Advanced Hydrazine Reduction Technology for Commercial Scale Production of 3-Aminophthalic Acid
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety and cost efficiency. A pivotal advancement in this domain is detailed in patent CN101012178A, which discloses a highly efficient method for preparing 3-aminophthalic acid from 3-nitrophthalic acid. This technology represents a significant departure from conventional hydrogenation techniques, utilizing a hydrazine hydrate reduction system catalyzed by iron trichloride and carbon. For R&D directors and procurement specialists alike, this patent offers a compelling solution to the longstanding challenges of producing this critical intermediate, which serves as a foundational building block for various active pharmaceutical ingredients and dye precursors. By shifting away from high-pressure hydrogenation or polluting iron powder methods, this approach delivers a yield of up to 95 percent with a purity exceeding 96 percent, establishing a new benchmark for reliability in the supply chain of complex aromatic amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminophthalic acid has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional methods often rely on catalytic hydrogenation using palladium on carbon (Pd/C), which, while effective, imposes severe safety risks due to the handling of high-pressure hydrogen gas and requires substantial capital investment in specialized pressure vessels. Furthermore, the Pd/C method often struggles to achieve purity levels beyond 90 percent, necessitating costly recrystallization steps that erode overall process margins. Alternatively, the older iron powder reduction technique, though avoiding high pressure, generates massive quantities of iron oxide sludge, creating a heavy environmental burden and complicating waste disposal protocols. This method also suffers from inherently low conversion rates, typically yielding only 50 to 60 percent of the desired product, which drastically inflates the cost of goods sold and limits the ability to meet high-volume demand consistently.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data utilizes a hydrazine hydrate reduction strategy that elegantly circumvents these traditional bottlenecks. By converting the starting material, 3-nitrophthalic acid, into its soluble sodium salt prior to reduction, the process ensures homogeneous reaction conditions that facilitate superior mass transfer and reaction kinetics. The introduction of an iron trichloride and activated carbon catalyst system allows the reduction to proceed efficiently at atmospheric pressure and moderate temperatures near 95 degrees Celsius. This eliminates the need for dangerous high-pressure infrastructure while simultaneously achieving a dramatic improvement in both yield and purity. The result is a streamlined workflow that not only enhances safety profiles for plant operators but also delivers a product quality that meets the stringent specifications required for downstream pharmaceutical applications without excessive purification burdens.
Mechanistic Insights into FeCl3-C Catalyzed Hydrazine Reduction
The core of this technological breakthrough lies in the synergistic interaction between hydrazine hydrate and the heterogeneous FeCl3/C catalyst system. In this mechanism, the activated carbon serves as a high-surface-area support that disperses the iron species, preventing agglomeration and maximizing the available active sites for electron transfer. When hydrazine hydrate is introduced to the heated reaction mixture, the iron catalyst facilitates the decomposition of hydrazine into reactive nitrogen species and protons, which are then selectively transferred to the nitro group of the substrate. This catalytic cycle effectively reduces the nitro functionality to an amino group while leaving the carboxylic acid moieties intact, a selectivity that is crucial for maintaining the structural integrity of the phthalic acid backbone. The presence of the sodium salt form during the reaction further stabilizes the intermediate species, preventing premature precipitation and ensuring that the reduction proceeds to completion with minimal formation of azo or hydrazo byproducts.
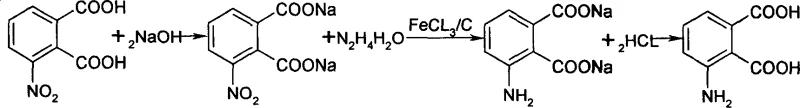
From an impurity control perspective, this mechanism offers distinct advantages over stoichiometric metal reductions. Because the catalyst loading is optimized between 0.2 to 1 times the weight of the substrate, and the reaction temperature is tightly controlled near reflux, the formation of over-reduced side products or polymeric tars is significantly suppressed. The subsequent acidification step with concentrated hydrochloric acid to a pH of 3 to 4 is critical, as it precisely targets the isoelectric point of the 3-aminophthalic acid, promoting selective crystallization while keeping soluble impurities in the mother liquor. This inherent selectivity of the crystallization process, driven by the clean reaction profile, is what allows the final product to consistently achieve purity levels greater than 96 percent as verified by HPLC analysis, reducing the need for energy-intensive chromatographic separations.
How to Synthesize 3-Aminophthalic Acid Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize the benefits of the catalytic system. The process begins with the formation of the sodium salt, followed by the addition of the catalyst and the controlled addition of the reducing agent. Maintaining the reaction temperature near 95 degrees Celsius is essential to drive the kinetics without causing excessive decomposition of the hydrazine.
- Dissolve 3-nitrophthalic acid in aqueous sodium hydroxide solution to form the sodium salt, maintaining a molar ratio between 1: 1.5 and 1:3.5.
- Add the FeCl3/C catalyst system to the solution, utilizing 0.2 to 1 times the weight of the starting acid relative to the catalyst consumption.
- Heat the mixture to near reflux at approximately 95 degrees Celsius and add hydrazine hydrate solution dropwise, maintaining reflux for 3 to 5 hours.
- Filter the hot reaction mixture, acidify the filtrate with concentrated hydrochloric acid to pH 3-4, and isolate the product via cooling crystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hydrazine-based reduction methodology offers profound strategic advantages that extend far beyond simple yield improvements. The elimination of high-pressure hydrogenation equipment removes a major capital expenditure barrier and significantly lowers the operational risk profile of the manufacturing facility, leading to reduced insurance premiums and maintenance costs. Furthermore, the avoidance of iron powder sludge generation simplifies waste management logistics, allowing for faster batch turnover and reduced downtime associated with reactor cleaning and waste disposal. These operational efficiencies translate directly into a more resilient supply chain capable of responding rapidly to market fluctuations without the bottlenecks typical of older, dirtier technologies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive noble metal catalysts and high-pressure infrastructure with inexpensive iron salts and standard atmospheric reactors. By removing the dependency on palladium, manufacturers avoid the volatility of precious metal markets and the complex recovery processes associated with spent noble metal catalysts. Additionally, the high yield of up to 96 percent means that less raw material is wasted per kilogram of finished product, effectively lowering the unit cost of production. The simplified workup procedure, which relies on straightforward filtration and acidification rather than complex extraction or chromatography, further reduces labor and utility costs, resulting in substantial overall cost savings for the production of this key intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of legacy manufacturing processes that are prone to equipment failure or regulatory shutdowns due to environmental non-compliance. This novel method utilizes readily available commodity chemicals such as hydrazine hydrate and sodium hydroxide, ensuring that raw material sourcing remains stable and unaffected by niche supply shortages. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without catastrophic failure, ensures consistent batch-to-batch quality. This reliability allows supply chain planners to forecast inventory levels with greater confidence and reduces the need for excessive safety stock, thereby optimizing working capital utilization across the organization.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions continue to tighten globally, the ability to scale production without increasing the environmental footprint is a critical competitive advantage. This process generates significantly less solid waste compared to iron powder reduction and avoids the safety hazards of high-pressure hydrogen, making it easier to obtain and maintain environmental permits. The scalability is further enhanced by the fact that the reaction can be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating rapid technology transfer from pilot scale to commercial tonnage production. This ease of scale-up ensures that suppliers can meet surging demand for pharmaceutical intermediates without requiring years of new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: What are the advantages of this hydrazine reduction method over traditional iron powder reduction?
A: Unlike traditional iron powder reduction which generates significant solid waste sludge and achieves lower yields of only 50-60 percent, this novel hydrazine hydrate method offers a cleaner process with yields reaching up to 96 percent and significantly reduced environmental pollution.
Q: How does the FeCl3/C catalyst system improve product purity?
A: The combination of iron trichloride and activated carbon facilitates a highly selective reduction of the nitro group to the amino group while minimizing side reactions, resulting in a final product purity exceeding 96 percent without the need for complex downstream purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process operates at atmospheric pressure and moderate temperatures around 95 degrees Celsius, eliminating the need for expensive high-pressure hydrogenation equipment required by Pd/C methods, thereby making it highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminophthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates like 3-aminophthalic acid requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the hydrazine reduction method are fully realized in a GMP-compliant environment. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the >96 percent purity threshold required by top-tier pharmaceutical clients, providing you with a partner who understands the critical nature of your supply chain.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your current sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to our manufactured grade can reduce your total landed costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless transition to a more efficient and reliable supply source.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
