Advanced Palladium-Catalyzed Synthesis of 4-(1',3'-Conjugated Dien-2'-yl)-2,5-Dihydrofuran Intermediates
Introduction to High-Stereoselective Dihydrofuran Synthesis
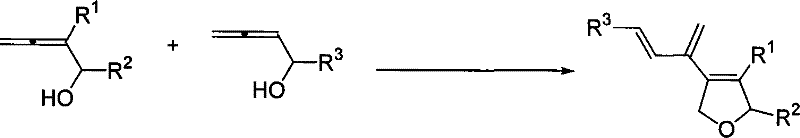
The development of efficient synthetic routes for complex heterocyclic structures remains a cornerstone of modern pharmaceutical research, particularly when targeting bioactive natural product analogs. Patent CN101250170B introduces a groundbreaking methodology for the high-stereoselective synthesis of 4-(1',3'-conjugated dien-2'-yl)-2,5-dihydrofuran derivatives, a structural motif frequently encountered in potent therapeutic agents. This innovation addresses critical bottlenecks in organic synthesis by leveraging a palladium iodide-catalyzed intermolecular cross-coupling reaction that operates under remarkably mild yet effective conditions. By utilizing a specific combination of 2-substituted and 2-unsubstituted 2,3-allenols in the presence of boron trifluoride etherate, the process achieves a level of stereocontrol and substituent diversity that was previously difficult to attain with conventional techniques. For R&D directors and procurement specialists alike, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with streamlined processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2,5-dihydrofuran rings containing 1,3-conjugated diene structures has been plagued by significant inefficiencies that hindered their widespread application in drug discovery pipelines. Prior art, including studies cited in the background of the patent such as those by Ma and Hashmi, often reported reaction yields that were unacceptably low for commercial viability, frequently struggling to exceed modest conversion rates even under optimized conditions. Furthermore, these traditional methodologies were severely restricted in terms of structural flexibility, typically allowing only for the introduction of单一 (single) types of substituents, which limited the chemical space available for medicinal chemists to explore structure-activity relationships. The inability to efficiently introduce diverse functional groups at multiple positions on the dihydrofuran core meant that synthesizing complex libraries required lengthy, multi-step sequences that drove up costs and extended lead times substantially.
The Novel Approach
In stark contrast to these historical challenges, the novel approach detailed in CN101250170B utilizes a sophisticated palladium iodide catalytic system that fundamentally transforms the efficiency of this transformation. By employing a precise intermolecular cross-coupling between a 2-substituted 2,3-allenol and a 2-unsubstituted 2,3-allenol, the reaction achieves high stereoselectivity while simultaneously introducing multiple distinct substituents in a single operational step. The use of dimethyl sulfoxide as the solvent medium, combined with the Lewis acid activation provided by boron trifluoride etherate, creates an environment where the reaction proceeds smoothly at 80°C, delivering yields ranging from 38% to 81% across various substrates. This robustness allows for the synthesis of a wide array of derivatives where R1 can be alkyl, phenyl, allyl, or ester groups, and R2 and R3 can vary between alkyl and aryl moieties, providing the structural diversity essential for modern drug development programs.
Mechanistic Insights into PdI2-Catalyzed Cyclization
The mechanistic pathway of this transformation relies on the unique ability of palladium iodide to activate the allene functionality towards nucleophilic attack while maintaining strict control over the stereochemical outcome. The catalytic cycle likely initiates with the coordination of the palladium species to the electron-rich pi-system of the 2,3-allenol, facilitating the formation of a key organopalladium intermediate that is stabilized by the adjacent hydroxyl group. The addition of one equivalent of boron trifluoride etherate plays a crucial role as a Lewis acid promoter, potentially activating the hydroxyl group for elimination or enhancing the electrophilicity of the intermediate, thereby driving the cyclization forward to form the five-membered dihydrofuran ring. This synergistic interaction between the transition metal catalyst and the Lewis acid additive ensures that the reaction proceeds with high fidelity, minimizing the formation of regioisomers or polymeric byproducts that often complicate allene chemistry.
From an impurity control perspective, the specific stoichiometry employed in this process is instrumental in ensuring product purity and simplifying downstream processing. The patent specifies a molar ratio of the 2-unsubstituted 2,3-allenol to the 2-substituted 2,3-allenol of approximately 1.1:1 to 1.3:1, which helps to drive the consumption of the more valuable substituted starting material to completion. Additionally, the use of palladium iodide at a loading of 0.05 equivalents relative to the substrate strikes a balance between catalytic activity and cost, avoiding the excessive metal loadings that can lead to difficult-to-remove heavy metal residues in the final API intermediate. The reaction is quenched simply by adding water at room temperature, followed by ether extraction, which effectively separates the organic product from the polar DMSO solvent and inorganic salts, resulting in a crude material that is amenable to rapid purification via flash column chromatography.
How to Synthesize 4-(1',3'-Conjugated Dien-2'-yl)-2,5-Dihydrofuran Efficiently
Executing this synthesis requires careful attention to the order of addition and temperature control to maximize the benefits of the patented protocol. The process begins at room temperature by preparing a mixture of the 2-unsubstituted 2,3-allenol and the palladium iodide catalyst, followed by the sequential addition of half the volume of the dimethyl sulfoxide solvent, the boron trifluoride etherate additive, the 2-substituted 2,3-allenol, and finally the remaining solvent. This specific addition sequence is designed to ensure proper mixing and activation of the catalytic species before the introduction of the second coupling partner, preventing premature side reactions. Once the reagents are combined, the mixture is heated to 80°C and stirred for a duration of 0.5 to 1.5 hours, after which the reaction is monitored for completion before proceeding to the workup phase involving aqueous quenching and organic extraction.
- Combine 2-substituted 2,3-allenol, palladium iodide catalyst, and dimethyl sulfoxide solvent with boron trifluoride etherate and 2-unsubstituted 2,3-allenol at room temperature.
- Heat the reaction mixture to 80°C and stir for 0.5 to 1.5 hours to facilitate the cross-coupling cyclization.
- Quench the reaction with water, extract with ether, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound advantages in terms of cost structure and operational reliability compared to legacy methods. The elimination of complex, multi-step sequences traditionally required to build the dihydrofuran core translates directly into reduced manufacturing costs, as fewer unit operations mean lower labor, energy, and equipment utilization expenses. Furthermore, the reliance on readily available starting materials such as simple 2,3-allenols and common reagents like palladium iodide and DMSO mitigates the risk of supply chain disruptions associated with exotic or proprietary building blocks, ensuring a steady flow of materials for continuous production campaigns. The simplicity of the workup procedure, which avoids intricate distillation or crystallization steps in favor of standard extraction and chromatography, further enhances the economic viability of the process by reducing solvent consumption and waste generation.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot cross-coupling reaction significantly lowers the overall cost of goods sold by consolidating multiple bond-forming events into a single vessel operation. By avoiding the need for expensive chiral ligands or harsh reaction conditions that require specialized containment, the process achieves substantial cost savings through simplified infrastructure requirements and reduced utility consumption. The high stereoselectivity inherent to the method also minimizes the loss of material to unwanted isomers, thereby improving the overall mass balance and yield efficiency which is critical for margin improvement in competitive pharmaceutical intermediate markets.
- Enhanced Supply Chain Reliability: The use of commodity chemicals and stable catalysts ensures that the raw material supply base is robust and less susceptible to geopolitical or logistical volatility. Since the reaction tolerates a wide range of substituents including alkyl, phenyl, and ester groups, manufacturers can flexibly source different grades of allenols without compromising the integrity of the final product, providing a buffer against specific raw material shortages. This flexibility allows for dynamic inventory management and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and clinical trial material requirements.
- Scalability and Environmental Compliance: The operational parameters of 80°C in DMSO are well within the safe operating limits of standard glass-lined or stainless steel reactors, facilitating seamless scale-up from laboratory grams to commercial tonnage without the need for re-engineering the process. The straightforward aqueous workup and the ability to recover solvents contribute to a reduced environmental footprint, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. This compliance readiness reduces the regulatory burden on the supply chain and ensures long-term sustainability of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on reaction performance and applicability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this chemistry into their existing production workflows or R&D pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method offers significantly higher stereoselectivity and allows for diverse substituent introduction compared to previous literature which suffered from low yields and limited structural variety.
Q: What are the optimal reaction conditions for this cross-coupling?
A: The reaction proceeds optimally at 80°C in dimethyl sulfoxide (DMSO) using palladium iodide as the catalyst and one equivalent of boron trifluoride etherate as an additive.
Q: Is this process suitable for large-scale pharmaceutical intermediate production?
A: Yes, the operation is simple, utilizes readily available reagents, and the products are easy to separate and purify, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(1',3'-Conjugated Dien-2'-yl)-2,5-Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for complex heterocyclic intermediates like 4-(1',3'-conjugated dien-2'-yl)-2,5-dihydrofuran in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality and stringent purity specifications. Our rigorous QC labs are equipped to verify the stereochemical integrity and impurity profiles of these sensitive diene-containing structures, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to contact our technical procurement team to discuss how this advanced synthetic technology can be leveraged for your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this efficient route. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and accelerate your path to market.
