Advanced Anhydrous Synthesis of Azelaic Acid Derivatives for High-Purity Pharmaceutical Intermediates
Advanced Anhydrous Synthesis of Azelaic Acid Derivatives for High-Purity Pharmaceutical Intermediates
The development of effective dermatological agents often hinges on the ability to modify active pharmaceutical ingredients to enhance their bioavailability and formulation stability. Patent CN102388017B introduces a significant technological advancement in the synthesis of azelaic acid amide ester derivatives, specifically addressing the limitations of traditional aqueous-based methods. This innovation is particularly relevant for manufacturers seeking a reliable pharmaceutical intermediates supplier capable of delivering high-purity compounds for cosmetic and therapeutic applications. The core breakthrough lies in the establishment of strictly anhydrous reaction conditions using aprotic solvents and organic bases, which fundamentally alters the reaction kinetics and product isolation profile compared to legacy processes.
Traditional azelaic acid derivatives, such as potassium azelayl diglycinate, have demonstrated superior efficacy in acne treatment at significantly lower concentrations than the parent acid, thereby reducing skin irritation. However, scaling the production of these complex molecules has historically been fraught with challenges related to moisture sensitivity and by-product formation. The methodology outlined in this patent provides a robust framework for overcoming these hurdles, offering a pathway to cost reduction in pharmaceutical intermediates manufacturing by improving overall process efficiency. By shifting the reaction environment from aqueous alkaline solutions to dry organic media, the process minimizes the degradation of valuable starting materials and streamlines the downstream purification steps required for commercial grade output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acylated amino acid derivatives has relied heavily on the Schotten-Baumann reaction, which typically proceeds in an aqueous alkaline medium. While this method is well-established for simple substrates, it presents severe drawbacks when applied to moisture-sensitive reagents like azelaic acid dichloride. In the presence of water, even under controlled pH conditions, the acid halide is prone to rapid hydrolysis, converting the valuable acylating agent back into azelaic acid, which acts as an impurity rather than a reactant. This side reaction not only consumes raw materials but also complicates the purification process, as separating the desired amide from the corresponding carboxylic acid can be energetically and chemically demanding.

Furthermore, the conventional aqueous approach often necessitates the use of strong inorganic bases to maintain pH levels, which can lead to racemization of chiral amino acid centers or unwanted side reactions with sensitive functional groups on the amino acid side chains. For instance, amino acids containing hydroxyl or sulfhydryl groups may require additional protection and de-protection steps to prevent polymerization or oxidation, adding significant time and cost to the manufacturing timeline. The resulting products are frequently highly water-soluble, making their isolation from the reaction mixture difficult and requiring extensive lyophilization or salt precipitation techniques that are not ideal for large-scale industrial operations.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by utilizing an anhydrous environment established through the use of aprotic solvents such as chloroform, dichloromethane, or tetrahydrofuran. In this system, the amino acid hydrohalide is first deprotonated using an organic base, typically pyridine, which serves a dual function as both a base and a scavenger for the hydrogen halide by-product generated during acylation. This modification effectively neutralizes the acidic by-product in situ, driving the equilibrium towards the formation of the desired amide bond without the risk of hydrolyzing the acid chloride. The reaction proceeds smoothly at ambient or slightly cooled temperatures, preserving the stereochemical integrity of chiral amino acids.
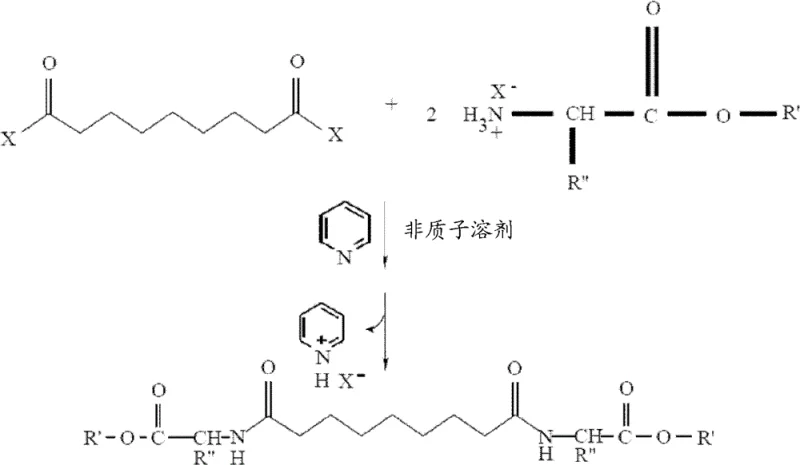
Moreover, this anhydrous protocol significantly simplifies the workup procedure. Since the reaction does not generate large volumes of aqueous waste containing inorganic salts, the product can be isolated through straightforward solvent evaporation and washing steps. The use of anhydrous salts like sodium sulfate ensures that any trace moisture introduced during reagent addition is sequestered, maintaining the integrity of the acid halide throughout the process. This results in a crude product with a much cleaner impurity profile, reducing the burden on final purification steps such as recrystallization and enabling higher overall yields across a broader range of amino acid substrates, including those with reactive side chains.
Mechanistic Insights into Anhydrous Acylation Catalysis
The mechanistic foundation of this synthesis relies on the precise control of nucleophilicity and electrophilicity within a non-aqueous medium. The process begins with the amino acid ester hydrohalide, which exists in a protonated ammonium state that renders the nitrogen atom non-nucleophilic. As illustrated in the structural representation below, the ammonium group must be deprotonated to generate a free amine capable of attacking the carbonyl carbon of the azelaic acid dichloride. The addition of an organic base like pyridine facilitates this deprotonation, creating a reactive nucleophile while simultaneously forming a pyridinium salt that remains soluble in the organic phase.
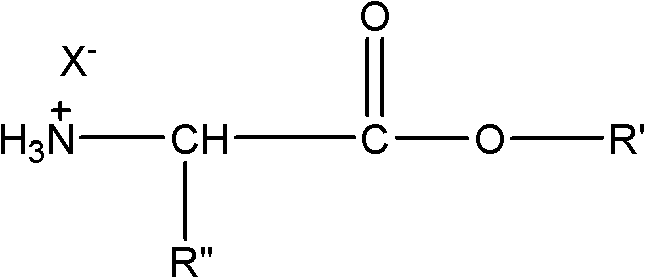
Once the free amine is generated, it undergoes a nucleophilic acyl substitution with the acid chloride. The carbonyl carbon of the azelaic acid dichloride is highly electrophilic due to the electron-withdrawing nature of the chlorine atoms. The attack by the amino group forms a tetrahedral intermediate, which subsequently collapses to expel the chloride ion. The expelled chloride ion then associates with the protonated pyridine, effectively removing it from the reaction equilibrium and preventing the reverse reaction or acid-catalyzed degradation of the product. This mechanism ensures that the acylation proceeds efficiently even with sterically hindered amino acids, provided that the solvent system maintains solubility for all species involved.
Impurity control is inherently built into this mechanism by excluding water. In aqueous systems, water competes with the amine as a nucleophile, leading to the hydrolysis of the acid chloride. By rigorously excluding moisture through the use of dried solvents and anhydrous drying agents post-reaction, the pathway for hydrolysis is blocked. Additionally, the choice of aprotic solvents prevents the solvation of the nucleophile to an extent that would reduce its reactivity, ensuring that the kinetic rate of amide formation outpaces any potential side reactions. This level of control is critical for producing high-purity intermediates where trace halide ions or carboxylic acid impurities could compromise the stability or safety of the final dermatological formulation.
How to Synthesize Azelaic Acid Amide Ester Efficiently
The practical execution of this synthesis requires careful attention to reagent stoichiometry and moisture control to maximize yield and purity. The patent outlines a generalized procedure where amino acid ester hydrohalides are reacted with azelaic acid dichloride in a molar ratio of approximately 2:1, ensuring that both acid chloride groups are fully consumed. The reaction is typically initiated by the dropwise addition of the acid chloride solution to the deprotonated amino acid mixture to manage the exotherm and minimize local concentration spikes that could lead to oligomerization. Detailed standardized synthesis steps for this process are provided in the guide below.
- Dissolve amino acid hydrohalide in an aprotic solvent and treat with an organic base like pyridine to effect deprotonation.
- React the deprotonated product with azelaic acid halide dissolved in an aprotic solvent under controlled temperature.
- Dry the reaction mixture using anhydrous salts such as sodium sulfate and remove the solvent via vacuum evaporation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this anhydrous synthesis method offers tangible strategic benefits beyond mere technical superiority. The elimination of aqueous workups drastically reduces the volume of wastewater generated, aligning with increasingly stringent environmental regulations and lowering the costs associated with waste treatment and disposal. Furthermore, the ability to use a wide variety of amino acid esters without complex protection strategies simplifies the raw material supply chain, allowing manufacturers to source standard, commercially available building blocks rather than custom-synthesized protected intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant improvement in raw material utilization efficiency. By preventing the hydrolysis of azelaic acid dichloride, a relatively expensive reagent, the process ensures that nearly all input material is converted into the desired product rather than wasted as carboxylic acid by-products. Additionally, the simplified isolation procedure, which avoids energy-intensive lyophilization or complex salt precipitation, reduces utility consumption and labor hours per batch. The use of recoverable organic solvents further enhances the economic viability, as solvent recycling loops can be easily integrated into the production line to minimize consumable expenses.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents or conditions that are difficult to scale often creates bottlenecks in the supply of critical intermediates. This method utilizes robust, scalable chemistry that relies on common organic solvents and bases, reducing the risk of supply disruptions caused by the unavailability of niche catalysts or reagents. The flexibility to accommodate various amino acid side chains means that production lines can be quickly adapted to manufacture different derivatives based on market demand without requiring extensive retooling or process re-validation, thereby enhancing the agility of the supply chain response to customer needs.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden inefficiencies, particularly in separation and purification stages. The anhydrous nature of this reaction facilitates straightforward scale-up, as the heat transfer and mixing dynamics in organic solvents are generally more predictable than in heterogeneous aqueous slurries. From an environmental perspective, the reduction in aqueous waste streams and the potential for solvent recovery contribute to a smaller environmental footprint. This alignment with green chemistry principles not only mitigates regulatory risk but also appeals to downstream customers who are increasingly prioritizing sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this anhydrous synthesis technology. These insights are derived directly from the experimental data and process descriptions found within the patent literature, providing a factual basis for evaluating the feasibility of adopting this method for large-scale production. Understanding these nuances is essential for technical teams assessing the integration of this chemistry into existing manufacturing portfolios.
Q: Why is the anhydrous method superior to conventional Schotten-Baumann conditions?
A: Conventional aqueous methods risk hydrolyzing the sensitive acid halide starting material into carboxylic acids, reducing yield. The anhydrous method eliminates water, preventing this side reaction and simplifying product isolation.
Q: What types of amino acids are compatible with this synthesis route?
A: This method is versatile and supports neutral alpha-amino acids like glycine and alanine, as well as those with functional side chains such as serine, cysteine, and methionine, without requiring complex protection groups.
Q: How does this process impact the purity of the final cosmetic ingredient?
A: By avoiding aqueous workups that can leave salt residues and minimizing hydrolysis by-products, the process yields derivatives with high purity profiles suitable for sensitive dermatological formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azelaic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced dermatological ingredients requires more than just a patented reaction; it demands a partner with the infrastructure to execute complex chemistry with precision and consistency. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azelaic acid derivative meets the exacting standards required for pharmaceutical and cosmetic applications.
We invite you to collaborate with our technical team to explore how this anhydrous synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals, ensuring a reliable supply of high-quality intermediates for your global operations.
