and disposal. This eco-friendly profile not only lowers operational costs but also enhances the corporate sustainability credentials of the manufacturer, a growing priority for multinational pharmaceutical partners evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clarithromycin synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Stakeholders are encouraged to review these points to assess the feasibility of integrating this technology into their existing manufacturing portfolios for improved efficiency and cost management.
Q: Why is methyl bromide preferred over methyl iodide in this clarithromycin synthesis?
A: Methyl bromide offers significant cost advantages and reduced toxicity compared to methyl iodide. Furthermore, the patent indicates that methyl bromide provides better methylation selectivity at lower temperatures (0-15°C), minimizing by-product formation and simplifying downstream purification.
Q: How does the new deprotection method improve product quality?
A: The novel process utilizes acetic acid instead of stronger acids like formic acid. This milder acidic condition prevents the decomposition of the sensitive macrolide structure during the high-temperature deprotection phase, resulting in fewer impurities and higher overall yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial suitability. It features simple post-treatment steps such as extraction and crystallization, reduces the generation of 'three wastes' (waste water, gas, and residue), and utilizes readily available reagents, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clarithromycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN102268053A can be seamlessly translated into reliable supply solutions. We are committed to delivering high-purity intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities which guarantee that every batch conforms to the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and quality targets. By collaborating with us, you can leverage our expertise to conduct a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this methyl bromide-based process. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value and stability in your supply chain.
