Advanced Plerixafor Manufacturing: Overcoming Purification Barriers for Commercial Scale-up
Advanced Plerixafor Manufacturing: Overcoming Purification Barriers for Commercial Scale-up
The pharmaceutical landscape for hematopoietic stem cell mobilizers has been significantly advanced by the development of robust synthetic routes for Plerixafor, a critical CXCR4 antagonist. As detailed in the groundbreaking patent CN102584732B, a novel methodology has emerged that addresses the longstanding challenges of low yield and complex purification associated with earlier synthesis attempts. This technical disclosure outlines a sophisticated approach utilizing tri-tosyl protection and segmental crystallization, which collectively eliminate the need for cumbersome column chromatography while ensuring exceptional product quality. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding these mechanistic improvements is vital for securing a stable supply of high-purity active ingredients. The structural complexity of the target molecule, characterized by its macrocyclic tetraamine architecture linked by a xylylene bridge, demands precise control over reaction conditions to prevent oligomerization and side reactions.
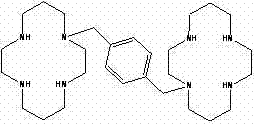
Furthermore, the ability to consistently achieve purity levels exceeding 99.5 percent with single impurity content maintained below 0.1 percent represents a paradigm shift in the manufacturing of this potent therapeutic agent. By leveraging mixed solvent systems and controlled temperature gradients during crystallization, the process effectively segregates unwanted byproducts that typically plague conventional syntheses. This level of purity is not merely a regulatory checkbox but a fundamental requirement for ensuring patient safety in oncology applications where Plerixafor is utilized alongside G-CSF. Consequently, this patent provides the foundational chemistry necessary for scaling production from laboratory benchtops to multi-ton commercial facilities without compromising on the stringent quality standards demanded by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Plerixafor and its analogues have historically suffered from significant operational inefficiencies that hinder cost reduction in API manufacturing. For instance, earlier routes described in literature often relied heavily on column chromatography for the purification of every intermediate, a technique that is notoriously difficult to scale and results in substantial solvent waste and product loss. Other approaches utilizing phosphorus-based protection groups faced severe challenges during the final deprotection step, where the stability of the protecting group led to incomplete reactions and prolonged production cycles. Additionally, routes employing metal-based protections required strictly anhydrous and oxygen-free conditions, imposing heavy burdens on equipment requirements and operational safety protocols. These traditional methods frequently yielded products with impurity profiles that exceeded acceptable limits, necessitating repeated recrystallization efforts that further eroded overall yield and economic viability.
The Novel Approach
In stark contrast, the innovative strategy disclosed in the patent introduces a streamlined pathway that replaces complex purification steps with efficient segmental crystallization techniques. By selecting p-toluenesulfonyl chloride as the protecting agent, the synthesis benefits from the formation of intermediates that possess favorable solubility characteristics, allowing for precise isolation through temperature-controlled crystallization rather than adsorption chromatography. This shift not only drastically simplifies the post-treatment workflow but also enhances the overall throughput of the manufacturing line by reducing cycle times. The bridging reaction with alpha,alpha'-dibromo-p-xylene is optimized in anhydrous acetonitrile with specific acid-binding agents, ensuring high conversion rates while minimizing the formation of polymeric byproducts. Ultimately, this novel approach transforms the synthesis from a labor-intensive, low-yield process into a robust, industrially viable operation capable of meeting the rigorous demands of the global pharmaceutical market.
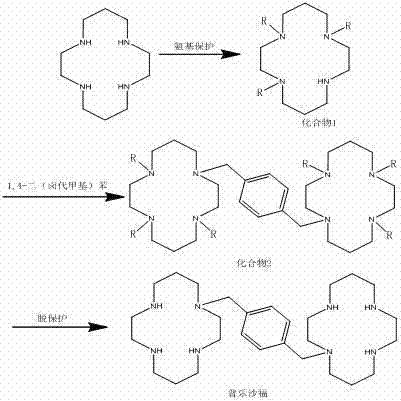
Mechanistic Insights into Tri-Tosyl Protection and Crystallization
The core of this synthetic breakthrough lies in the meticulous control of the protection and deprotection cycles, specifically utilizing the tri-tosyl derivative of 1,4,8,11-tetraazacyclotetradecane. Mechanistically, the reaction of the starting macrocycle with p-toluenesulfonyl chloride is conducted in methylene dichloride with DIPEA as an acid scavenger, creating a kinetic environment that favors the formation of the tri-substituted species over the tetra-substituted analogue. This selectivity is crucial because the remaining free amine serves as the nucleophilic site for the subsequent alkylation with the dibromo-xylene linker. The patent highlights that the purity of this tri-protected intermediate directly dictates the quality of the final product; therefore, the implementation of a mixed solvent system comprising lower alkyl alcohols and aprotic solvents is essential. By manipulating the polarity and temperature of this solvent matrix, operators can induce the precipitation of the desired intermediate while keeping impurities in solution, a phenomenon driven by subtle differences in lattice energy and solvation shells.
Following the bridging alkylation, the deprotection mechanism involves the cleavage of the sulfonamide bonds using a mixture of glacial acetic acid and hydrochloric acid under reflux conditions. This acidic environment protonates the nitrogen atoms, facilitating the departure of the tosyl groups and regenerating the free amines required for biological activity. However, the true innovation is found in the final purification stage, where the crude Plerixafor is subjected to a multi-stage crystallization protocol involving tetrahydrofuran and non-polar anti-solvents like n-hexane. This process exploits the differential solubility of the target molecule versus residual organic impurities at varying temperatures, specifically cooling from 40-45 degrees Celsius down to 0-5 degrees Celsius in controlled increments. Such precision ensures that the crystal lattice forms slowly and orderly, excluding impurity molecules and resulting in a final product that meets the strict specifications for injection-grade raw materials without the need for further chromatographic polishing.
How to Synthesize Plerixafor Efficiently
The practical execution of this synthesis requires careful attention to solvent ratios and temperature profiles to maximize the efficiency of the segmental crystallization steps. Operators must strictly adhere to the specified molar ratios of reactants and the sequence of solvent additions to ensure that the tri-protected intermediate precipitates with the highest possible purity before proceeding to the bridging reaction. The detailed standardized synthesis steps outlined below provide a roadmap for replicating the high yields and exceptional purity reported in the patent embodiments, serving as a critical guide for process chemists aiming to implement this technology. By following these protocols, manufacturing teams can avoid the common pitfalls of over-protection or incomplete alkylation that often plague macrocyclic syntheses. The integration of these specific crystallization parameters transforms what was once a finicky laboratory procedure into a predictable and scalable industrial process.
- React 1,4,8,11-tetraazacyclotetradecane with p-toluenesulfonyl chloride using segmental crystallization to obtain the tri-protected intermediate with purity greater than 95%.
- Perform a bridging reaction between the protected intermediate and alpha,alpha'-dibromo-p-xylene in anhydrous acetonitrile with an acid-binding agent.
- Execute deprotection in mixed acid followed by pH adjustment and multi-stage crystallization to achieve final product purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound benefits that extend far beyond simple chemical yield improvements. The elimination of column chromatography represents a massive reduction in operational expenditure, as it removes the need for expensive silica gel, vast quantities of elution solvents, and the specialized equipment required for large-scale flash purification. This simplification of the downstream processing directly translates to significant cost savings in Plerixafor manufacturing, allowing suppliers to offer more competitive pricing without sacrificing margin. Furthermore, the reduced solvent consumption aligns perfectly with modern green chemistry initiatives, lowering the environmental footprint of the production facility and minimizing the costs associated with waste disposal and solvent recovery. For supply chain managers, this means a more resilient production line that is less susceptible to bottlenecks caused by complex purification stages.
- Cost Reduction in Manufacturing: The replacement of chromatographic purification with crystallization significantly lowers the variable costs associated with solvent purchase and disposal. By avoiding the use of large volumes of toxic eluents and stationary phases, the process becomes inherently more economical and safer to operate on a multi-kilogram scale. This efficiency gain allows for a more favorable cost structure, making the final API more accessible for healthcare systems while maintaining high profitability for manufacturers. Additionally, the higher overall yield achieved through reduced product loss during purification further amplifies these financial benefits, ensuring that raw material inputs are converted into saleable product with maximum efficiency.
- Enhanced Supply Chain Reliability: The robustness of the crystallization-based purification method ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug product manufacturers. Unlike column chromatography, which can suffer from channeling or variable flow rates that delay production, crystallization is a highly predictable unit operation that can be easily scaled and automated. This reliability reduces the risk of batch failures and reprocessing, thereby shortening the effective lead time for high-purity pharmaceutical intermediates. Suppliers utilizing this method can guarantee tighter delivery schedules and greater volume flexibility, providing peace of mind to procurement teams managing complex global logistics networks.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common reagents and standard reaction conditions that do not require exotic catalysts or extreme pressures. This ease of scale-up facilitates the rapid transition from pilot plant trials to full commercial production, enabling suppliers to respond quickly to surges in market demand. Moreover, the reduced reliance on hazardous solvents and the minimization of waste streams ensure that the manufacturing process remains compliant with increasingly stringent environmental regulations. This sustainability aspect not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, appealing to ethically conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Plerixafor synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality benchmarks associated with this technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production needs or for procurement officers assessing supplier capabilities. The answers provided reflect the consensus on best practices for achieving the reported purity and yield metrics in a commercial setting.
Q: How does the new method improve Plerixafor purity compared to conventional routes?
A: The patented method utilizes a unique segmental crystallization technique instead of column chromatography, effectively removing impurities to achieve purity greater than 99.5% and single impurity content below 0.1%.
Q: What are the key advantages of the tri-tosyl protection strategy?
A: Using p-toluenesulfonyl groups allows for easier handling and purification via crystallization compared to phosphorus or metal-based protections, significantly simplifying the post-treatment process and reducing solvent consumption.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the elimination of column chromatography and the use of standard crystallization steps make the process highly scalable, cost-effective, and compliant with strict environmental and safety regulations for mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plerixafor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality intermediates that meet the rigorous standards of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early clinical trials through to full market launch. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Plerixafor we produce adheres to the highest quality benchmarks, including the >99.5% purity threshold defined in leading patents. Our infrastructure is designed to handle complex macrocyclic syntheses with precision, leveraging advanced crystallization technologies to deliver products that are ready for immediate formulation.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of our production methods compared to traditional routes. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements. Let us demonstrate how our commitment to innovation and quality can make us your trusted partner in the successful commercialization of Plerixafor and other critical oncology therapeutics.
