Scalable Total Synthesis of Daptomycin: A Strategic Breakthrough for API Manufacturing
Scalable Total Synthesis of Daptomycin: A Strategic Breakthrough for API Manufacturing
The pharmaceutical landscape for treating Gram-positive bacterial infections has been profoundly influenced by the introduction of lipopeptide antibiotics, with Daptomycin standing as a cornerstone therapy for complicated skin and soft tissue infections. As resistance to traditional agents like vancomycin escalates globally, the demand for reliable, high-purity sources of this critical active pharmaceutical ingredient (API) has never been more urgent. Patent CN101235080B introduces a transformative synthetic methodology that shifts the production paradigm from biological fermentation to a robust chemical synthesis involving a strategic solid-liquid phase combination. This innovation addresses the critical supply chain vulnerabilities associated with obtaining the natural producer strain, Streptomyces roseosporus, while offering a pathway to superior impurity control. By leveraging advanced peptide coupling technologies, this method enables the precise assembly of the complex cyclic structure, ensuring that the final product meets the stringent quality standards required for parenteral administration. The following analysis details how this chemical route provides a sustainable alternative for reliable API intermediate supplier networks seeking to diversify their sourcing strategies beyond fermentation-dependent models.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Daptomycin has relied almost exclusively on the fermentation of Streptomyces roseosporus, a biological process fraught with inherent inconsistencies and regulatory hurdles. Fermentation pathways are notoriously sensitive to minor fluctuations in nutrient media, temperature, and aeration, leading to significant batch-to-batch variability in yield and impurity profiles that complicate downstream purification. Furthermore, the reliance on a specific biological organism creates a single point of failure in the supply chain; any contamination or genetic drift in the seed culture can halt production for extended periods, jeopardizing supply chain reliability for hospitals and healthcare systems. The extraction of the antibiotic from a complex fermentation broth also necessitates extensive chromatographic separation steps to remove structurally related analogs and cellular debris, driving up the cost of goods sold (COGS) and generating substantial organic waste. Additionally, intellectual property constraints surrounding the natural producing strain have historically limited the number of qualified manufacturers, creating a monopolistic market environment that stifles competition and keeps prices artificially high for this life-saving medication.
The Novel Approach
In stark contrast to the biological limitations, the method disclosed in patent CN101235080B utilizes a hybrid solid-liquid phase synthesis strategy that offers unprecedented control over the molecular architecture of Daptomycin. This approach initiates with the anchoring of the C-terminal amino acid onto a 2-chlorotrityl chloride resin, allowing for the sequential addition of protected amino acids using standard Fmoc chemistry protocols that are well-established in the fine chemical industry. By constructing the linear decapeptide chain on a solid support, the method simplifies purification between each coupling step through simple filtration and washing, effectively removing excess reagents and soluble by-products without the need for complex isolation procedures. The transition to liquid phase for the critical macrocyclization step allows for precise optimization of reaction conditions, such as concentration and solvent choice, which are vital for forming the large lactone ring without inducing oligomerization. This chemical synthesis route not only bypasses the need for the difficult-to-obtain Streptomyces strain but also facilitates the production of structural analogs for research and development, thereby accelerating the discovery of next-generation antibiotics with improved pharmacokinetic properties.
Mechanistic Insights into Solid-Liquid Phase Peptide Assembly
The core of this synthetic breakthrough lies in the meticulous orchestration of protecting group strategies and coupling reagents to preserve the stereochemical integrity of the thirteen chiral centers present in the Daptomycin molecule. The process employs Fmoc (9-fluorenylmethoxycarbonyl) protection for the alpha-amino groups, which allows for orthogonal deprotection using mild bases like piperidine, ensuring that acid-labile side-chain protecting groups such as tBu and Boc remain intact during chain elongation. A critical mechanistic feature is the use of the DIC (N,N'-Diisopropylcarbodiimide) and DMAP (4-Dimethylaminopyridine) system for the esterification step involving the Kynurenine residue; this specific catalytic combination activates the carboxylic acid efficiently while minimizing the risk of racemization at the adjacent chiral center, a common pitfall in peptide synthesis. The subsequent macrocyclization is mediated by PyBOP (Benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate) and HOAT (1-Hydroxy-7-azabenzotriazole), reagents chosen for their ability to suppress epimerization and promote rapid amide bond formation even in dilute conditions required for intramolecular reactions. This careful selection of reagents ensures that the final cyclized product possesses the exact three-dimensional conformation necessary for binding to the bacterial cell membrane and exerting its bactericidal effect.
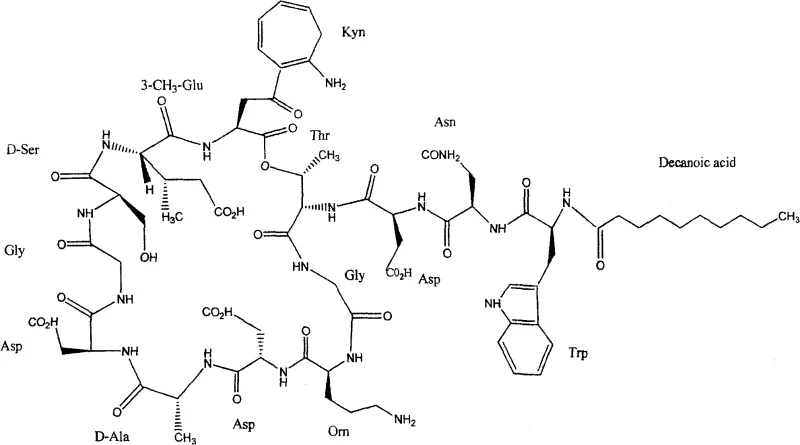
Impurity control is another paramount aspect of this mechanism, particularly regarding the formation of deletion sequences and diastereomers that can arise during the repetitive coupling cycles. The protocol mandates the use of the Kaiser test after each amino acid addition to monitor coupling completeness; if a positive result indicates unreacted amines, the coupling step is repeated to ensure quantitative conversion before proceeding to the next residue. This rigorous quality control checkpoint prevents the accumulation of truncated peptide chains that would otherwise co-elute with the target product during final purification, significantly reducing the burden on preparative HPLC columns. Furthermore, the choice of the 2-chlorotrityl linker allows for the cleavage of the fully protected peptide from the resin under very mild acidic conditions (1-2% TFA), which preserves the acid-sensitive ester linkage within the macrocycle that might be compromised by stronger acids like HF or TFMSA used in Boc chemistry. The final global deprotection utilizes a scavenger-rich cocktail of TFA, water, and thioanisole to capture reactive carbocations generated during the removal of Trt and tBu groups, preventing alkylation side reactions on the electron-rich indole ring of the Tryptophan residue and ensuring a clean final product profile.
How to Synthesize Daptomycin Efficiently
The execution of this synthetic route requires a seamless integration of automated solid-phase synthesizers for the linear assembly and specialized glassware reactors for the solution-phase cyclization and deprotection steps. Operators must strictly adhere to the specified molar ratios of coupling reagents, such as the 1:1:1 weight ratio of Fmoc-AA, DIC, and HOBt during standard elongation, and the 3:3:1 ratio involving DMAP for the difficult esterification steps, to maintain high reaction efficiency. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process chemists to replicate this high-yielding synthesis in a GMP-compliant environment.
- Perform solid-phase synthesis using 2-chlorotrityl chloride resin to assemble the protected decapeptide chain, sequentially adding Fmoc-protected amino acids and decanoic acid.
- Cleave the fully protected peptide from the resin using a mild acidic solution (1-2% TFA in DCM) to obtain the linear precursor.
- Execute liquid-phase macrocyclization using PyBOP and HOAT coupling reagents, followed by global deprotection with TFA/water/thioanisole mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from fermentation to this chemical synthesis route represents a strategic opportunity to mitigate risk and optimize cost structures within the antibiotic supply network. By decoupling production from biological variables, manufacturers can offer more predictable lead times and consistent pricing, shielding customers from the volatility often associated with agricultural-style fermentation processes. The ability to source high-quality starting materials, such as protected amino acids and resins, from multiple established chemical vendors further enhances supply security, reducing the dependency on single-source biological inputs that are prone to shortage. Moreover, the simplified purification workflow inherent in solid-phase synthesis reduces the consumption of expensive chromatography resins and solvents, translating into tangible economic benefits that can be passed down the value chain.
- Cost Reduction in Manufacturing: The elimination of fermentation tanks and the associated sterilization, aeration, and harvesting infrastructure drastically lowers the capital expenditure (CAPEX) and operational expenditure (OPEX) required for facility setup. Chemical synthesis allows for a smaller physical footprint and reduced energy consumption per kilogram of product, as the process does not require the maintenance of sterile conditions over multi-day fermentation cycles. Additionally, the recovery and recycling of solvents like DMF and DCM, which are used in large volumes during the washing and cleavage steps, can be implemented more efficiently in a chemical plant than in a biotech facility, leading to substantial cost savings in raw material procurement. The higher overall yield and purity achieved through the controlled stepwise assembly also mean less product loss during final purification, maximizing the output from every batch of starting materials.
- Enhanced Supply Chain Reliability: Unlike fermentation, which is susceptible to phage attacks and strain degeneration, chemical synthesis offers a robust and reproducible manufacturing platform that ensures continuous supply continuity. The modular nature of solid-phase peptide synthesis allows for rapid scale-up by simply increasing the batch size of the resin or running parallel reactors, enabling manufacturers to respond quickly to surges in market demand without the long lead times required to expand fermentation capacity. This agility is crucial for maintaining stock levels of critical antibiotics during public health emergencies or seasonal spikes in infection rates. Furthermore, the stability of the chemical intermediates allows for strategic stockpiling of key precursors, providing an additional buffer against potential disruptions in the global logistics network.
- Scalability and Environmental Compliance: The process is inherently scalable from gram-scale R&D to multi-ton commercial production using standard chemical engineering unit operations, facilitating a smooth technology transfer from the laboratory to the pilot and production plants. From an environmental perspective, the containment of reagents on the solid support minimizes the release of hazardous chemicals into the wastewater stream compared to the nutrient-rich effluent of fermentation, simplifying waste treatment compliance. The use of modern coupling reagents and the potential for implementing green chemistry principles, such as solvent substitution or flow chemistry adaptations in the liquid phase steps, further aligns the manufacturing process with increasingly stringent global environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology, providing clarity for stakeholders evaluating the feasibility of adopting this new supply source. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of the solid-liquid combined synthesis method for Daptomycin compared to fermentation?
A: The solid-liquid combined method eliminates the reliance on Streptomyces roseosporus fermentation, which is subject to biological variability and patent restrictions. This chemical approach offers better control over impurity profiles, consistent batch-to-batch reproducibility, and avoids the complex downstream purification required for fermentation broths.
Q: How does the DIC/DMAP system improve the esterification step in this synthesis?
A: The use of the DIC/DMAP system for esterification, specifically when connecting the Kynurenine residue, provides ideal reaction kinetics and minimizes racemization. This ensures high stereochemical purity at the critical ester linkage, which is essential for the biological activity of the final lipopeptide antibiotic.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. By utilizing standard solid-phase peptide synthesis (SPPS) equipment for the linear assembly and transitioning to liquid phase only for the critical cyclization step, the process balances the efficiency of automation with the precision of solution-phase chemistry, making it viable for multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Daptomycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex antibiotics like Daptomycin for the global healthcare market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles against international pharmacopoeia standards. Our dedication to quality assurance means that every batch of Daptomycin intermediate or API produced under our supervision undergoes comprehensive testing to guarantee safety and efficacy for the end patient.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this advanced solid-liquid synthesis route can be integrated into your supply chain to drive value and security. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and regional market dynamics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your position in the competitive antibiotic marketplace.
