Advanced Tungstate-Catalyzed Synthesis of 5-Substituted Tetrazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously demand robust, safe, and scalable synthetic routes for high-value heterocyclic intermediates, particularly 5-substituted tetrazoles which serve as critical bioisosteres for carboxylic acids in drug design. Patent CN100591674C introduces a transformative methodology utilizing tungstate catalysts to facilitate the [2+3] cycloaddition between nitriles and sodium azide, effectively addressing long-standing safety and efficiency bottlenecks in tetrazole manufacturing. This innovation represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering complex heterocycles with superior process safety profiles. By replacing hazardous hydrazoic acid generation with a solid-state azide protocol mediated by earth-abundant tungsten salts, this technology not only enhances operator safety but also streamlines the downstream purification process, thereby offering substantial potential for cost reduction in API manufacturing. The versatility of this catalytic system allows for the processing of a wide array of aromatic and heteroaromatic nitriles, ensuring that supply chains for key drug substances remain resilient and uninterrupted.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-substituted tetrazoles has been plagued by severe safety hazards and environmental concerns associated with traditional catalytic systems. Conventional protocols frequently rely on the in situ generation of hydrazoic acid (HN3) from sodium azide and strong mineral acids, a practice that poses an unacceptable risk of explosion and toxicity in large-scale reactors. Furthermore, alternative methods employing organotin reagents or soluble zinc salts often result in difficult-to-remove heavy metal impurities, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. These legacy processes also suffer from poor atom economy and generate significant volumes of hazardous waste, complicating disposal and increasing the overall environmental footprint of the manufacturing campaign. The inability to easily recover homogeneous catalysts in these traditional routes further exacerbates production costs, making the economic viability of such processes increasingly questionable in a competitive global market.
The Novel Approach
In stark contrast, the tungstate-catalyzed method disclosed in the patent data offers a paradigm shift towards greener and more sustainable chemical manufacturing. By utilizing insoluble tungstate salts such as barium tungstate (BaWO4) or calcium tungstate (CaWO4), the reaction proceeds through a heterogeneous catalytic mechanism that eliminates the release of free hydrazoic acid gas. This fundamental change in reaction engineering allows for the safe handling of sodium azide in polar aprotic solvents like DMF or DMSO at elevated temperatures without the risk of catastrophic pressure buildup. The solid nature of the catalyst facilitates its physical separation from the reaction mixture via simple filtration or centrifugation post-reaction, enabling potential catalyst recycling and drastically reducing metal contamination in the final product. This approach not only simplifies the workflow but also aligns perfectly with modern green chemistry principles, providing a clear pathway for the commercial scale-up of complex heterocycles with minimal environmental impact.

Mechanistic Insights into Tungstate-Catalyzed Cycloaddition
The efficacy of this synthetic route lies in the unique Lewis acidic properties of the tungstate anion, which activates the nitrile triple bond towards nucleophilic attack by the azide ion. In the proposed mechanistic cycle, the tungsten center coordinates with the nitrogen atom of the nitrile group, increasing the electrophilicity of the carbon atom and lowering the activation energy required for the initial addition of the azide species. This coordination stabilizes the transition state leading to the formation of the tetrazole ring, allowing the reaction to proceed efficiently at 120 degrees Celsius without the need for extreme pressures or hazardous promoters. The use of polar solvents like DMF further enhances the solubility of the ionic sodium azide while maintaining the suspension of the solid tungstate catalyst, creating an optimal interfacial environment for the reaction to occur. Detailed kinetic studies suggest that the reaction rate is dependent on the surface area of the tungstate particles and the concentration of dissolved azide, indicating a mechanism that bridges homogeneous and heterogeneous catalysis regimes.
From an impurity control perspective, this mechanism offers distinct advantages over zinc-mediated processes where metal chelation can lead to persistent impurities. Since the tungstate catalyst remains largely insoluble throughout the reaction course, there is minimal leaching of tungsten into the organic phase, resulting in a crude product profile that is significantly cleaner than those obtained from homogeneous catalysis. The subsequent workup involving neutralization with hydrochloric acid and extraction with ethyl acetate effectively removes any residual inorganic salts and unreacted starting materials, yielding a high-purity 5-substituted tetrazole suitable for direct use in subsequent coupling reactions. This inherent purity reduces the burden on downstream crystallization or chromatography steps, thereby shortening the overall production timeline and reducing lead time for high-purity intermediates required for clinical trial material synthesis.
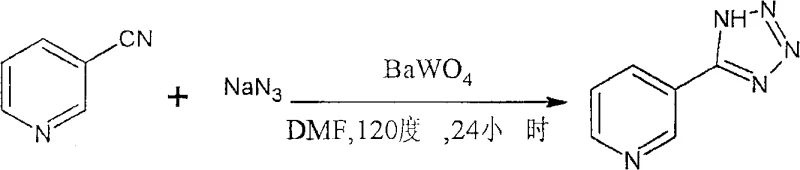
How to Synthesize 5-Substituted Tetrazole Efficiently
The operational simplicity of this patented method makes it highly attractive for both laboratory-scale optimization and multi-ton commercial production. The process begins with the precise weighing of the nitrile substrate and sodium azide, which are dissolved in a specified volume of DMF or DMSO to ensure homogeneity of the reactants prior to catalyst addition. Following the addition of the tungstate catalyst, the mixture is heated under reflux conditions, where the temperature is strictly maintained to drive the equilibrium towards product formation while preventing thermal decomposition of the sensitive tetrazole ring. Upon completion, the cooling and filtration steps are critical for maximizing catalyst recovery, and the subsequent acid-base workup ensures the isolation of the free tetrazole acid form. For a detailed breakdown of the specific molar ratios, solvent volumes, and temperature profiles required for different substrates, please refer to the standardized synthesis guide below.
- Dissolve the nitrile substrate and sodium azide in an organic solvent such as DMF or DMSO with a molar ratio of 1: 1.05-2.1, then add the tungstate catalyst (e.g., BaWO4) at 10-30 mol% relative to the nitrile.
- Heat the reaction mixture to 120 degrees Celsius and maintain stirring for 12 to 24 hours to ensure complete cycloaddition and formation of the tetrazole ring.
- Cool the reaction mixture, filter or centrifuge to recover the solid tungstate catalyst, neutralize the filtrate with 6mol/L hydrochloric acid, and extract the product with ethyl acetate followed by rotary evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tungstate-catalyzed technology translates directly into tangible strategic benefits regarding cost stability and operational continuity. The elimination of hazardous hydrazoic acid generation removes the need for specialized scrubbing equipment and extensive safety monitoring systems, leading to a significant reduction in capital expenditure and operational overheads associated with safety compliance. Furthermore, the ability to recover and reuse the tungstate catalyst multiple times without significant loss of activity creates a circular economy within the reactor, drastically lowering the raw material cost per kilogram of the final product. This efficiency gain is compounded by the use of commodity chemicals like sodium azide and benzonitriles, which are readily available from multiple global suppliers, ensuring that the supply chain remains robust against regional disruptions or price volatility.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the heterogeneous nature of the catalyst, which eliminates the expensive and complex steps required to scavenge heavy metals from the final product. Unlike traditional methods that require costly resin treatments or multiple recrystallizations to meet ppm-level metal specifications, this method yields a product with inherently low metal content, thereby reducing the consumption of auxiliary materials and solvents. Additionally, the high conversion rates and selectivity observed across various substrates minimize the formation of by-products, leading to higher overall yields and less waste disposal costs. These factors collectively contribute to a leaner manufacturing process that maximizes resource utilization and improves the gross margin for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on stable, solid-state reagents rather than unstable gaseous intermediates significantly de-risks the manufacturing schedule. Traditional routes prone to safety incidents or regulatory shutdowns due to hydrazoic acid emissions are replaced by a benign process that can run continuously with minimal interruption. The robustness of the tungstate catalyst against moisture and air exposure further simplifies logistics, as the catalyst does not require inert atmosphere storage or special transportation protocols. This reliability ensures that production timelines are met consistently, providing downstream partners with a predictable flow of critical intermediates necessary for maintaining their own inventory levels and meeting market demand.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of exothermic runaway risks associated with hydrazoic acid evolution. The reaction can be safely conducted in standard glass-lined or stainless steel reactors equipped with standard heating and agitation systems, removing the need for custom-engineered high-pressure vessels. From an environmental standpoint, the process generates significantly less hazardous waste, as the primary by-product is inorganic salt which can be treated via standard wastewater protocols. This alignment with strict environmental regulations facilitates faster permitting and approval processes for new manufacturing sites, accelerating the time-to-market for new drug candidates relying on this tetrazole scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tungstate-catalyzed synthesis route. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams negotiating supply agreements based on this specific intellectual property.
Q: What are the safety advantages of this tungstate-catalyzed method compared to traditional tetrazole synthesis?
A: Traditional methods often involve the generation of highly toxic and explosive hydrazoic acid (HN3) in situ or the use of hazardous organotin reagents. This patented process utilizes solid sodium azide and a stable tungstate catalyst in a closed system, significantly mitigating the risk of explosive gas release and improving overall operational safety for industrial scale-up.
Q: Can the tungstate catalyst be recovered and reused in this process?
A: Yes, a key feature of this invention is the heterogeneous nature of the tungstate catalyst (such as BaWO4). After the reaction is complete, the solid catalyst can be easily separated from the liquid reaction mixture via simple filtration or centrifugation, allowing for potential recycling and reducing the overall cost of goods sold (COGS) by minimizing catalyst consumption.
Q: What types of nitrile substrates are compatible with this synthetic route?
A: The method demonstrates broad substrate scope, successfully converting aromatic nitriles with various substituents including electron-withdrawing groups like nitro and chloro, as well as heteroaromatic nitriles like pyridine-carbonitriles. It also tolerates dinitrile substrates, making it versatile for synthesizing diverse tetrazole derivatives used in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted Tetrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocycle synthesis plays in the rapid development of life-saving medications. Our technical team has extensively analyzed the potential of the tungstate-catalyzed route described in CN100591674C and is fully prepared to implement this technology to meet your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require milligram quantities for screening or metric tons for commercial launch, our facilities are equipped to deliver. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how switching to this catalytic system can optimize your specific bill of materials. We encourage you to contact us today to discuss your requirements,索取 specific COA data for our tetrazole library, and obtain comprehensive route feasibility assessments tailored to your target molecules. Let us be your strategic partner in transforming innovative patent chemistry into commercial reality.
