Optimizing Sulfasalazine Purity: A Novel DMF-Based Purification Strategy for Global API Supply Chains
Optimizing Sulfasalazine Purity: A Novel DMF-Based Purification Strategy for Global API Supply Chains
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their critical intermediates to ensure patient safety and regulatory compliance. A significant breakthrough in this domain is documented in patent CN1261413C, which details a sophisticated purification process for 5-[pair-(2-pyridine sulfamic) benzene] azo salicylic acid, commonly known as Sulfasalazine (SASP). This compound is a vital therapeutic agent primarily used for treating nonspecific ulcerative colitis and rheumatoid arthritis, functioning by releasing sulfapyridine and 5-aminosalicylic acid within the intestinal wall. As market requirements for SASP specifications become increasingly rigorous, particularly for export to regulated markets, the ability to consistently produce high-purity material is a decisive competitive advantage. The technology described in this patent offers a robust solution to the longstanding challenges of impurity removal, utilizing a dimethylformamide (DMF) mixed solvent system that drastically enhances product quality while streamlining the manufacturing workflow.
For R&D Directors and Technical Leads, the significance of this patent lies in its ability to address the complex impurity profile inherent to SASP synthesis. Traditional methods often struggle to eliminate structurally similar byproducts, leading to batches that fail to meet the strict limits set by major pharmacopoeias. By adopting the methodology outlined in CN1261413C, manufacturers can achieve a finished product that not only complies with the Chinese Pharmacopoeia 2000 edition but also satisfies the demanding criteria of BP2000 and Ph.Eur.4. This level of compliance is essential for any reliable pharmaceutical intermediate supplier aiming to secure long-term contracts with multinational pharmaceutical corporations. The process represents a shift from brute-force purification to a more nuanced, solubility-driven approach that maximizes yield without compromising on the stringent purity profiles required for modern anti-inflammatory therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Sulfasalazine has relied heavily on conventional techniques involving reflux with glacial acetic acid followed by alkaline dissolution and acid precipitation. While this method was sufficient for older quality standards, it presents severe drawbacks in a modern manufacturing environment. The primary issue is the aggressive nature of glacial acetic acid reflux, which causes serious corrosion to production equipment, leading to increased maintenance costs and potential downtime. Furthermore, this traditional approach is notoriously inefficient at removing specific, structurally similar impurities that co-precipitate with the target molecule. The process generates a significant volume of acidic and alkaline waste, creating a heavy burden on environmental treatment facilities and complicating the "three wastes" management strategy. Consequently, the final product quality often fluctuates, making it difficult to consistently reach the regulation of the European Pharmacopoeia, thereby limiting the commercial viability of the batch for high-value markets.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a DMF mixing solution to dissolve coarse products, followed by a controlled acid separation process. This method fundamentally changes the solubility dynamics of the system, allowing for the selective precipitation of pure Sulfasalazine while keeping impurities in the solution phase. The operation is simple and convenient, requiring standard industrial equipment without the need for exotic materials to resist extreme corrosion. By optimizing the solvent ratio and pH control, this technique achieves an obvious impurity removing effect, specifically targeting the nine known structural impurities that plague the synthesis. The result is a finished product with greatly improved quality and a refining yield that is significantly higher than traditional methods. For procurement teams, this translates to a more stable supply of high-grade material, reducing the risk of batch rejection and ensuring continuity in the downstream formulation of anti-inflammatory medications.
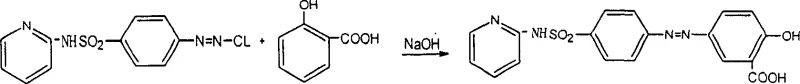
Mechanistic Insights into DMF-Mediated Solubility Control
The core mechanism driving the success of this purification process is the precise manipulation of solubility through a mixed solvent system and pH gradient. Sulfasalazine contains both acidic (carboxylic acid, phenolic hydroxyl) and basic (azo, pyridine) functional groups, making its solubility highly sensitive to pH and solvent polarity. In the patented process, the crude product is first dissolved in a 15-70% DMF aqueous solution with the addition of alkali. DMF acts as a powerful polar aprotic solvent that effectively breaks down the crystal lattice of the crude solid, ensuring that even occluded impurities are released into the solution. This complete dissolution is critical; unlike partial washing methods where impurities remain trapped within the solid matrix, this step ensures a homogeneous starting point for separation. The subsequent addition of acid to maintain a pH of 1-3 at elevated temperatures (60-95°C) triggers the selective crystallization of the zwitterionic form of Sulfasalazine, while the more soluble or differently charged impurities remain in the mother liquor.
Furthermore, the process effectively targets specific problematic impurities identified in the synthesis pathway, such as 4,4'-[(4-hydroxyl-1,3-phenylene) two (diazenediyls)] two [N-(pyridine-2-yl) benzsulfamide] and various hydroxyl-phenylformic acid derivatives. These impurities often have pKa values or solubility profiles slightly different from the target molecule. By controlling the acidification rate and temperature, the process exploits these subtle differences. For instance, impurities like "Whitfield's ointment" (a colloidal term used in the patent context for specific gum-like byproducts) and other azo-coupling side products are either kept in solution or filtered out during the hot filtration steps. The use of activated carbon for decolorization in the second stage further polishes the product by adsorbing colored organic byproducts. This multi-stage purification logic ensures that the final crystal lattice is formed from a solution depleted of contaminants, resulting in a product with superior chromatographic purity as evidenced by HPLC analysis.
How to Synthesize Sulfasalazine Efficiently
The implementation of this purification protocol requires careful attention to solvent ratios and pH endpoints to maximize recovery. The process begins with the dissolution of the crude SASP in a specific concentration of DMF and water, followed by a controlled acidification to precipitate a semi-finished product. This intermediate is then subjected to a second purification cycle involving alkaline dissolution and re-precipitation to ensure ultimate purity. The detailed operational parameters, including exact temperatures and stirring rates, are critical for reproducibility on a commercial scale. For a comprehensive understanding of the standardized synthetic steps and critical process parameters (CPPs) required to replicate this high-yield purification, please refer to the technical guide below.
- Dissolve the Sulfasalazine crude product in a 15-70% DMF aqueous solution (2-6 times volume) with heating and stirring, then slowly add alkali until fully dissolved.
- Add acid to the solution to maintain a pH of 1-3 at a temperature of 60-95°C, then centrifuge to obtain the semi-finished product.
- Redissolve the semi-finished product in aqueous alkali, perform heat decolorization with activated carbon, filter, adjust filtrate to pH 1-3 with acid, and dry the final precipitate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this DMF-based purification technology offers substantial strategic benefits for supply chain managers and procurement officers. The primary advantage lies in the drastic simplification of the purification workflow, which eliminates the need for harsh reflux conditions and reduces the complexity of waste handling. By moving away from the corrosive glacial acetic acid reflux method, manufacturers can significantly extend the lifespan of their reactor vessels and piping systems, leading to lower capital expenditure on equipment replacement and maintenance. Additionally, the improved yield reported in the patent data suggests a more efficient utilization of raw materials, meaning that less crude starting material is required to produce the same amount of finished API intermediate. This efficiency directly contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing material loss and reducing the volume of solvent that needs to be recovered or disposed of.
- Cost Reduction in Manufacturing: The elimination of severe equipment corrosion and the reduction in solvent consumption create a leaner manufacturing process. By avoiding the energy-intensive and time-consuming steps associated with traditional acid reflux and multiple recrystallizations from neat solvents, the overall production cost per kilogram is optimized. The ability to recover and reuse the DMF mother liquor through vacuum distillation further enhances the economic viability of the process, turning a potential waste stream into a valuable resource. This closed-loop solvent management not only saves money but also aligns with green chemistry principles, reducing the environmental footprint of the facility.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a consistent supply of high-quality Sulfasalazine. Traditional methods often suffer from batch-to-batch variability due to the difficulty in controlling impurity levels, which can lead to production delays and missed delivery deadlines. The new process, with its clear endpoints and effective impurity removal, stabilizes the production schedule. This reliability is crucial for maintaining the continuity of supply for downstream drug manufacturers who rely on just-in-time inventory models. Furthermore, the simplified operation reduces labor intensity and the potential for human error, adding another layer of security to the supply chain.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges, but this DMF-based method is designed for industrial applicability. The use of common industrial acids and alkalis, combined with standard filtration and drying equipment, makes the transition from pilot to commercial scale straightforward. Moreover, the significant reduction in "three wastes" (waste water, waste gas, waste residue) simplifies regulatory compliance. In an era of increasing environmental scrutiny, having a purification process that inherently generates less hazardous waste provides a distinct competitive advantage and mitigates the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Sulfasalazine. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production lines.
Q: What are the primary impurities removed by this novel DMF purification process?
A: The process effectively removes nine known structural impurities, including difficult-to-separate compounds such as 4,4'-[(4-hydroxyl-1,3-phenylene) two (diazenediyls)] two [N-(pyridine-2-yl) benzsulfamide] and various hydroxyl-phenylformic acid derivatives that typically persist in conventional acetic acid reflux methods.
Q: How does this method compare to traditional glacial acetic acid reflux in terms of equipment safety?
A: Unlike the conventional method which requires extensive reflux with glacial acetic acid causing severe equipment corrosion and difficult three-waste treatment, the DMF mixed solvent method operates under milder conditions with significantly reduced corrosivity, extending equipment lifespan and simplifying waste management.
Q: Does this purification technique meet international pharmacopoeia standards?
A: Yes, the purified Sulfasalazine produced via this method fully meets the stringent quality requirements of the Chinese Pharmacopoeia (2000 edition), British Pharmacopoeia (BP2000), and European Pharmacopoeia (Ph.Eur.4), ensuring global market compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfasalazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of intermediates like Sulfasalazine is the foundation of safe and effective final drug products. Our technical team has extensively analyzed advanced purification pathways, including the DMF-based methodology described in CN1261413C, to ensure we offer the highest quality standards to our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition innovative lab-scale processes into robust, GMP-compliant manufacturing operations. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities and enforcing stringent purity specifications that meet or exceed international pharmacopoeia requirements, ensuring that every batch we deliver supports your regulatory filings and market success.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable, cost-effective, and high-purity supply of Sulfasalazine that drives your business forward.
