Advanced Configuration Inversion for High-Purity N-Ethyl-L-Menthyl Formamide Manufacturing
The fine chemical industry continuously seeks efficient pathways to produce high-value cooling agents, specifically N-ethyl-L-menthyl formamide, which is renowned for its potent cooling sensation and lack of pungent odor compared to traditional menthol. A significant technological breakthrough in this domain is documented in patent CN109851522B, which discloses a novel method for preparing N-ethyl-L-menthyl formamide through the stereochemical inversion of its neo-isomer counterpart. This innovation addresses a critical bottleneck in the manufacturing of high-end daily chemical and food additives by transforming what was previously considered a difficult-to-separate impurity into a valuable product. By utilizing a specific base-catalyzed system, the process achieves high conversion rates under relatively mild conditions, offering a robust solution for industrial scale-up. This report analyzes the technical merits of this inversion strategy, providing actionable insights for R&D directors seeking purity optimization and procurement managers aiming for cost-effective supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
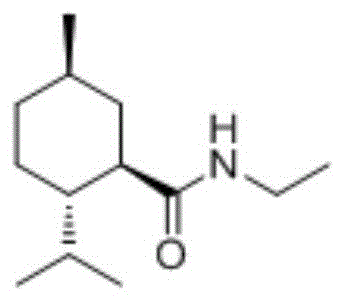
The Limitations of Conventional Methods
Traditionally, the production of N-ethyl-L-menthyl formamide has relied on multi-step syntheses starting directly from expensive L-menthol, or alternatively, through the dehydration condensation of menthic acid derivatives. A significant challenge in these conventional routes is the inevitable formation of stereoisomers, particularly the neo-isomer, which possesses inferior cooling properties and acts as an impurity. Prior art, such as the methods described in US20120283469A1, attempted to address isomer issues by inverting the configuration of menthic acid isomers using strong acid catalysts like p-toluenesulfonic acid or Lewis acids. However, these acid-catalyzed processes suffer from severe limitations, including the requirement for excessively high reaction temperatures often exceeding 175°C to achieve meaningful conversion. Furthermore, even under such harsh thermal conditions, the conversion rate of the isomer remains suboptimal at approximately 85%, necessitating complex and energy-intensive separation procedures to isolate the pure L-isomer. These factors collectively drive up equipment investment and operational costs, making the final product less competitive in the sensitive flavor and fragrance market.
The Novel Approach
In stark contrast to the harsh acidic conditions of the past, the method disclosed in CN109851522B introduces a sophisticated base-catalyzed inversion strategy that operates directly on the amide functionality. This novel approach utilizes sterically hindered tertiary amines as catalysts to facilitate the epimerization of N-ethyl-neo-menthyl formamide into the thermodynamically more stable N-ethyl-L-menthyl formamide. The process operates at significantly milder temperatures, typically ranging from 110°C to 150°C, which drastically reduces energy consumption and thermal stress on the reaction vessel. By targeting the amide directly rather than the precursor acid, the method streamlines the synthetic route, eliminating the need for hydrolysis and re-condensation steps. The result is a highly efficient transformation where conversion rates can reach approximately 91% with selectivity exceeding 99%, effectively turning a waste stream of neo-isomers into high-purity product. This shift represents a paradigm change in process chemistry, moving from separation-heavy workflows to conversion-focused manufacturing.
Mechanistic Insights into Base-Catalyzed Stereochemical Inversion
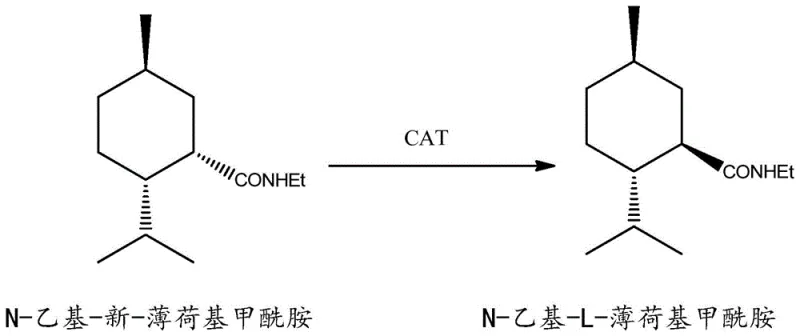
The core of this technological advancement lies in the precise mechanistic interaction between the substrate and the sterically hindered tertiary amine catalyst. Unlike small base molecules that might attack the carbonyl carbon leading to hydrolysis or side reactions, the bulky structure of catalysts such as N,N'-dicyclohexylmethylamine or N,N'-diisopropylethylamine prevents nucleophilic attack on the amide bond. Instead, these catalysts function exclusively as Brønsted bases, abstracting the acidic proton located at the alpha-carbon adjacent to the amide carbonyl group. This deprotonation generates a planar carbanion intermediate, which temporarily loses its stereochemical information. Subsequently, the proton is returned to the alpha-carbon from the less hindered face, favoring the formation of the more stable equatorial conformation found in the L-isomer. This delicate balance of steric bulk and basicity ensures that the reaction proceeds with high fidelity, minimizing degradation of the sensitive amide linkage while maximizing the thermodynamic drive towards the desired L-configuration.
Furthermore, the control of impurities is intrinsically built into this mechanism due to the high selectivity of the catalyst. The reaction environment is tuned such that the equilibrium strongly favors the L-isomer over the neo-isomer, driven by the lower steric strain of the L-configuration in the cyclohexane ring system. The use of inert solvents like toluene or cyclohexane further supports this selectivity by providing a non-polar medium that stabilizes the transition state without interfering with the proton transfer steps. Post-reaction workup is equally elegant; the catalyst and any residual acidic byproducts are easily removed through simple aqueous washing until a neutral pH is achieved. This simplicity in purification means that the final product requires minimal downstream processing to meet stringent quality standards for food and cosmetic applications, ensuring a clean impurity profile that is critical for regulatory compliance in global markets.
How to Synthesize N-Ethyl-L-Menthyl Formamide Efficiently
Implementing this configuration inversion process requires careful attention to reaction parameters to maximize the yield and purity of the final cooling agent. The procedure begins by dissolving the feedstock, which may be pure N-ethyl-neo-menthyl formamide or a mixture containing both neo and L isomers, into a suitable organic solvent such as toluene. The choice of solvent is critical, with a mass ratio of raw material to solvent typically maintained between 1:4 and 1:7 to ensure adequate solubility and heat transfer. Once the solution is prepared, a specific loading of the sterically hindered tertiary amine catalyst is introduced, usually in a mass ratio ranging from 10:1 to 60:1 relative to the substrate. The reaction mixture is then heated to a controlled temperature window of 120°C to 145°C under a protective nitrogen atmosphere to prevent oxidation. Stirring is continued for a duration of 20 to 36 hours to allow the equilibrium to shift fully towards the L-isomer. Following the reaction period, the mixture is cooled and subjected to a straightforward aqueous workup to remove the catalyst, followed by solvent recovery to isolate the high-purity product. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Dissolve the mixture of N-ethyl-neo-menthyl formamide and N-ethyl-L-menthyl formamide in an organic solvent such as toluene or cyclohexane.
- Add a sterically hindered tertiary amine catalyst (e.g., N,N'-dicyclohexylmethylamine) and heat the mixture to 110-150°C under nitrogen protection.
- After stirring for 20-36 hours, wash the reaction mixture with water to neutral pH, separate the organic layer, and recover the solvent to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this configuration inversion technology offers profound benefits for supply chain stability and cost management. Traditional manufacturing routes often suffer from low overall yields due to the accumulation of isomeric impurities that must be discarded or recycled through energy-intensive distillation columns. By chemically converting these impurities into the desired product, the effective yield of the manufacturing process is significantly increased without requiring additional raw material inputs. This improvement in atom economy translates directly into reduced raw material costs per kilogram of finished goods. Moreover, the ability to utilize mixed isomer feedstocks provides procurement teams with greater flexibility in sourcing, as they are no longer strictly dependent on high-purity starting materials that command premium pricing. This flexibility acts as a buffer against market volatility in the supply of natural menthol derivatives.
- Cost Reduction in Manufacturing: The elimination of harsh acidic conditions and high-temperature requirements leads to substantial savings in energy consumption and equipment maintenance. The mild reaction conditions allow for the use of standard stainless steel reactors rather than specialized high-pressure or corrosion-resistant vessels, lowering capital expenditure. Additionally, the simplified workup procedure involving simple water washes and distillation reduces the consumption of auxiliary chemicals and solvents. The high selectivity of the reaction minimizes the formation of byproducts that would otherwise require costly chromatographic separation or multiple recrystallization steps, thereby streamlining the entire production cycle and reducing labor costs associated with complex purification protocols.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with major FMCG clients. Because the process can tolerate variations in the initial isomer ratio of the feedstock, manufacturers are less vulnerable to supply disruptions of specific high-grade intermediates. The use of common, commercially available solvents like toluene and readily accessible tertiary amine catalysts further secures the supply chain against shortages of exotic reagents. This reliability enables producers to maintain steady inventory levels and meet tight delivery schedules, reinforcing their reputation as dependable partners in the global fragrance and flavor supply network.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method align perfectly with modern environmental regulations and corporate sustainability goals. The absence of heavy metal catalysts or corrosive mineral acids reduces the generation of hazardous waste streams, simplifying wastewater treatment and disposal compliance. The high atom utilization rate means less waste is generated per unit of product, contributing to a lower environmental footprint. Furthermore, the process is inherently scalable; the exothermic nature of the reaction is manageable under the specified conditions, allowing for safe expansion from pilot plant scales to multi-ton commercial production without significant re-engineering of the process flow, ensuring that supply can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this configuration inversion technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, aimed at clarifying the operational feasibility for potential licensees or manufacturing partners. Understanding these nuances is crucial for R&D teams planning to integrate this methodology into their existing production lines.
Q: What is the primary advantage of using a base catalyst over acid catalysts for this inversion?
A: Base catalysis using sterically hindered tertiary amines allows for milder reaction temperatures (110-150°C) compared to acid-catalyzed methods which often require temperatures above 175°C, resulting in higher conversion rates and better selectivity.
Q: Can this process handle mixtures containing significant amounts of the neo-isomer?
A: Yes, the process is specifically designed to handle mixtures where the molar ratio of neo-isomer to L-isomer varies, effectively converting the unwanted neo-isomer into the valuable L-isomer to maximize overall yield.
Q: Which solvents are compatible with this configuration inversion reaction?
A: The patent specifies non-polar organic solvents such as cyclohexane and toluene as preferred media, which facilitate the reaction while allowing for easy recovery and recycling via distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Ethyl-L-Menthyl Formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced stereochemical inversion technologies in the production of high-value cooling agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN109851522B are seamlessly translated into robust industrial operations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N-ethyl-L-menthyl formamide meets the exacting standards required by the global food and personal care industries. We are committed to leveraging our technical expertise to optimize yield and minimize impurities, delivering a product that provides the intense cooling sensation consumers demand without compromising on safety or quality.
We invite procurement leaders and technical directors to collaborate with us to explore how this efficient synthesis route can enhance your product portfolio. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
