Advanced Manufacturing of 4-Amino-3-Nitrophenol via Optimized One-Pot Nitration
Introduction to Advanced Intermediate Synthesis
The synthesis of 4-amino-3-nitrophenol represents a critical capability in the production of high-value fine chemical intermediates, particularly for the pharmaceutical and dye industries. As detailed in patent CN101066929B, a novel manufacturing route has been developed that fundamentally restructures the traditional approach to accessing this vital molecular scaffold. This technology leverages a streamlined one-pot strategy that combines acetylation and nitration steps, thereby eliminating the need for intermediate isolation and significantly reducing processing time. For R&D directors and procurement specialists seeking a reliable 4-amino-3-nitrophenol supplier, understanding the mechanistic advantages of this process is essential for evaluating supply chain resilience. The method utilizes p-aminophenol as a cost-effective starting material, reacting it with acetic anhydride to form a protected intermediate which is subsequently nitrated without changing reactors. This integration not only simplifies the operational workflow but also minimizes the generation of waste acid, addressing key environmental compliance concerns that modern chemical manufacturers face today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-3-nitrophenol has been plagued by significant safety hazards and low efficiency, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Prior art, such as the route reported by Kehrmann, relied heavily on the use of o-azidonitrobenzene as a precursor, a compound known for its inherent instability and potential for explosive decomposition. Furthermore, the selective reduction required in these legacy pathways is notoriously difficult to control, often resulting in poor yields and a complex impurity profile that necessitates expensive downstream purification. The reliance on hazardous azide chemistry imposes severe restrictions on plant operations, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Additionally, multi-step processes that involve isolating unstable intermediates increase the risk of material loss and exposure to toxic substances, making them economically unviable for large-scale production in a competitive market environment.
The Novel Approach
In stark contrast to these dangerous legacy methods, the innovative process described in the patent data utilizes a robust and safe pathway starting from readily available p-aminophenol. By employing acetic anhydride as both an acylating agent and a solvent component, the reaction system effectively protects the amino functionality prior to the harsh nitration step. This strategic protection prevents the oxidative degradation of the amine group, which is a common side reaction when exposed to strong oxidizing agents like nitric acid. The ability to perform both acetylation and nitration in a single reactor vessel drastically reduces the physical footprint of the manufacturing process and eliminates the need for transferring reactive intermediates between different units. This consolidation of unit operations leads to a substantial reduction in labor costs and energy consumption, while the use of commodity reagents ensures a stable and predictable supply chain for raw materials.
Mechanistic Insights into One-Pot Acetylation-Nitration
The core chemical innovation lies in the precise control of electrophilic aromatic substitution facilitated by the acetyl protecting groups. During the initial phase, p-aminophenol reacts with acetic anhydride in a molar ratio ranging from 1:2.0 to 1:3.0 to form N-(4-hydroxyphenyl)acetamide derivatives in situ. The acetyl group on the nitrogen atom decreases the electron density of the aromatic ring slightly but, more importantly, prevents the formation of tarry oxidation byproducts that typically occur with free amines under acidic conditions. Subsequently, the introduction of 98% nitric acid (5-7 molar equivalents) generates the nitronium ion necessary for electrophilic attack. The presence of the acetoxy group at the para-position and the acetamido group directs the incoming nitro group specifically to the ortho-position relative to the oxygen, ensuring high regioselectivity for the 3-nitro isomer. This electronic directing effect is crucial for minimizing the formation of undesired isomers that would complicate the purification of high-purity pharmaceutical intermediates.
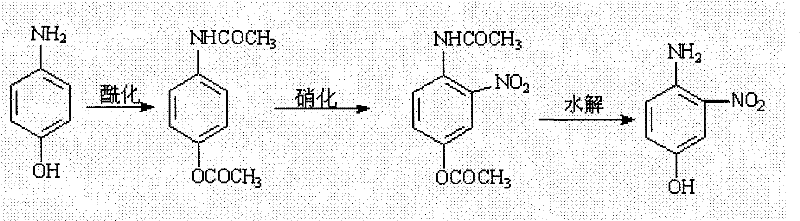
Following the nitration event, the reaction mixture is quenched in ice water to precipitate the intermediate 2-acetamido-5-acetoxynitrobenzene as yellow crystals. The subsequent hydrolysis step is equally critical, utilizing sodium hydroxide solutions with concentrations between 1mol/L and 5mol/L to cleave both acetyl groups simultaneously. The mechanism involves nucleophilic attack by hydroxide ions on the carbonyl carbons of the ester and amide functionalities. Maintaining the reaction temperature between 50-100°C ensures complete deprotection without degrading the sensitive nitro-phenolic structure. Finally, careful pH adjustment to the range of 3-4 using hydrochloric acid protonates the amino group and precipitates the final zwitterionic product, effectively separating it from inorganic salts and soluble organic impurities. This rigorous control over pH and temperature is what allows the process to achieve yields exceeding 75% in pilot examples, demonstrating excellent reproducibility.
How to Synthesize 4-Amino-3-Nitrophenol Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent to ensure optimal yield and safety. The process begins with the reflux of p-aminophenol and acetic anhydride, followed by controlled cooling before the exothermic addition of nitric acid. Operators must monitor the temperature closely during the nitration phase to prevent runaway reactions, adding dilute nitric acid in a secondary stage to maximize conversion. The isolation of the intermediate via filtration and washing is a standard unit operation that prepares the solid for the final hydrolysis step. For detailed procedural specifics regarding reagent addition rates and stirring speeds, please refer to the standardized synthesis guide below which breaks down the exact operational parameters.
- Perform acetylation of p-aminophenol with acetic anhydride at reflux, followed by direct nitration in the same reactor using concentrated nitric acid.
- Isolate the intermediate 2-acetamido-5-acetoxynitrobenzene by pouring the reaction mixture into ice water, filtering, and washing to neutrality.
- Hydrolyze the isolated intermediate using sodium hydroxide solution at 50-100°C, then adjust pH to 3-4 with hydrochloric acid to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing technology offers profound advantages for cost reduction in pharmaceutical intermediates manufacturing by fundamentally simplifying the production workflow. The elimination of hazardous azide reagents removes the need for expensive safety infrastructure and specialized waste disposal services associated with explosive materials. Furthermore, the one-pot nature of the acetylation and nitration steps significantly reduces the cycle time per batch, allowing manufacturing facilities to increase throughput without expanding their physical plant capacity. The use of acetic anhydride and nitric acid, which are bulk commodity chemicals, ensures that raw material costs remain low and supply is not subject to the volatility often seen with specialty reagents. This stability in input costs allows for more accurate long-term pricing contracts with downstream customers in the dye and API sectors.
- Cost Reduction in Manufacturing: The integration of acetylation and nitration into a single reactor vessel eliminates the need for intermediate drying and transfer operations, which are traditionally labor-intensive and energy-consuming steps. By reducing the number of unit operations, the process lowers utility consumption for heating and cooling, directly impacting the variable cost of goods sold. Additionally, the recycling of acetic acid generated during the reaction as a solvent component further minimizes raw material waste, contributing to a leaner and more economical production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Sourcing p-aminophenol as the primary feedstock provides a significant logistical advantage, as it is a widely produced chemical with a robust global supply network. Unlike niche precursors that may suffer from shortages or long lead times, the availability of p-aminophenol ensures continuous production scheduling and reduces the risk of stockouts. The simplified process flow also means that manufacturing can be easily replicated across different sites, providing redundancy and flexibility in the supply chain to meet fluctuating market demands for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates significantly less waste acid compared to traditional nitration methods due to the optimized stoichiometry and the dual role of reaction byproducts as solvents. This reduction in effluent volume simplifies wastewater treatment requirements and lowers the environmental compliance burden on the manufacturing site. The mild hydrolysis conditions and the absence of heavy metal catalysts mean that the final product is free from toxic metal residues, meeting stringent regulatory standards for pharmaceutical applications without the need for complex purification steps like chromatography.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and quality control of this specific synthesis route. Understanding these details is vital for partners evaluating the feasibility of integrating this intermediate into their own drug substance manufacturing processes. The answers provided are derived directly from the experimental data and technical specifications found within the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why is the acetylation step critical before nitration in this process?
A: Acetylation protects the sensitive amino group from oxidation by strong nitric acid and prevents polymerization, while also directing the nitro group to the desired ortho-position relative to the hydroxyl group.
Q: How does this method improve safety compared to traditional routes?
A: Unlike older methods that utilize hazardous azide compounds with explosion risks, this process relies on stable, commodity chemicals like p-aminophenol and acetic anhydride, significantly enhancing operational safety.
Q: What are the purity specifications achievable with this hydrolysis method?
A: By controlling the hydrolysis temperature between 50-100°C and carefully adjusting the pH to 3-4 during precipitation, the process yields high-purity crystals with melting points consistent with pharmaceutical grade standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-3-Nitrophenol Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our technical team is adept at optimizing the acetylation-nitration-hydrolysis sequence to meet stringent purity specifications required by global regulatory bodies. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our 4-amino-3-nitrophenol consistently meets the high standards expected by top-tier pharmaceutical and agrochemical companies. Our commitment to quality assurance extends beyond simple testing, encompassing a holistic approach to process safety and environmental stewardship.
We invite potential partners to contact our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies translate into competitive pricing for your supply chain. We are prepared to provide specific COA data and route feasibility assessments to support your vendor qualification process, ensuring a transparent and collaborative partnership focused on mutual growth and innovation in the fine chemicals sector.
