Advanced One-Pot Synthesis of Oxabispiperidine Besylates for Commercial Scale-Up
Advanced One-Pot Synthesis of Oxabispiperidine Besylates for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex heterocyclic intermediates, particularly those intended for cardiac arrhythmia treatments. Patent CN1514836A introduces a groundbreaking methodology for the preparation of N-oxoalkyl-N'-anilinoalkyloxabispidine benzenesulfonate salts, addressing significant bottlenecks in traditional synthetic routes. This innovation centers on a direct coupling reaction that bypasses the cumbersome anion exchange steps previously deemed necessary for obtaining high-purity besylate salts. By leveraging a specific interaction between an N-oxoalkyl oxabispiperidine core and a benzenesulfonic acid anilinoalkyl ester, the process achieves superior purity profiles while drastically simplifying the operational workflow. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the methods disclosed in International Patent Application WO 01/28992, typically relied on a multi-step sequence involving the initial formation of a different salt form, often a tosylate, followed by a subsequent anion exchange to achieve the desired besylate. This conventional approach necessitates additional unit operations, including neutralization and extensive solvent exchange procedures, which inherently increase the risk of introducing impurities from the reagents used in the exchange process. Furthermore, the requirement for very pure starting materials to mitigate these impurity risks drives up raw material costs and complicates supply chain logistics. The need to manage multiple solvent systems and perform intermediate isolations not only extends the overall production timeline but also reduces the overall mass efficiency of the process, creating significant waste streams that require careful environmental management.
The Novel Approach
In stark contrast, the method described in CN1514836A enables the direct formation of the target benzenesulfonate salt in a simplified, essentially one-pot fashion. The core innovation involves reacting a compound of formula II directly with a compound of formula III, where the leaving group and counter-ion are pre-aligned to yield the besylate salt immediately upon completion of the nucleophilic substitution. 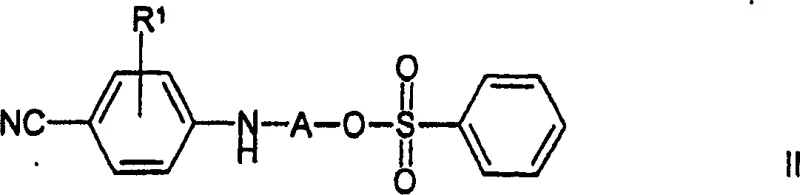 This eliminates the need for any post-reaction anion exchange, thereby removing the associated purification burdens and solvent swap requirements. The process operates effectively in hydroxylic solvents like ethanol, allowing for straightforward precipitation of the product by the addition of water, which significantly enhances the ease of isolation and reduces the reliance on hazardous chlorinated solvents often used in extraction-heavy workflows.
This eliminates the need for any post-reaction anion exchange, thereby removing the associated purification burdens and solvent swap requirements. The process operates effectively in hydroxylic solvents like ethanol, allowing for straightforward precipitation of the product by the addition of water, which significantly enhances the ease of isolation and reduces the reliance on hazardous chlorinated solvents often used in extraction-heavy workflows.
Mechanistic Insights into Direct Besylate Salt Formation
The chemical mechanism underpinning this synthesis relies on a nucleophilic attack by the secondary amine of the oxabispiperidine ring onto the alkyl chain of the benzenesulfonic acid ester. The reaction is facilitated by the specific electronic properties of the benzenesulfonate leaving group, which ensures high reactivity under moderate thermal conditions. Preferably conducted in ethanol at temperatures ranging from 20°C to 80°C, with a specific embodiment at 74°C, the reaction kinetics are optimized to favor the formation of the desired salt without promoting degradation of the sensitive oxabispiperidine core. The stoichiometric ratio is tightly controlled, ideally at 1:1, ensuring that neither reactant is in significant excess, which minimizes the formation of side products and simplifies the downstream purification strategy. This precise control over reaction parameters is critical for maintaining the structural integrity of the bicyclic system while achieving high conversion rates.
Impurity control is inherently built into this design by avoiding the introduction of extraneous ions during an exchange phase. In traditional methods, the exchange resin or alternative salt reagents can introduce trace metals or organic contaminants that are difficult to remove. By forming the besylate salt directly, the impurity profile is largely limited to unreacted starting materials, which are effectively removed through a simple recrystallization from ethanol and water.  The preparation of the key intermediate, Formula III, itself follows a robust pathway involving the reaction of 9-oxa-3,7-diazabicyclo[3.3.1]nonane with a ketone derivative, ensuring that the input quality remains high. This holistic approach to impurity management ensures that the final API intermediate meets stringent regulatory specifications required for cardiovascular drug manufacturing.
The preparation of the key intermediate, Formula III, itself follows a robust pathway involving the reaction of 9-oxa-3,7-diazabicyclo[3.3.1]nonane with a ketone derivative, ensuring that the input quality remains high. This holistic approach to impurity management ensures that the final API intermediate meets stringent regulatory specifications required for cardiovascular drug manufacturing.
How to Synthesize Oxabispiperidine Besylate Efficiently
Executing this synthesis requires careful attention to solvent quality and temperature profiling to maximize yield and purity. The process begins with the dissolution of the N-oxoalkyl oxabispiperidine and the anilinoalkyl benzenesulfonate in ethanol, followed by heating to reflux to drive the reaction to completion. After the reaction period, the mixture is cooled, and water is introduced to induce crystallization, a technique that leverages the differential solubility of the product in aqueous-organic mixtures. The detailed standardized synthesis steps, including specific molar ratios and cooling rates, are outlined in the guide below to ensure reproducibility across different manufacturing scales.
- React N-oxoalkyl oxabispiperidine with anilinoalkyl benzenesulfonate in ethanol at reflux temperature (approx. 74°C) for 6 hours.
- Cool the mixture to room temperature and stir for an extended period (e.g., 65 hours) to ensure complete conversion.
- Precipitate the product by adding water to the ethanolic solution, followed by cooling to 0°C and filtration to isolate the high-purity solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic advantages beyond mere technical elegance. The elimination of the anion exchange step translates directly into a reduction in processing time and resource consumption, as there is no longer a need to procure and manage specialized ion-exchange resins or additional salt reagents. This streamlining of the process flow reduces the overall footprint of the manufacturing operation, allowing for higher throughput within existing facility constraints. Furthermore, the use of ethanol as the primary solvent aligns with green chemistry initiatives, reducing the environmental burden associated with volatile organic compound emissions and hazardous waste disposal, which can lead to lower compliance costs and improved sustainability metrics for the organization.
- Cost Reduction in Manufacturing: The direct formation of the besylate salt removes the necessity for expensive purification steps associated with anion exchange, such as solvent swapping and neutralization. By consolidating the reaction and salt formation into a single operational phase, manufacturers can significantly reduce utility costs related to heating, cooling, and distillation. The ability to use technical grade ethanol and simple water for precipitation further lowers raw material expenses compared to processes requiring high-purity specialty solvents. Additionally, the higher recovery rates observed during recrystallization minimize material loss, ensuring that the cost per kilogram of the final intermediate is optimized for commercial viability.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the number of critical raw materials required, thereby mitigating supply chain risks associated with sourcing multiple specialized reagents. The reliance on commodity chemicals like ethanol and water, alongside readily available precursors, ensures a more resilient supply chain that is less susceptible to market fluctuations or vendor disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and stoichiometric variations, also means that production schedules are less likely to be impacted by minor process deviations, ensuring consistent delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous reaction conditions and simple precipitation techniques that translate well from laboratory to pilot and commercial scales. The avoidance of chlorinated solvents in the final isolation steps simplifies waste treatment protocols and reduces the regulatory burden associated with handling hazardous materials. This environmental compatibility not only facilitates easier permitting for new production lines but also enhances the corporate social responsibility profile of the manufacturing entity, making it a more attractive partner for global pharmaceutical companies with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering a clear understanding of the operational realities and potential benefits for stakeholders considering this route for their supply chain.
Q: What is the primary advantage of this synthesis method over prior art?
A: The primary advantage is the elimination of the anion exchange step. Unlike previous methods requiring conversion from tosylate to besylate, this process directly forms the benzenesulfonate salt, simplifying purification and reducing impurity risks.
Q: What solvents are preferred for the reaction and recrystallization?
A: The reaction is preferably carried out in ethanol. For purification, a mixture of ethanol and water is used for recrystallization, which effectively yields products with purity exceeding 99%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes common solvents like ethanol and operates at moderate temperatures (around 74°C), making it highly scalable. The direct precipitation method avoids complex solvent swaps typical in lab-scale anion exchanges.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxabispiperidine Besylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement complex chemistries, such as the direct besylate formation described in CN1514836A, positions us as a strategic partner capable of solving your most challenging supply chain hurdles.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits of switching to this streamlined synthesis. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and commercial reliability.
