Advanced Organocatalytic Synthesis of Optically Active Oxazolin-2-One Derivatives for Commercial Scale
Introduction to Next-Generation Oxazolin-2-One Synthesis
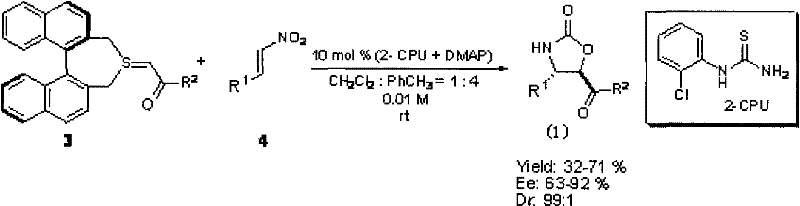
The landscape of heterocyclic chemistry has been significantly transformed by the innovations detailed in patent CN101585817B, which introduces a highly efficient method for synthesizing optically active oxazolin-2-one derivatives. These compounds are not merely academic curiosities; they represent a critical class of scaffolds with profound implications for both pharmaceutical and agrochemical industries, echoing the success of landmark molecules like Linezolid. The patent discloses a novel organocatalytic cascade reaction involving C2 symmetrical chiral sulfur ylides and nitroalkenes, achieving exceptional stereocontrol without the need for harsh conditions or precious metal catalysts. This technological breakthrough addresses the longstanding challenge of constructing complex chiral centers in a single operational step, thereby streamlining the supply chain for high-value intermediates. For R&D directors and procurement specialists alike, understanding this methodology is crucial for securing a reliable supply of advanced agrochemical intermediates that meet stringent purity specifications while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazolin-2-one core has relied heavily on the utilization of chiral amino acids and their derivatives as starting materials. While effective, these traditional pathways often necessitate multi-step synthetic sequences involving protection and deprotection strategies, which inherently accumulate waste and reduce overall atom economy. Furthermore, many classical methods require stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts that pose significant challenges for downstream purification and regulatory compliance regarding heavy metal residues. The reliance on cryogenic temperatures in some asymmetric protocols further exacerbates energy consumption and operational complexity, creating bottlenecks in commercial scale-up. These factors collectively contribute to higher manufacturing costs and extended lead times, making it difficult for suppliers to respond agilely to market demands for cost reduction in agrochemical intermediate manufacturing without compromising on optical purity.
The Novel Approach
In stark contrast, the methodology presented in the patent leverages a sophisticated organocatalytic system comprising o-chlorothiourea (2-CPU) and DMAP to drive a cascade reaction between stable sulfur ylides and nitroalkenes. This approach operates under remarkably mild conditions, typically at room temperature (10-30°C), eliminating the energy-intensive requirements of cryogenic cooling. The reaction proceeds with high diastereoselectivity, often achieving dr values of 99:1, and impressive enantioselectivity with ee values reaching up to 92% in optimized examples. By bypassing the need for chiral pool starting materials, this route offers a more direct and convergent synthesis strategy. The use of organocatalysts not only reduces the cost of goods sold by avoiding precious metals but also simplifies the workup procedure, as the catalysts can often be removed or degraded more easily than metal complexes. This represents a paradigm shift towards greener, more sustainable chemistry that aligns perfectly with modern supply chain goals for reducing lead time for high-purity agrochemical intermediates.
Mechanistic Insights into 2-CPU and DMAP Catalyzed Cascade Reaction
The mechanistic elegance of this transformation lies in the synergistic activation provided by the dual catalyst system. The o-chlorothiourea (2-CPU) acts as a hydrogen-bond donor, activating the nitroalkene electrophile through specific non-covalent interactions that organize the transition state geometry. Simultaneously, DMAP likely facilitates the generation or stabilization of the reactive sulfur ylide species, ensuring a steady concentration of the nucleophile. This cooperative catalysis creates a highly ordered chiral environment around the reacting centers, which is critical for discriminating between the pro-chiral faces of the substrates. The result is a concerted cyclization that forms the oxazolin-2-one ring with precise control over the newly generated stereocenters. Such mechanistic precision is vital for R&D teams aiming to replicate these results, as it ensures that the impurity profile remains manageable and predictable. The high stereoselectivity observed minimizes the formation of unwanted diastereomers, which are often difficult to separate and can compromise the biological efficacy of the final agrochemical product.
From an impurity control perspective, the robustness of this organocatalytic cycle is particularly noteworthy. The reaction tolerates a wide range of substituents on both the nitroalkene and the sulfur ylide components, including electron-withdrawing and electron-donating groups on the aromatic rings. This substrate scope flexibility allows for the generation of diverse libraries of derivatives without needing to re-optimize reaction conditions for each new analog. The absence of metal catalysts means there is no risk of metal-catalyzed side reactions such as homocoupling or oxidation, which are common pitfalls in transition metal chemistry. Consequently, the crude reaction mixtures are cleaner, facilitating simpler purification via standard column chromatography. For quality assurance teams, this translates to a more consistent product quality and reduced analytical burden, ensuring that every batch of high-purity oxazolin-2-one derivatives meets the rigorous standards required for biological testing and subsequent commercial application.
How to Synthesize Optically Active Oxazolin-2-One Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific protocol outlined in the patent to maximize yield and stereoselectivity. The general procedure involves dissolving the nitroalkene substrate along with the catalytic amounts of 2-CPU and DMAP in a mixed solvent system of toluene and dichloromethane. This specific solvent ratio is critical for maintaining the solubility of all components while optimizing the hydrogen-bonding network essential for catalysis. Once the catalyst mixture is equilibrated, the chiral sulfur ylide is introduced, initiating the cascade sequence. The reaction progress is monitored via TLC until completion, after which standard aqueous workup and chromatographic purification yield the target compound. The detailed standardized synthesis steps see the guide below for precise molar ratios and handling instructions.
- Prepare the reaction mixture by stirring nitroalkene with 2-CPU and DMAP catalysts in a toluene and dichloromethane solvent system at room temperature.
- Add the C2 symmetrical chiral sulfur ylide to the reaction flask and continue stirring until TLC analysis confirms complete consumption of starting materials.
- Purify the crude reaction mixture using column chromatography with a petroleum ether, ethyl acetate, and dichloromethane gradient to isolate the target oxazolin-2-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of expensive transition metal catalysts directly correlates to a significant reduction in raw material costs, as organocatalysts like thiourea derivatives are generally far cheaper and more abundant than palladium or rhodium complexes. Moreover, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to lower utility costs and a smaller carbon footprint. The simplified purification process, driven by the cleaner reaction profile, reduces the consumption of silica gel and solvents during chromatography, further driving down the cost of goods. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in the price of precious metals or energy markets, ensuring stable pricing for customers seeking cost reduction in agrochemical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the replacement of costly chiral auxiliaries and metal catalysts with inexpensive organocatalysts. By utilizing a cascade reaction that builds complexity in a single step, the number of unit operations is drastically reduced, leading to lower labor and equipment overheads. The high atom economy of the reaction ensures that a greater proportion of the starting materials ends up in the final product, minimizing waste disposal costs. Additionally, the ability to run the reaction at room temperature removes the need for specialized cryogenic equipment, allowing for the use of standard glass-lined reactors which are more readily available and cheaper to maintain. This holistic reduction in operational expenditure makes the commercial production of these derivatives financially viable even at large scales.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available starting materials such as nitroalkenes and stable sulfur ylides, which can be sourced from multiple global suppliers. Unlike specialized chiral amino acids that may have limited suppliers and long lead times, the precursors for this reaction are commodity chemicals with robust supply networks. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in temperature or moisture, reducing the risk of batch failures and production delays. This reliability is crucial for maintaining consistent inventory levels and meeting delivery commitments to downstream formulators. Furthermore, the absence of regulatory hurdles associated with heavy metal residues simplifies the registration process for new agrochemical products, accelerating time-to-market.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is straightforward due to the exothermic nature of the reaction being manageable at room temperature. The use of common organic solvents like toluene and dichloromethane allows for easy recovery and recycling, aligning with green chemistry principles and environmental regulations. The organocatalytic nature of the synthesis ensures that the final product is free from toxic metal contaminants, a critical requirement for agrochemicals that are applied to crops and enter the ecosystem. This environmental compatibility reduces the burden on wastewater treatment facilities and minimizes the generation of hazardous waste. Consequently, manufacturers can achieve commercial scale-up of complex agrochemical intermediates with greater ease and confidence, knowing that the process adheres to strict environmental, health, and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these oxazolin-2-one derivatives. The answers are derived directly from the experimental data and background information provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines or R&D programs. We encourage stakeholders to review these insights to fully appreciate the versatility and robustness of this synthetic methodology.
Q: What are the key advantages of this organocatalytic method over traditional amino acid routes?
A: This method eliminates the need for multi-step conversions from chiral amino acids, utilizing a direct cascade reaction that offers high diastereoselectivity (up to 99:1 dr) and excellent enantioselectivity under mild room temperature conditions.
Q: What is the biological activity profile of these oxazolin-2-one derivatives?
A: The synthesized derivatives demonstrate significant fungicidal activity against pathogens such as Fusarium oxysporium and Rhizoctonia solani, as well as herbicidal effects on dicotyledonous and monocotyledonous plants.
Q: Is this synthesis scalable for industrial production?
A: Yes, the process uses readily available starting materials, operates at room temperature without cryogenic conditions, and avoids expensive transition metal catalysts, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolin-2-One Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic synthesis methods described in patent CN101585817B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped to handle organocatalytic reactions with precision, maintaining stringent purity specifications through our rigorous QC labs. We understand that the consistency of stereochemistry is paramount for the biological activity of agrochemicals, and our processes are designed to deliver the high dr and ee values demonstrated in the patent literature consistently across large batches.
We invite you to collaborate with us to leverage this advanced chemistry for your next-generation agrochemical or pharmaceutical projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data for our catalog of oxazolin-2-one derivatives or to discuss route feasibility assessments for your custom synthesis needs. Together, we can optimize your supply chain and bring innovative solutions to the market faster and more efficiently.
