Scalable Synthesis of High-Performance Phosphorus-Nitrogen Flame Retardant Intermediates for Epoxy Systems
Scalable Synthesis of High-Performance Phosphorus-Nitrogen Flame Retardant Intermediates for Epoxy Systems
The global shift towards halogen-free materials has intensified the demand for advanced organophosphorus flame retardants that do not compromise the mechanical or thermal properties of the host polymer. Patent CN102617645A introduces a significant breakthrough in this domain by disclosing a series of 1-oxo-4-hydroxymethyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane derivatives designed specifically for epoxy resin applications. These novel compounds leverage a unique caged phosphate structure combined with nitrogen-containing functional groups to achieve a synergistic flame retardant effect. Unlike traditional halogenated additives that release toxic corrosive gases upon combustion, this technology promotes char formation through a phosphorus-nitrogen synergy mechanism, effectively insulating the underlying material from heat and oxygen. For R&D directors and procurement specialists in the polymer industry, this represents a viable pathway to meet stringent environmental regulations like RoHS and REACH while maintaining high performance standards in electronic encapsulation and composite materials.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formulation of flame-retardant epoxy resins has relied heavily on brominated compounds or simple phosphate esters, both of which present significant drawbacks in modern high-performance applications. Brominated flame retardants, while effective, are increasingly restricted due to their persistence in the environment and the generation of dioxins during incineration, posing severe supply chain risks for manufacturers targeting green markets. On the other hand, conventional phosphate esters often suffer from poor thermal stability and migration issues, where the additive leaches out of the polymer matrix over time, leading to a loss of flame retardancy and contamination of the surface. Furthermore, many existing organophosphorus additives require high loading levels to achieve desired UL-94 ratings, which frequently degrades the mechanical strength and glass transition temperature (Tg) of the cured epoxy, limiting their utility in structural applications where integrity is paramount.
The Novel Approach
The technology described in the patent overcomes these limitations by utilizing a rigid bicyclic phosphate cage, specifically the 1-oxo-4-hydroxymethyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane scaffold, as the core building block. This caged structure provides exceptional thermal stability, with decomposition temperatures exceeding 200°C, ensuring that the additive remains intact during the typical curing cycles of epoxy resins. By functionalizing the hydroxymethyl group with various nitrogen-containing moieties, such as diphenylamino or azobenzene groups, the inventors have created a library of derivatives that exhibit a powerful P-N synergistic effect. This synergy allows for effective flame retardancy at lower loading levels, typically around 10% by weight, thereby preserving the mechanical properties of the epoxy matrix. The result is a material that not only self-extinguishes rapidly but also forms a robust char layer that acts as a physical barrier against heat transfer, addressing both safety and performance requirements simultaneously.
Mechanistic Insights into Phosphorus-Nitrogen Synergistic Flame Retardancy
The efficacy of these derivatives stems from a dual-action mechanism involving both condensed phase and gas phase interactions during thermal decomposition. In the condensed phase, the phosphorus component of the caged structure promotes the dehydration and carbonization of the polymer matrix, leading to the formation of a stable, intumescent char layer. This char acts as an insulating shield, preventing the underlying material from reaching its ignition temperature and blocking the diffusion of oxygen to the pyrolysis zone. Simultaneously, the nitrogen functionalities incorporated into the R-group of the molecule decompose to release non-flammable gases such as ammonia (NH3) and nitrogen oxides (NO). These gases dilute the concentration of combustible volatiles and oxygen in the immediate vicinity of the flame, effectively suppressing the combustion reaction. The patent data highlights that compound I-3, containing an azobenzene linkage, achieves a remarkable char residue rate of 78% at 400°C, demonstrating the superior capability of this molecular design to promote solid-phase protection over volatile fuel generation.
From a synthetic chemistry perspective, the preparation of these compounds relies on a nucleophilic substitution reaction that is highly controllable and scalable. The key intermediate, 1-oxo-4-chloroacyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane (designated as Ma in the patent), serves as an electrophilic acylating agent. This reactive species is coupled with various nucleophiles (Mb), which can be primary or secondary amines, phenols, or alcohols, depending on the desired final functionality. The reaction is typically conducted in polar aprotic solvents like dichloroethane or acetonitrile, using organic bases such as triethylamine or diisopropylethylamine to scavenge the hydrochloric acid byproduct. This mild reaction condition avoids the harsh temperatures or strong acids that might degrade the sensitive caged phosphate structure, ensuring high purity and yield. The versatility of this approach allows for the rapid generation of a diverse library of flame retardants tailored to specific polymer compatibility requirements without the need for complex multi-step syntheses.
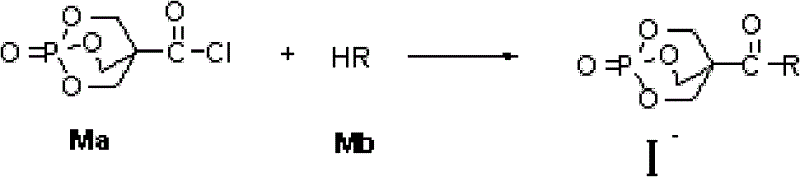
How to Synthesize 1-oxo-4-hydroxymethyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane Derivatives Efficiently
The synthesis protocol outlined in the patent offers a robust and reproducible method for producing these high-value flame retardant intermediates on a commercial scale. The process begins with the dissolution of the chloroacyl precursor in a suitable organic solvent, followed by the controlled addition of the amine or alcohol intermediate and a stoichiometric amount of organic base. Temperature control is critical during the addition phase, typically maintained between -10°C and 50°C to manage the exotherm and prevent side reactions. Once the addition is complete, the reaction mixture is allowed to stir at elevated temperatures, ranging from ambient up to 150°C depending on the reactivity of the nucleophile, for a period of 1 to 20 hours. The workup procedure is notably simple, involving filtration of the precipitated product followed by thorough washing with water to remove salt byproducts, eliminating the need for complex chromatographic purification steps which are often cost-prohibitive in bulk manufacturing. Detailed standardized synthesis steps follow below.
- Dissolve 1-oxo-4-chloroacyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane (Ma) in a polar organic solvent such as dichloroethane or acetonitrile under stirring.
- Prepare a mixture of the intermediate Mb (amine or alcohol) and an organic base like triethylamine or diisopropylethylamine, then add dropwise to the Ma solution at low temperature (-10 to 50°C).
- Allow the reaction to proceed at temperatures between -10°C and 150°C for 1 to 20 hours, followed by filtration, washing with water, and drying to obtain the target flame retardant derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers distinct strategic advantages centered around regulatory compliance and process efficiency. The primary benefit lies in the transition to a halogen-free formulation, which mitigates the risk of future regulatory bans on brominated flame retardants and aligns with the sustainability goals of major electronics and automotive OEMs. By securing a supply of these phosphorus-nitrogen synergistic additives, manufacturers can future-proof their product lines against evolving environmental legislation, ensuring uninterrupted market access in regions with strict chemical controls like the European Union. Furthermore, the high thermal stability of these derivatives means they can be processed alongside high-performance engineering thermosets without degradation, reducing waste and rework costs associated with thermal instability during compounding.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive transition metal catalysts or complex purification technologies, relying instead on straightforward nucleophilic substitution and filtration. This simplification of the downstream processing significantly lowers the operational expenditure per kilogram of produced additive. Additionally, the high efficiency of the flame retardant allows for lower loading levels in the final epoxy formulation compared to conventional fillers, which reduces the total raw material cost per unit of finished resin while maintaining or even enhancing mechanical performance.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including the caged phosphate precursor and common amines or phenols, are derived from widely available industrial feedstocks. This reduces dependency on niche or geographically concentrated raw material sources that are prone to volatility. The robustness of the reaction conditions, which tolerate a range of solvents and bases, further enhances supply chain resilience by allowing for flexibility in sourcing auxiliary chemicals, ensuring consistent production continuity even during market fluctuations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproduct is an organic amine salt that can be easily separated and potentially recycled or treated. The absence of halogenated solvents or reagents simplifies waste management and reduces the environmental footprint of the manufacturing facility. This aligns with corporate social responsibility mandates and facilitates easier permitting for capacity expansion, enabling suppliers to scale production from pilot batches to multi-ton annual volumes without significant infrastructure upgrades for pollution control.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these flame retardant derivatives in industrial epoxy systems. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for engineering and procurement decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this new chemistry into existing production lines and product portfolios.
Q: What is the thermal stability range of these phosphorus-nitrogen flame retardant derivatives?
A: According to patent data, these compounds exhibit excellent thermal stability, typically beginning decomposition above 200°C. Specific derivatives like compound I-3 show exceptional char formation rates of up to 78% at 400°C, making them suitable for high-temperature polymer processing.
Q: How does the phosphorus-nitrogen synergy improve flame retardancy in epoxy resins?
A: The P-N bond enhances the flame retardant effect on cellulose and polymer matrices. Upon heating, the system generates small amounts of NO and NH3 gases, which capture free radicals essential for combustion, thereby terminating the chain reaction and promoting char formation rather than flammable gas release.
Q: Are these intermediates compatible with standard epoxy curing agents?
A: Yes, the patent demonstrates successful integration with polyamide resin curing agents (such as resin 650). The derivatives can be added at loadings of 1% to 30% by mass without compromising the curing process, achieving UL-94 ratings and improved Limiting Oxygen Index (LOI) values.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-oxo-4-hydroxymethyl-1-phospha-2,6,7-trioxabicyclo[2,2,2]octane Derivative Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this patented laboratory methodology into a reliable commercial supply chain. We understand that scaling complex organophosphorus chemistry requires precise control over reaction parameters and rigorous quality assurance to ensure batch-to-batch consistency. Our facilities are equipped to handle the specific solvent systems and temperature profiles required for this synthesis, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to verify that every shipment meets the thermal stability and composition standards necessary for high-performance epoxy applications, guaranteeing that your final products perform exactly as designed.
We invite you to collaborate with us to optimize this flame retardant solution for your specific resin systems. Our technical team can provide a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this halogen-free alternative in your current formulations. Please contact our technical procurement team to request specific COA data for our pilot batches and comprehensive route feasibility assessments tailored to your volume requirements, ensuring a smooth and efficient transition to next-generation flame retardant technology.
