Scalable Solvent-Free Synthesis of Phenothiazinyl Chalcone Derivatives for Pharmaceutical Applications
Scalable Solvent-Free Synthesis of Phenothiazinyl Chalcone Derivatives for Pharmaceutical Applications
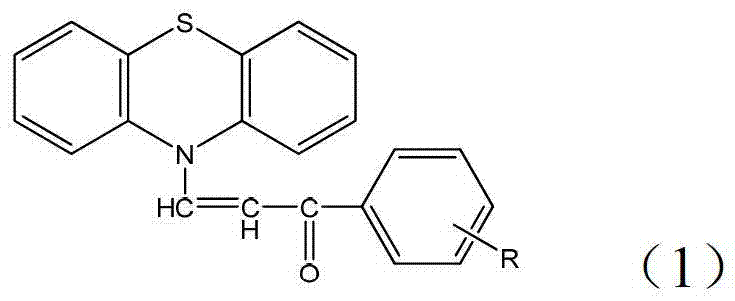
The pharmaceutical industry is constantly seeking more efficient, environmentally benign, and cost-effective pathways for synthesizing complex heterocyclic intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN103214429A, which details a novel preparation method for 1-substituted phenyl-3-(N-phenothiazinyl)prop-2-en-1-one derivatives. These compounds represent a unique fusion of the pharmacologically active phenothiazine scaffold with the versatile chalcone structure, creating molecules with potent antibacterial properties. The patent highlights a mechanochemical approach that bypasses traditional solvent-heavy processes, offering a streamlined route that appeals to modern green chemistry standards. By utilizing a simple grinding technique with inorganic bases, this method achieves high yields and purity without the need for expensive catalysts or prolonged heating, positioning it as a highly attractive option for large-scale manufacturing of biomedical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of chalcone derivatives and their heterocyclic analogues typically relies on solution-phase chemistry, often requiring volatile organic solvents such as ethanol, methanol, or dichloromethane. These conventional routes frequently necessitate extended reaction times under reflux conditions, which not only consumes significant energy but also increases the risk of thermal degradation of sensitive functional groups. Furthermore, the reliance on liquid media complicates the downstream processing; removing residual solvents often requires energy-intensive distillation or rotary evaporation steps, and the resulting waste streams demand costly treatment protocols to meet environmental regulations. The use of homogeneous catalysts in solution can also lead to difficulties in product isolation, often requiring column chromatography which is impractical for industrial scale-up due to high silica gel consumption and low throughput.
The Novel Approach
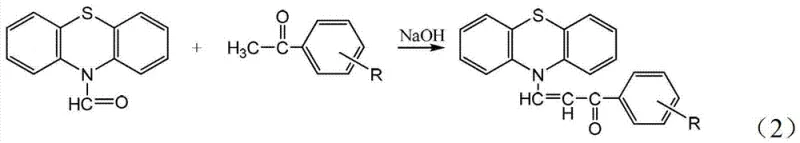
In stark contrast, the methodology described in CN103214429A employs a solvent-free mechanochemical strategy that fundamentally alters the reaction dynamics. By physically grinding the solid reactants—N-formylphenothiazine and various substituted acetophenones—together with a solid base catalyst like sodium hydroxide or potassium hydroxide, the reaction proceeds rapidly at ambient conditions. This approach eliminates the solvation shell effects that can sometimes hinder reactivity in solution, allowing for direct molecular contact that accelerates the condensation process. The absence of bulk solvent means that the reaction mixture is highly concentrated, driving the equilibrium towards product formation efficiently. Moreover, the workup is remarkably simplified; the crude solid can be directly washed with water to remove inorganic salts and unreacted base, followed by simple filtration and drying, thereby drastically reducing the operational complexity and time required to obtain the final high-purity crystalline product.
Mechanistic Insights into Base-Catalyzed Mechanochemical Condensation
The core chemical transformation driving this synthesis is a classic Claisen-Schmidt condensation, adapted here for a solid-state environment. The mechanism initiates when the strong inorganic base, such as sodium hydroxide, abstracts an acidic alpha-proton from the methyl group of the substituted acetophenone. In the absence of solvent, this deprotonation generates a highly reactive enolate species that is immediately available for nucleophilic attack. This enolate then targets the electrophilic carbonyl carbon of the N-formylphenothiazine, forming a beta-hydroxy ketone intermediate. Under the mechanical stress of grinding and the basic conditions, this intermediate undergoes rapid dehydration to yield the conjugated alpha,beta-unsaturated ketone system characteristic of chalcones. The rigid crystal lattice disruption caused by grinding ensures fresh surfaces are constantly exposed, maintaining high reaction kinetics throughout the process without the diffusion limitations often seen in viscous solution mixtures.
Impurity control in this mechanochemical system is managed through precise stoichiometric balancing and real-time monitoring. The patent specifies a molar ratio range where N-formylphenothiazine is slightly in excess or balanced (A:B = 5:(4-7)), ensuring that the more valuable or difficult-to-remove acetophenone derivative is fully consumed. Thin Layer Chromatography (TLC) is utilized as a critical process analytical technology (PAT) to monitor the disappearance of the starting material spots. By stopping the grinding process exactly when the limiting reagent is consumed, the formation of side products such as polymerization byproducts or over-condensation species is minimized. This precise endpoint determination, combined with the simplicity of washing away the inorganic base with water, results in a product profile with exceptional purity, often negating the need for further recrystallization steps that would otherwise reduce overall yield.
How to Synthesize 1-Substituted Phenyl-3-(N-Phenothiazinyl)prop-2-en-1-one Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates with minimal infrastructure requirements. The process is designed to be operationally simple, relying on standard laboratory or industrial grinding equipment rather than complex reactor vessels. The key to success lies in the thorough mixing of the solid components to ensure uniform contact between the organic substrates and the base catalyst. Detailed standardized synthetic steps for implementing this grinding methodology are provided in the guide below, ensuring reproducibility across different batch sizes.
- Mix N-formylphenothiazine and substituted acetophenone with sodium hydroxide or potassium hydroxide in a reaction vessel.
- Grind the mixture thoroughly while monitoring reaction progress via TLC until starting materials are consumed.
- Wash the crude product with water, filter the suspension, and vacuum dry the filter cake to obtain the pure ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free grinding technology represents a strategic opportunity to optimize both cost structures and operational reliability. The elimination of organic solvents removes a major variable from the supply chain, mitigating risks associated with solvent price volatility and availability shortages. Furthermore, the simplified downstream processing reduces the burden on utility systems, as there is no need for large-scale solvent recovery units or extensive wastewater treatment facilities dedicated to organic load removal. This translates directly into a leaner manufacturing footprint and reduced overhead costs, making the production of these antibacterial intermediates more economically sustainable in the long term.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete removal of organic solvents from the reaction matrix. In traditional chemical manufacturing, solvent purchase, storage, recovery, and disposal can account for a substantial portion of the total production cost. By shifting to a neat grinding method, these expenses are effectively eradicated. Additionally, the use of commodity inorganic bases like sodium hydroxide instead of expensive organic catalysts or transition metals further drives down the raw material bill. The high yields reported in the patent examples, ranging significantly above typical solution-phase averages for similar condensations, mean that less raw material is wasted, maximizing the output per unit of input and enhancing the overall cost-efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Relying on a process that uses readily available, stable solid reagents enhances supply chain resilience. Organic solvents often require specialized transport and storage conditions due to flammability and toxicity concerns, which can introduce logistical bottlenecks. In contrast, solid powders like N-formylphenothiazine and substituted acetophenones are easier to handle, store, and transport safely. The robustness of the grinding method also implies that production is less susceptible to fluctuations in utility quality, such as steam pressure for heating or cooling water capacity, since the reaction proceeds largely at ambient temperatures. This stability ensures consistent delivery schedules and reduces the risk of production stoppages due to equipment failure or utility constraints.
- Scalability and Environmental Compliance: Scaling up mechanochemical processes is increasingly feasible with modern industrial milling technology, offering a direct path from gram-scale optimization to tonnage production. The environmental benefits are profound; the E-factor (mass of waste per mass of product) is drastically improved because the primary waste stream is aqueous salt solution rather than toxic organic sludge. This aligns perfectly with tightening global environmental regulations and corporate sustainability goals. Facilities adopting this technology can expect smoother regulatory approvals and lower compliance costs related to emissions and waste disposal, securing their social license to operate and appealing to eco-conscious partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these phenothiazinyl chalcone derivatives. The answers are derived directly from the experimental data and technical specifications provided in the underlying patent documentation, ensuring accuracy and relevance for potential partners and technical stakeholders.
Q: What are the primary advantages of the grinding method over traditional solution synthesis?
A: The grinding method eliminates the need for organic solvents, significantly reducing environmental waste and solvent recovery costs. It also simplifies the workup process to a simple water wash and filtration, leading to higher overall efficiency and purity compared to conventional heating methods.
Q: What is the typical molar ratio of reactants used in this synthesis?
A: The optimal molar ratio of N-formylphenothiazine to substituted acetophenone to the base catalyst (NaOH or KOH) is generally maintained at 5:(4-7):(6-9), ensuring complete conversion while minimizing excess reagent waste.
Q: Does the final product exhibit specific biological activity?
A: Yes, the synthesized 1-substituted phenyl-3-(N-phenothiazinyl)prop-2-en-1-one derivatives demonstrate significant antibacterial activity against Gram-positive and Gram-negative bacteria, including Staphylococcus aureus and Escherichia coli, making them valuable for antimicrobial drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted Phenyl-3-(N-Phenothiazinyl)prop-2-en-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the synthetic route described in CN103214429A for generating high-value antibacterial intermediates. As a leading CDMO partner, we possess the technical expertise to translate this innovative grinding methodology into a robust, commercial-scale manufacturing process. Our facilities are equipped with advanced milling and solid-state reaction capabilities, allowing us to offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 1-substituted phenyl-3-(N-phenothiazinyl)prop-2-en-1-one meets the exacting standards required for pharmaceutical applications, guaranteeing consistency and safety for your downstream drug development programs.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your specific project needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this solvent-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our available derivatives and to discuss detailed route feasibility assessments for your target molecules, ensuring a seamless integration of these high-quality intermediates into your supply chain.
