Advanced Synthesis of 5-Substituted-2,3-Pyridinedicarboxylic Acids for Agrochemical Applications
Introduction to Novel Pyridine Intermediate Synthesis
The development of efficient synthetic routes for functionalized pyridine derivatives is critical for the agrochemical and pharmaceutical industries, particularly for the production of imidazolinone herbicides and related bioactive compounds. Patent CN1094398A discloses a sophisticated methodology for preparing 5,6-disubstituted-3-pyridylmethyl ammonium halides, which serve as pivotal intermediates in the synthesis of 5-(substituted methyl)-2,3-pyridinedicarboxylic acids. This technology addresses the longstanding challenge of introducing specific substituents at the 5-position of the pyridine ring with high regioselectivity and minimal byproduct formation. By utilizing a quaternary ammonium salt strategy, the process transforms a relatively inert methyl group into a highly reactive leaving group, enabling subsequent nucleophilic displacement under mild conditions.
The significance of this invention lies in its ability to produce high-purity intermediates that are essential for downstream coupling reactions. Traditional methods often suffer from over-oxidation or poor selectivity when attempting to functionalize the methyl group directly on the pyridine nucleus. In contrast, the disclosed method employs a controlled radical halogenation followed by quaternization, creating a versatile platform for introducing oxygen, sulfur, or nitrogen-containing substituents. As a reliable agrochemical intermediate supplier, understanding these mechanistic nuances is vital for ensuring the quality and consistency of the final active ingredients. The structural versatility of the intermediates, defined by Formula I in the patent, allows for the customization of side chains to meet specific biological activity requirements.
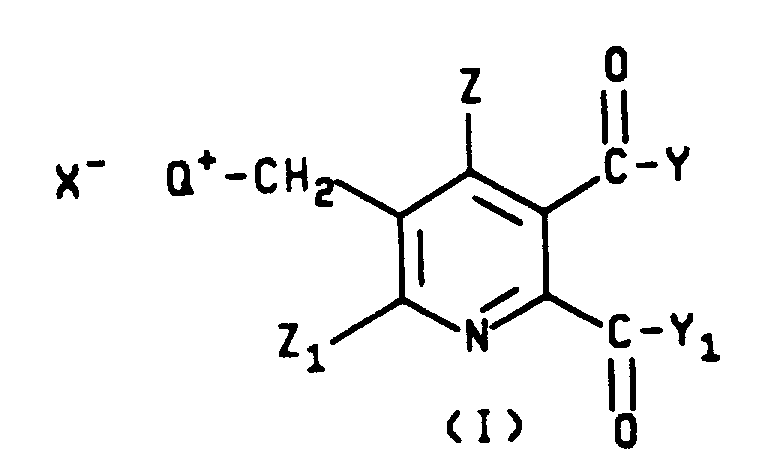
Furthermore, the patent outlines a comprehensive scope of substituents, including various esters, amides, and cyclic amines, demonstrating the broad applicability of this chemistry. The ability to tune the electronic and steric properties of the pyridine ring through these substitutions is paramount for optimizing the efficacy of the final herbicidal or pharmaceutical products. For R&D teams focused on cost reduction in agrochemical manufacturing, this route offers a streamlined pathway that avoids expensive transition metal catalysts or harsh oxidative conditions, thereby reducing both material costs and environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the 5-methyl group on 2,3-pyridinedicarboxylic acid derivatives has been fraught with challenges regarding selectivity and yield. Conventional approaches often rely on direct oxidation or free-radical halogenation without adequate control, leading to complex mixtures of mono-, di-, and tri-halogenated species. Separating these closely related byproducts is technically demanding and economically inefficient, often requiring multiple recrystallization steps or preparative chromatography, which drastically reduces overall throughput. Additionally, direct nucleophilic substitution on a simple halomethyl pyridine can be sluggish due to the electron-withdrawing nature of the pyridine nitrogen and the adjacent ester groups, necessitating high temperatures that may degrade sensitive functional groups.
Another significant drawback of older methodologies is the handling of free carboxylic acids during the functionalization steps. The presence of acidic protons can interfere with basic reagents or catalysts, leading to salt formation that complicates isolation and purification. Furthermore, traditional routes may lack the flexibility to introduce diverse substituents such as thioethers or bulky alkoxy groups without extensive protection-deprotection sequences. These inefficiencies translate into longer lead times and higher production costs, creating bottlenecks in the supply chain for high-value pharmaceutical intermediates and crop protection agents.
The Novel Approach
The innovative strategy presented in CN1094398A overcomes these hurdles by converting the 5-methyl group into a quaternary ammonium salt, which acts as a superior leaving group. This transformation activates the benzylic position for nucleophilic attack, allowing reactions to proceed rapidly at lower temperatures with excellent conversion rates. The use of ester protection for the carboxylic acid functions ensures compatibility with the basic conditions required for quaternization and subsequent substitution, preventing unwanted side reactions. This approach not only simplifies the purification process—since the ammonium salts often crystallize readily—but also enhances the overall atom economy of the synthesis.
Moreover, the modular nature of this synthesis allows for the easy variation of the amine component used in the quaternization step. By selecting different tertiary amines, such as trimethylamine, pyridine, or heterocyclic amines, chemists can fine-tune the reactivity of the intermediate to suit specific downstream transformations. This flexibility is crucial for the commercial scale-up of complex agrochemical intermediates, as it provides multiple levers to optimize yield and purity. The process effectively decouples the halogenation and substitution steps, allowing each to be optimized independently for maximum efficiency and minimal waste generation.
Mechanistic Insights into Radical Halogenation and Quaternization
The core of this synthetic route involves a two-stage activation of the 5-methyl position. Initially, a radical halogenation is performed using agents like N-bromosuccinimide (NBS) or 1,3-dibromo-5,5-dimethylhydantoin in the presence of a radical initiator such as 2,2'-azobis(2-methylbutyronitrile). This step selectively replaces one hydrogen atom of the methyl group with a halogen, generating a reactive halomethyl intermediate. The choice of initiator and solvent, typically chlorobenzene or tetrachloroethane, is critical to suppress poly-halogenation and ensure high selectivity for the mono-bromo species. Careful control of stoichiometry and temperature (typically 80-100°C) minimizes the formation of dibromo byproducts, which are difficult to separate and can compromise the purity of the final product.
Following halogenation, the crude mixture is treated with a tertiary amine to effect quaternization. This nucleophilic attack by the amine on the halomethyl carbon displaces the halide ion, forming the stable quaternary ammonium salt (Formula I). This salt is isolable and often possesses high crystallinity, serving as an effective purification point in the process. The positive charge on the nitrogen significantly enhances the electrophilicity of the adjacent methylene carbon, making it highly susceptible to displacement by weaker nucleophiles like alkoxides or thiolates in the subsequent step. This mechanistic sequence ensures that the final substitution occurs cleanly at the 5-position without affecting the ester groups or the pyridine ring itself.
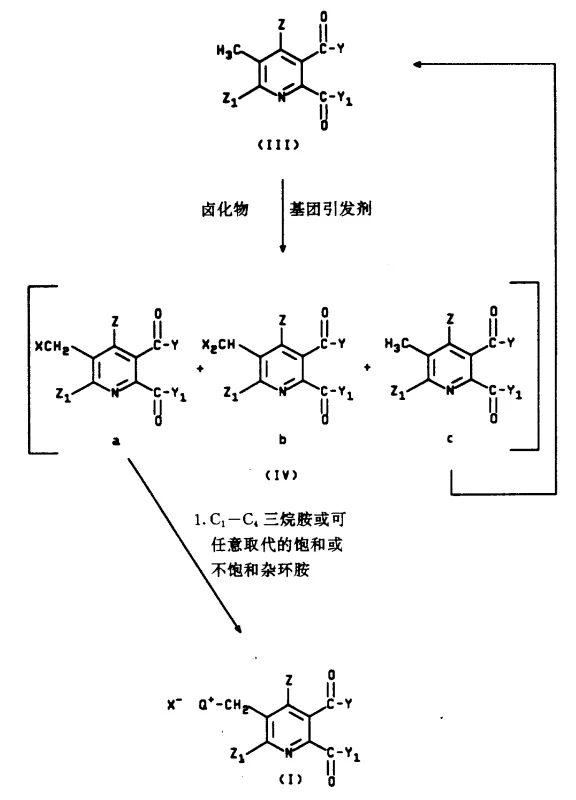
Impurity control is inherently built into this mechanism. By isolating the ammonium salt, any unreacted starting material or dibromo impurities can be washed away or left in the mother liquor. The subsequent nucleophilic displacement is typically conducted with alkali metal alkoxides (e.g., sodium methoxide) in alcoholic solvents. The driving force for this reaction is the formation of a neutral tertiary amine leaving group, which is thermodynamically favorable. Finally, hydrolysis of the ester groups under basic conditions followed by acidification yields the target 5-(substituted methyl)-2,3-pyridinedicarboxylic acid with purity levels exceeding 99%, as confirmed by HPLC analysis in the patent examples.
How to Synthesize 5-(Substituted Methyl)-2,3-Pyridinedicarboxylic Acids Efficiently
The synthesis of these valuable intermediates requires precise control over reaction parameters to maximize yield and minimize impurities. The process begins with the radical bromination of 5-methyl-2,3-pyridinedicarboxylic acid dimethyl ester, followed by quaternization with a selected amine. The resulting ammonium salt is then subjected to nucleophilic substitution and hydrolysis. Detailed operational procedures, including specific molar ratios, temperature profiles, and workup protocols, are essential for reproducibility on a manufacturing scale. The following guide summarizes the critical stages identified in the patent literature for producing these compounds efficiently.
- Perform radical halogenation on 5-methyl-2,3-pyridinedicarboxylic acid esters using N-bromosuccinimide or similar agents with a radical initiator.
- React the resulting halomethyl intermediate with a tertiary amine (e.g., trimethylamine, pyridine) to form the quaternary ammonium salt.
- Displace the quaternary ammonium group with an alkoxide or thioether nucleophile, followed by ester hydrolysis to yield the final dicarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply reliability. The reagents employed, such as N-bromosuccinimide, trimethylamine, and common organic solvents, are commodity chemicals available from multiple global sources, reducing the risk of single-supplier dependency. The robustness of the chemistry allows for flexible manufacturing schedules, as the intermediate ammonium salts can be stored and transported if necessary, decoupling the halogenation and substitution steps geographically if needed. This modularity enhances supply chain resilience against logistical disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of straightforward crystallization for purification significantly lower the cost of goods sold (COGS). By avoiding complex chromatographic separations and reducing solvent consumption through efficient recycling protocols, manufacturers can achieve substantial cost savings. The high selectivity of the radical halogenation step minimizes raw material waste, further contributing to economic efficiency. Additionally, the ability to use technical grade solvents without compromising product quality adds another layer of cost optimization.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on stable, non-hazardous reagents that are easy to source and handle, ensuring consistent production uptime. The high purity of the isolated intermediates reduces the likelihood of batch failures in downstream processing, leading to more predictable delivery schedules. This reliability is crucial for maintaining just-in-time inventory levels for downstream API or agrochemical production. The process is also adaptable to continuous flow chemistry, which can further increase throughput and reduce footprint.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproducts are succinimide (from NBS) and tertiary amines, which can often be recovered or treated easily. The use of alcohol and water in the final hydrolysis and isolation steps aligns with green chemistry principles, reducing the environmental burden. Scalability is demonstrated by the patent examples which utilize multi-hundred gram scales, indicating readiness for tonnage production. Compliance with environmental regulations is simplified due to the absence of heavy metals and the use of standard waste treatment protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyridine intermediates. The answers are derived from the detailed experimental data and claims found in the patent documentation, providing clarity on the feasibility and advantages of this technology for potential partners and licensees.
Q: What is the primary advantage of using quaternary ammonium salts in this synthesis?
A: The quaternary ammonium group serves as an excellent leaving group, facilitating smooth nucleophilic substitution to introduce diverse substituents (methoxy, thioether, etc.) at the 5-position with high regioselectivity and purity.
Q: Can this process be scaled for industrial production of herbicide intermediates?
A: Yes, the patent describes robust conditions using common solvents like chlorobenzene and alcohols, and reagents like N-bromosuccinimide, which are suitable for large-scale batch or continuous processing.
Q: How is the purity of the final dicarboxylic acid controlled?
A: High purity is achieved through the crystallization of the intermediate ammonium salts and careful control of the halogenation step to minimize dibromo byproducts, followed by standard hydrolysis and filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(Substituted Methyl)-2,3-Pyridinedicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of next-generation crop protection and pharmaceutical solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of radical chemistry and quaternization is maintained at every scale. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-(substituted methyl)-2,3-pyridinedicarboxylic acid meets the exacting standards required for regulatory filings and commercial manufacturing.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By leveraging our expertise in process chemistry and scale-up, we can help you accelerate your time-to-market while maintaining the highest standards of quality and compliance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
