Advanced Selective Synthesis of Fluorochemical Monoisocyanates for Commercial Scale-up
Advanced Selective Synthesis of Fluorochemical Monoisocyanates for Commercial Scale-up
The development of environmentally sustainable fluorinated materials has become a critical priority for the global specialty chemicals industry, driven by increasing regulatory scrutiny regarding bioaccumulative substances. Patent CN1902164A introduces a groundbreaking process for the selective preparation of fluorochemical monoisocyanates, specifically targeting the synthesis of short-chain perfluorinated intermediates that serve as precursors for water and oil-repellent acrylic resins. This technology addresses the urgent market demand for alternatives to long-chain perfluorooctyl compounds, which are known to persist in living organisms, by utilizing fluorochemical alcohols with carbon chain lengths of n=2 to 5. The innovation lies not only in the molecular design but also in the reaction engineering, which employs a selective precipitation strategy to achieve high purity levels exceeding 85% without the need for energy-intensive purification steps. For R&D directors and procurement specialists seeking reliable fluorochemical intermediate suppliers, this patent represents a significant leap forward in balancing performance with environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorochemical isocyanates relied heavily on processes that required substantial excesses of diisocyanate reactants to drive conversion, often leading to complex downstream purification challenges. Traditional methods, such as those referenced in US Patent 5,446,118, frequently resulted in reaction mixtures containing significant amounts of unreacted diisocyanate and bis-urethane byproducts, necessitating rigorous distillation or chromatographic separation to isolate the desired monoisocyanate. These purification steps are not only capital-intensive but also pose safety risks due to the thermal instability of isocyanates at elevated temperatures. Furthermore, conventional solvents often kept the product in solution, preventing the natural separation of the target compound from the reaction matrix, thereby increasing the overall processing time and waste generation. The reliance on long-chain perfluorinated precursors in older methodologies also created a supply chain vulnerability as global regulations began to restrict the use of C8-based chemistry due to toxicity concerns.
The Novel Approach
The novel approach detailed in the patent data utilizes a clever manipulation of solubility parameters to drive the reaction equilibrium and facilitate product isolation simultaneously. By conducting the reaction between fluorochemical alcohols and 4,4'-diphenylmethane diisocyanate (MDI) in a non-polar solvent where the resulting monoisocyanate is insoluble, the product precipitates out of the solution as it forms. This selective precipitation effectively removes the product from the reactive environment, preventing further reaction to form bis-urethanes and minimizing the need for excess diisocyanate. The process operates efficiently at mild temperatures ranging from 25°C to 50°C, significantly reducing energy consumption compared to high-temperature distillation methods. This methodology allows for the direct filtration and drying of the product to obtain a white powder with high purity, streamlining the manufacturing workflow and enhancing the economic viability of producing short-chain fluorochemical intermediates for commercial applications.
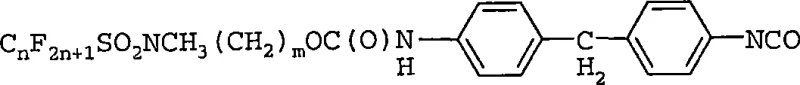
Mechanistic Insights into MDI-Mediated Urethane Formation
The core chemical transformation involves the nucleophilic attack of the hydroxyl group of the fluorochemical alcohol on the electrophilic carbon of the isocyanate group in MDI. This reaction is typically catalyzed by organotin compounds such as dibutyltin dilaurate (DBTDL) or tertiary amines like DABCO, which activate the isocyanate group towards nucleophilic addition. The mechanism proceeds through a transition state where the catalyst coordinates with the isocyanate, increasing its susceptibility to attack by the alcohol oxygen. As the urethane linkage forms, the steric bulk of the perfluorinated chain combined with the rigid aromatic structure of the MDI moiety reduces the solubility of the resulting molecule in non-polar hydrocarbon solvents like n-heptane. This physicochemical property is exploited to drive the reaction forward; as the product precipitates, Le Chatelier's principle dictates that the equilibrium shifts to favor further product formation, thereby maximizing yield without requiring a large molar excess of the expensive fluorochemical alcohol.
Impurity control is inherently managed through this precipitation mechanism, as the soluble byproducts and unreacted starting materials remain in the supernatant liquid phase. The patent data indicates that maintaining a molar ratio of fluorochemical alcohol to MDI between 1:1 and 1:1.5 is optimal for minimizing the formation of the bis-adduct, where both isocyanate groups of the MDI react. If the ratio of MDI is too high, there is an increased risk of forming oligomeric species or leaving residual free isocyanate in the solid cake, although the washing steps with fresh solvent effectively remove these surface impurities. The choice of solvent is critical; solvents with a Hildebrand solubility parameter less than 8.3 (cal/cm3)1/2 and a low hydrogen bond index ensure that the polar urethane-fluorochemical hybrid molecule crashes out of solution immediately upon formation, acting as a self-purifying crystallization process.
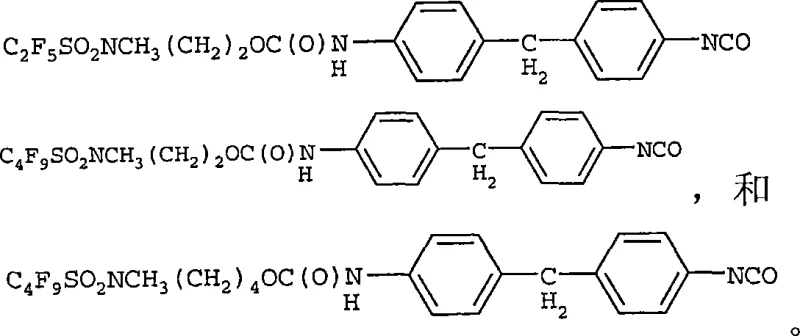
How to Synthesize Fluorochemical Monoisocyanates Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of solvent selection and temperature control to achieve consistent quality. The process begins with the preparation of a dry, non-polar reaction medium, followed by the controlled addition of reactants to manage the exotherm and particle size of the precipitating solid. Detailed operational parameters, including specific stirring rates and addition times, are crucial for preventing agglomeration and ensuring uniform particle growth, which facilitates efficient filtration. The following guide summarizes the critical operational steps derived from the experimental examples provided in the intellectual property documentation.
- Prepare the reaction vessel with a non-polar solvent such as n-heptane or petroleum ether, ensuring the solvent has a Hildebrand solubility parameter less than 8.3 (cal/cm3)1/2.
- Dissolve 4,4'-diphenylmethane diisocyanate (MDI) in the solvent and add a catalytic amount of dibutyltin dilaurate (DBTDL) or a tertiary amine catalyst.
- Slowly add the fluorochemical alcohol (e.g., C4F9SO2NCH3(CH2)2OH) to the MDI solution at 25°C to 50°C, maintaining a molar ratio between 1: 1 and 1:1.5 to induce selective precipitation of the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this selective precipitation technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of complex purification trains, such as vacuum distillation columns required for heat-sensitive isocyanates, translates directly into reduced capital expenditure and lower operating costs for manufacturing facilities. By avoiding the need for extensive downstream processing, manufacturers can significantly shorten the production cycle time, allowing for faster turnaround on customer orders and improved responsiveness to market fluctuations. The ability to use stoichiometric or near-stoichiometric amounts of MDI reduces raw material waste, contributing to a more sustainable and cost-effective supply chain model that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The process drastically simplifies the production workflow by integrating reaction and separation into a single unit operation, removing the need for expensive solvent recovery systems associated with distillation. Since the product is isolated via simple filtration and drying, the energy intensity of the process is markedly lower than conventional methods, leading to substantial utility savings. Furthermore, the reduced requirement for excess diisocyanate minimizes the cost of goods sold by optimizing raw material utilization efficiency, ensuring that every kilogram of input contributes maximally to the final yield.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically MDI and short-chain fluorochemical alcohols, are commercially available from major global chemical suppliers, ensuring a stable and diversified supply base. The robustness of the reaction conditions, which tolerate mild temperatures and ambient pressure, reduces the risk of production delays caused by equipment failure or stringent safety interlocks often associated with high-pressure reactors. This reliability ensures consistent delivery schedules for downstream customers in the coatings and textile industries who depend on a steady flow of high-quality intermediates for their own formulation processes.
- Scalability and Environmental Compliance: The use of short-chain perfluorinated groups addresses the growing regulatory pressure against long-chain PFAS, future-proofing the supply chain against potential bans or restrictions. The process generates less hazardous waste due to the absence of heavy purification steps and the ability to recycle the non-polar solvent washings, supporting a greener manufacturing footprint. Scalability is enhanced by the simplicity of the solid-liquid separation step, which can be easily adapted from laboratory filter funnels to industrial centrifuges or filter presses without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorochemical synthesis technology. These insights are derived directly from the experimental data and claims within the patent specification, providing clarity on purity standards, solvent compatibility, and application suitability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing polymer formulations.
Q: What is the primary advantage of the selective precipitation method described in CN1902164A?
A: The primary advantage is the ability to produce fluorochemical monoisocyanates with greater than 85% purity without requiring complex purification steps like distillation, significantly simplifying the downstream processing.
Q: Why are short-chain perfluorinated groups preferred over long-chain groups in this synthesis?
A: Short-chain perfluorinated groups (n=2 to 5) are preferred because they exhibit significantly lower bioaccumulation potential and toxicity compared to traditional long-chain (C8+) perfluorinated compounds, aligning with modern environmental regulations.
Q: Which solvents are most effective for this reaction process?
A: Non-polar solvents where the product is insoluble are most effective, specifically n-heptane, petroleum ether, or methyl nonafluorobutyl ether (HFE-7100), as they facilitate the immediate precipitation of the solid product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorochemical Monoisocyanates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of transitioning to safer, high-performance fluorinated intermediates that meet both regulatory standards and end-user performance expectations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorochemical monoisocyanate meets the exacting requirements necessary for producing premium water and oil-repellent coatings.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this precipitation-based method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific application needs, ensuring a reliable partnership for your specialty chemical requirements.
