Optimizing Nadifloxacin Intermediate Production via Advanced Cyclization Technology
Optimizing Nadifloxacin Intermediate Production via Advanced Cyclization Technology
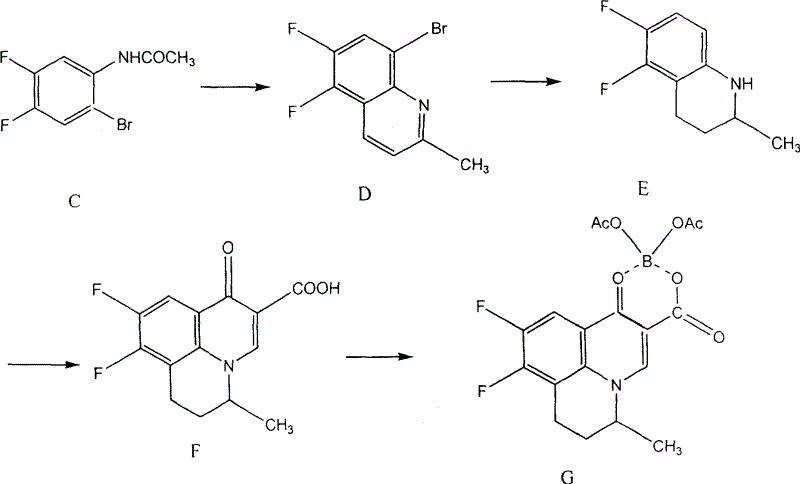
The pharmaceutical industry constantly seeks robust synthetic routes for broad-spectrum antibiotics, particularly for dermatological applications where agents like Nadifloxacin play a pivotal role. Patent CN100462357C introduces a transformative preparation method for 8-bromo-5,6-difluoro-2-methylquinoline, a critical pharmaceutical intermediate in the synthesis of this potent antibacterial drug. This technical insight report dissects the proprietary chemistry disclosed in the patent, highlighting how shifting from solid to liquid oxidants in the cyclization step resolves long-standing efficiency bottlenecks. For R&D directors and procurement strategists, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at competitive margins. The following analysis details the mechanistic advantages and commercial implications of this optimized pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-bromo-5,6-difluoro-2-methylquinoline has relied on cyclization protocols utilizing sodium m-nitrobenzenesulfonate as the primary oxidizing agent. While chemically feasible, this conventional approach presents significant logistical and economic drawbacks for large-scale manufacturing. The solid nature of sodium m-nitrobenzenesulfonate necessitates the use of substantial volumes of acidic solvents, typically concentrated hydrochloric acid, to maintain reaction homogeneity. This excessive solvent consumption not only inflates raw material costs but also complicates the downstream workup, requiring large quantities of alkali for neutralization. Furthermore, literature and patent data indicate that this traditional route suffers from suboptimal conversion rates, often yielding less than 60% of the desired quinoline derivative, which severely impacts the overall cost of goods for the final antibiotic API.
The Novel Approach
The innovative methodology described in the patent fundamentally alters the oxidation landscape by substituting the expensive solid oxidant with liquid alternatives, specifically concentrated sulfuric acid or concentrated nitric acid. This strategic substitution offers immediate benefits in terms of reagent availability and handling safety, as these mineral acids are commodity chemicals with stable supply chains. By employing liquid oxidants, the process effectively reduces the dependency on concentrated hydrochloric acid, acting partially as the solvent medium itself. This dual functionality streamlines the reaction mixture, leading to a drastic reduction in the volume of waste acid generated. Consequently, the post-reaction neutralization step requires significantly less base, directly translating to lower operational expenditures and a reduced environmental footprint for the manufacturing facility.
Mechanistic Insights into Liquid Phase Oxidative Cyclization
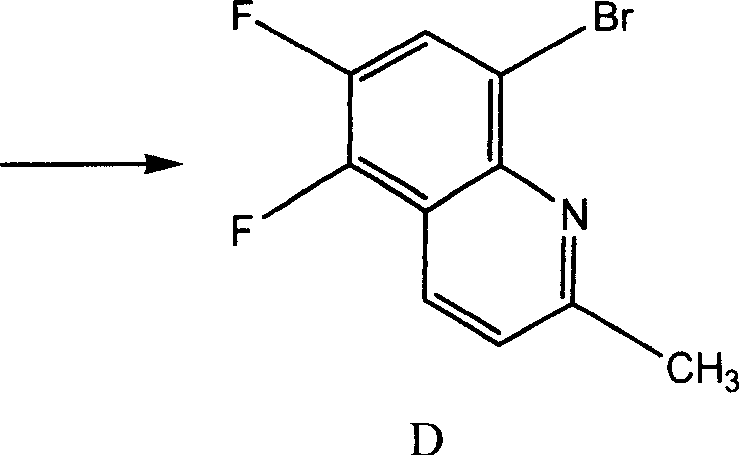
The core of this technological advancement lies in the oxidative cyclization of 3,4-difluoro-6-bromoacetanilide with trans-crotonaldehyde. In this mechanism, the choice of oxidant plays a critical role in facilitating the aromatization of the dihydroquinoline intermediate to the final quinoline structure. Concentrated sulfuric and nitric acids provide a highly acidic and oxidizing environment that promotes the dehydration and dehydrogenation steps necessary for ring closure. Unlike the solid sulfonate oxidant which may suffer from mass transfer limitations due to poor solubility, the liquid acids ensure intimate contact between the reactants and the oxidizing species. This homogeneous reaction environment minimizes side reactions and polymerization of the aldehyde component, thereby enhancing the selectivity towards the target 8-bromo-5,6-difluoro-2-methylquinoline scaffold.
Impurity control is another paramount consideration in the synthesis of fluoroquinolone intermediates, where halogenated byproducts can be difficult to remove. The use of liquid mineral acids allows for tighter thermal control within the 80-105°C range specified in the patent. Maintaining this temperature window is crucial; temperatures that are too low result in incomplete cyclization, while excessive heat can lead to decomposition of the sensitive fluorinated aromatic ring. The inclusion of a moderator, such as boric acid, further refines the reaction kinetics by buffering the acidity and preventing runaway exotherms. This precise control over the reaction parameters ensures a cleaner crude product profile, reducing the burden on purification units and ensuring that the final intermediate meets the stringent purity specifications required for subsequent coupling reactions in Nadifloxacin production.
How to Synthesize 8-Bromo-5,6-Difluoro-2-Methylquinoline Efficiently
Implementing this synthesis route requires careful attention to the sequential preparation of precursors before the final cyclization event. The process begins with the acylation of 3,4-difluoroaniline to protect the amine group, followed by a regioselective bromination to install the critical halogen atom at the 8-position. These preliminary steps set the stage for the high-yield cyclization discussed previously. Operators must adhere to strict molar ratios, particularly regarding the oxidant to substrate ratio which is optimized between 0.8 to 1.5 equivalents. The detailed standardized operating procedures for executing these steps safely and efficiently are outlined below, providing a roadmap for process chemists to replicate the patent's success in a pilot or production setting.
- Prepare 3,4-difluoroacetanilide by acylating 3,4-difluoroaniline with acetic anhydride in the presence of a catalyst.
- Synthesize 3,4-difluoro-6-bromoacetanilide via bromination using elemental bromine and an iodine catalyst in acetic acid.
- Perform the key cyclization reaction using concentrated sulfuric or nitric acid as the oxidant, reacting with trans-crotonaldehyde at 80-105°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of specialized solid oxidants removes a potential single-point-of-failure in the supply chain, as mineral acids are universally available from multiple vendors globally. This diversification of raw material sources enhances supply security and mitigates the risk of price volatility associated with niche chemical reagents. Furthermore, the reduction in solvent and neutralization agent consumption directly lowers the variable costs per kilogram of production. These efficiencies compound over large production runs, offering substantial margin improvements for contract manufacturing organizations and API producers alike without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The replacement of sodium m-nitrobenzenesulfonate with concentrated sulfuric or nitric acid eliminates the premium pricing associated with specialized oxidants. Additionally, the liquid nature of the new reagents reduces the volume of hydrochloric acid required, which subsequently lowers the cost of caustic soda needed for waste neutralization. This cascading effect of savings across multiple input categories results in a leaner cost structure for the entire production campaign.
- Enhanced Supply Chain Reliability: Sourcing commodity chemicals like sulfuric acid and nitric acid is inherently more stable than procuring fine chemical oxidants. This reliability ensures consistent production schedules and reduces the likelihood of delays caused by raw material shortages. For global supply chains, the ability to source these acids locally in various regions further reduces logistics costs and lead times, ensuring a steady flow of intermediates to downstream API synthesis sites.
- Scalability and Environmental Compliance: The simplified workup procedure, characterized by reduced salt generation from neutralization, eases the burden on wastewater treatment facilities. This environmental benefit is increasingly critical as regulatory scrutiny on pharmaceutical effluent intensifies. Moreover, the robust nature of the liquid-phase reaction supports seamless scale-up from laboratory to multi-ton production, allowing manufacturers to respond rapidly to market demand fluctuations for Nadifloxacin and related derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of using liquid oxidants in this synthesis?
A: Using concentrated sulfuric or nitric acid replaces expensive solid oxidants like sodium m-nitrobenzenesulfonate, significantly reducing raw material costs and simplifying post-treatment neutralization.
Q: How does this method improve yield compared to conventional routes?
A: The patented cyclization process achieves a yield increase of over 7% compared to prior art methods, pushing total yields above 65-73% which is critical for commercial viability.
Q: Is this process scalable for industrial antibiotic production?
A: Yes, the use of readily available liquid reagents and moderate temperatures (80-105°C) makes the process highly suitable for large-scale manufacturing with improved safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Bromo-5,6-Difluoro-2-Methylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 8-bromo-5,6-difluoro-2-methylquinoline meets the exacting standards required for GMP API synthesis. Our infrastructure is designed to handle complex fluorinated chemistries safely and efficiently.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical strategy available in the market.
