Advanced NHC-Catalyzed Synthesis of Beta-Sulfonyl Chiral Amino Compounds for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral building blocks, particularly those containing sulfur functionalities which are prevalent in bioactive molecules. Patent CN112479948A introduces a significant advancement in this domain by disclosing a novel preparation method for beta-sulfonyl chiral amino compounds. This technology leverages an asymmetric Michael addition reaction catalyzed by nitrogen heterocyclic carbenes (NHC), offering a distinct alternative to traditional metal-catalyzed or organocatalytic routes. The core innovation lies in the ability to introduce diverse functional group substituents at the R1, R2, FG1, and FG2 positions, thereby generating a library of structurally varied chiral amines. 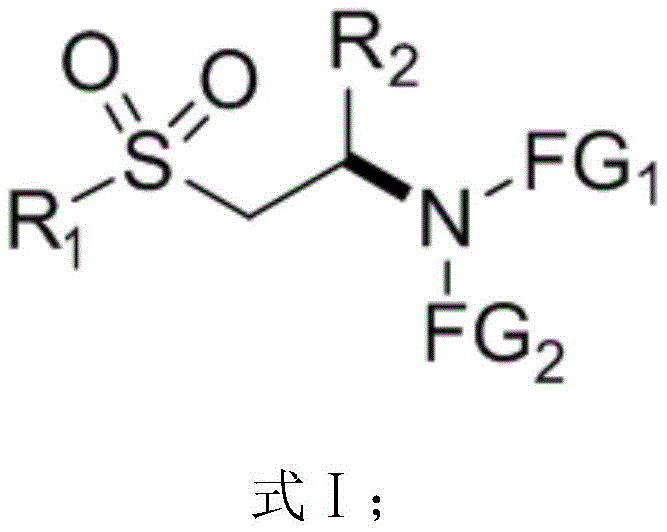 As illustrated in the general formula, the versatility of this scaffold allows for the subsequent removal of nitrogen functional groups to yield naked amino derivatives, providing a critical research basis for screening new drug candidates and functional materials with high precision.
As illustrated in the general formula, the versatility of this scaffold allows for the subsequent removal of nitrogen functional groups to yield naked amino derivatives, providing a critical research basis for screening new drug candidates and functional materials with high precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amino compounds has relied heavily on asymmetric addition reactions catalyzed by Cinchona alkaloids or their derivatives. While these natural product-derived catalysts have served the industry well, they possess inherent limitations that restrict their utility in modern complex molecule synthesis. The primary drawback is the relatively weak Bronsted basicity of Cinchona alkaloids, which limits the range of substrates that can be effectively activated. Consequently, the substrate scope is often confined to specific conjugated ketene compounds, such as chalcone derivatives, severely restricting the structural diversity of the resulting beta-sulfonyl chiral amino compounds. Furthermore, traditional methods may struggle with achieving high enantioselectivity across a broad range of electronic and steric environments, often requiring extensive optimization for each new substrate, which increases both time and cost in the R&D phase.
The Novel Approach
The methodology presented in patent CN112479948A overcomes these historical bottlenecks by employing an activated N-heterocyclic carbene (NHC) catalytic system. This novel approach facilitates the formation of a rigid transition state through specific non-covalent interactions between the catalyst, the Michael acceptor, and the nucleophilic reagent. 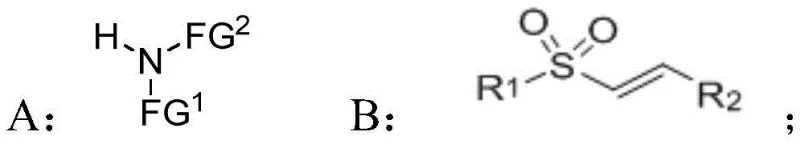 By enhancing the nucleophilicity of the reagent and selectively attacking the double bond of the Michael acceptor from one specific side, the reaction achieves superior asymmetric induction. This strategy not only expands the designability of the target molecules by accommodating a wide variety of R and FG groups but also simplifies the chemical conversion means required to access these valuable intermediates. The result is a streamlined pathway to beta-sulfonyl chiral amino compounds with potential application value that was previously difficult or impossible to access efficiently.
By enhancing the nucleophilicity of the reagent and selectively attacking the double bond of the Michael acceptor from one specific side, the reaction achieves superior asymmetric induction. This strategy not only expands the designability of the target molecules by accommodating a wide variety of R and FG groups but also simplifies the chemical conversion means required to access these valuable intermediates. The result is a streamlined pathway to beta-sulfonyl chiral amino compounds with potential application value that was previously difficult or impossible to access efficiently.
Mechanistic Insights into NHC-Catalyzed Asymmetric Michael Addition
The mechanistic elegance of this transformation relies on the unique electronic properties of the nitrogen heterocyclic carbene catalyst. In the reaction system, the NHC catalyst works synergistically with a base reagent to activate the substrates. Specifically, the carbene catalyst interacts with the conjugated olefin sulfone compound (Michael acceptor), effectively lowering its Lowest Unoccupied Molecular Orbital (LUMO) energy level. Simultaneously, through non-covalent interactions, the catalyst raises the Highest Occupied Molecular Orbital (HOMO) of the nucleophilic reagent. 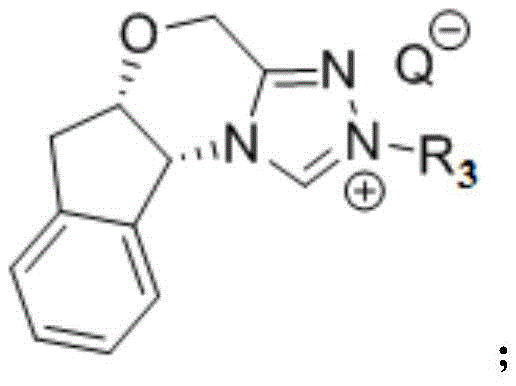 This dual activation mechanism significantly reduces the activation energy of the reaction, thereby increasing reaction activity and allowing the process to proceed smoothly under mild conditions. The formation of a rigid, chiral transition state ensures that the nucleophilic attack occurs with high facial selectivity, which is the fundamental driver for the observed high enantioselectivity in the final product.
This dual activation mechanism significantly reduces the activation energy of the reaction, thereby increasing reaction activity and allowing the process to proceed smoothly under mild conditions. The formation of a rigid, chiral transition state ensures that the nucleophilic attack occurs with high facial selectivity, which is the fundamental driver for the observed high enantioselectivity in the final product.
From an impurity control perspective, this organocatalytic route offers distinct advantages over transition metal catalysis. Since the system is metal-free, there is no risk of heavy metal contamination, which is a critical quality attribute for pharmaceutical intermediates intended for human use. The byproducts generated are minimal, and the reaction residues exhibit low toxicity, simplifying the purification workflow. The use of inorganic bases like potassium phosphate further supports a green chemistry profile by avoiding harsh acidic or basic workups that could lead to racemization or decomposition of the sensitive chiral centers. This mechanistic robustness ensures that the optical purity of the beta-sulfonyl chiral amino compound is maintained throughout the synthesis, delivering high-purity materials suitable for stringent regulatory requirements.
How to Synthesize Beta-Sulfonyl Chiral Amino Compound Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these high-value intermediates. The process begins with the preparation of the catalytic system, where an indanol-derived triazole carbene catalyst is mixed with a base in an anhydrous solvent under an inert atmosphere. Following the activation of the catalyst, the nucleophilic reagent is introduced, followed by the slow addition of the beta-unsaturated sulfone compound. The reaction is typically stirred at temperatures ranging from -90°C to 35°C for a period of 6 to 48 hours, depending on the specific substrate reactivity.
- Prepare the reaction system by mixing an N-heterocyclic carbene catalyst and a base reagent in an anhydrous solvent under inert atmosphere.
- Add the nucleophilic reagent compound A to the catalytic mixture and stir to allow for deprotonation and activation.
- Introduce the conjugated olefin sulfone compound B to the system to initiate the asymmetric Michael addition reaction, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHC-catalyzed technology represents a strategic opportunity to optimize manufacturing costs and secure supply continuity. The shift from metal-based catalysis to organocatalysis eliminates the need for expensive transition metals and the associated rigorous purification steps required to remove metal traces to ppm levels. This reduction in downstream processing complexity translates directly into significant cost savings and shorter production cycles. Furthermore, the mild reaction conditions reduce energy consumption and enhance operational safety, contributing to a more sustainable and economically viable manufacturing process for complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver associated with both the raw material price of the metal and the specialized scavenging resins or filtration systems needed for cleanup. Additionally, the high atom utilization rate and the ability to use readily available starting materials without extensive pre-modification streamline the supply chain. By reducing the number of unit operations and simplifying the workup procedure, manufacturers can achieve substantial cost reductions in pharma intermediate manufacturing while maintaining high product quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available organic catalysts and common inorganic bases mitigates the supply risks often associated with scarce precious metals. The robustness of the reaction across a wide temperature range allows for greater flexibility in production scheduling and facility utilization. This reliability ensures consistent delivery of high-purity beta-sulfonyl chiral amino compounds, reducing lead times and preventing bottlenecks in the downstream synthesis of active pharmaceutical ingredients.
- Scalability and Environmental Compliance: The low toxicity of the catalytic system and the minimal generation of hazardous waste align perfectly with modern environmental regulations. The process is inherently safer and easier to scale from laboratory to commercial production without the need for specialized containment equipment required for toxic metals. This ease of scale-up facilitates the rapid transition from pilot batches to multi-ton production, ensuring that supply can meet market demand without compromising on environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders evaluating this methodology for their own production needs.
Q: What are the advantages of using NHC catalysis over traditional Cinchona alkaloids for this synthesis?
A: Unlike traditional Cinchona alkaloid catalysts which are limited to specific conjugated ketene compounds like chalcones, the N-heterocyclic carbene (NHC) catalytic system described in patent CN112479948A offers a much broader substrate scope. It enables the efficient synthesis of diverse beta-sulfonyl chiral amino compounds with high enantioselectivity through a rigid transition state formed by non-covalent interactions, overcoming the limitations of weak Bronsted basicity found in older methods.
Q: Is this synthesis method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the method is highly suitable for scale-up due to its metal-free nature and mild reaction conditions ranging from -90°C to 35°C. The use of readily available starting materials and the elimination of toxic heavy metal residues significantly simplify the downstream purification process, reducing environmental impact and production costs, which are critical factors for commercial-scale API intermediate manufacturing.
Q: What level of enantioselectivity can be achieved with this protocol?
A: The patent data demonstrates that this asymmetric Michael addition protocol can achieve high enantioselectivity, with specific examples reporting ee values up to 94%. This high level of stereocontrol is achieved through the synergistic effect of the chiral N-heterocyclic carbene catalyst and the base reagent, ensuring the production of optically pure intermediates essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Sulfonyl Chiral Amino Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be successfully translated into industrial reality. We are equipped with rigorous QC labs and adhere to stringent purity specifications to deliver beta-sulfonyl chiral amino compounds that meet the highest global standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge NHC-catalyzed technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in asymmetric organocatalysis can drive efficiency and innovation in your supply chain.
