Advanced Synthesis of Piperazine Bridged Naphthalimide Aminothiazole Oximes for Antimicrobial Applications
The pharmaceutical industry is constantly seeking novel scaffolds to combat the rising tide of antimicrobial resistance, and Patent CN112480105B presents a significant breakthrough in this domain with the disclosure of piperazine bridged naphthalimide aminothiazole oxime compounds. This intellectual property details a sophisticated class of chemical entities designed by merging the electron-rich naphthalimide core with the potent pharmacophore of aminothiazole oxime, linked via a flexible piperazine bridge. The general structure, as depicted in the patent, allows for extensive structural diversification at the N-position of the naphthalimide ring, enabling fine-tuning of physicochemical properties and biological activity. 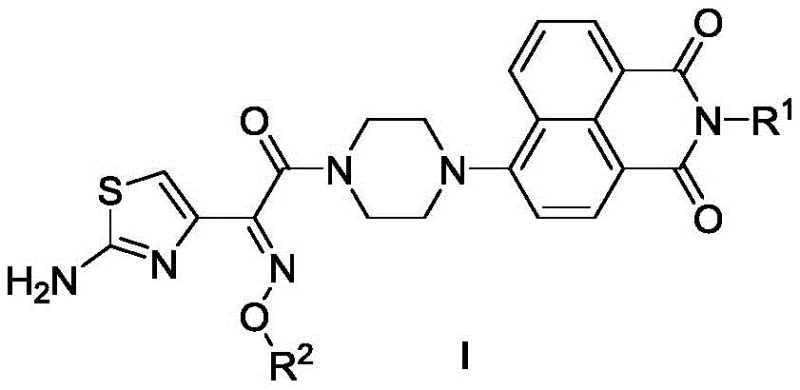 These compounds have demonstrated remarkable efficacy against a wide array of pathogens, including methicillin-resistant Staphylococcus aureus (MRSA) and various fungal strains, positioning them as critical candidates for next-generation anti-infective therapies. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these molecules is paramount for integrating them into future drug development pipelines.
These compounds have demonstrated remarkable efficacy against a wide array of pathogens, including methicillin-resistant Staphylococcus aureus (MRSA) and various fungal strains, positioning them as critical candidates for next-generation anti-infective therapies. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these molecules is paramount for integrating them into future drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex naphthalimide derivatives has been plagued by multi-step sequences that often require harsh reaction conditions and expensive catalysts. Conventional routes frequently involve the protection and deprotection of functional groups, leading to lower overall yields and increased waste generation. Furthermore, introducing specific substituents at the imide nitrogen or the aromatic core often necessitates specialized reagents that are not readily available on a commercial scale, creating bottlenecks in the supply chain. The purification of intermediates in these legacy processes can be particularly challenging due to the formation of closely related byproducts, which drives up the cost of goods sold (COGS) and extends lead times. For procurement managers, these inefficiencies translate into higher prices and less reliable supply continuity for essential pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN112480105B offers a streamlined, three-step synthetic strategy that dramatically simplifies the manufacturing process. The novel approach utilizes 4-bromo-1,8-naphthalic anhydride as a cost-effective starting material, which undergoes a direct amidation with various amino compounds to form Intermediate II. 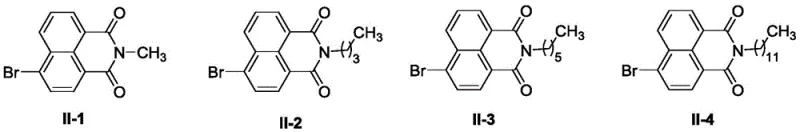 This step is followed by a nucleophilic substitution with anhydrous piperazine to install the crucial bridging moiety, yielding Intermediate III. The final coupling with the aminothiazole oxime fragment is achieved under exceptionally mild conditions, avoiding the thermal stress that often degrades sensitive functional groups. This concise route not only improves the overall atom economy but also significantly reduces the environmental footprint by minimizing solvent usage and waste disposal requirements, aligning perfectly with modern green chemistry principles.
This step is followed by a nucleophilic substitution with anhydrous piperazine to install the crucial bridging moiety, yielding Intermediate III. The final coupling with the aminothiazole oxime fragment is achieved under exceptionally mild conditions, avoiding the thermal stress that often degrades sensitive functional groups. This concise route not only improves the overall atom economy but also significantly reduces the environmental footprint by minimizing solvent usage and waste disposal requirements, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into the Three-Step Synthetic Route
The chemical elegance of this synthesis lies in its reliance on robust nucleophilic substitution and condensation mechanisms that are highly predictable and controllable. In the first step, the nucleophilic attack of the amine on the anhydride carbonyl is facilitated by the polar protic solvent ethanol at a moderate temperature of 80°C, ensuring high conversion rates without excessive energy input. The subsequent displacement of the bromine atom by piperazine in the second step is driven by the strong nucleophilicity of the secondary amine and the electron-withdrawing nature of the flanking carbonyl groups, which activates the aromatic ring for substitution. 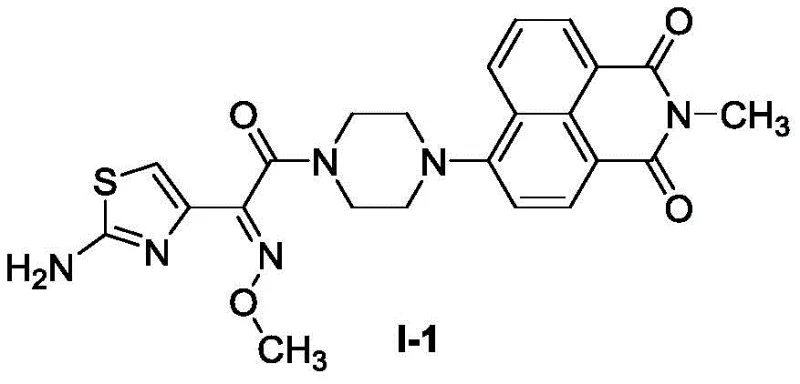 The final step involves the formation of an amide bond between the piperazine nitrogen and the activated ester of the aminothiazole oxime. This reaction proceeds efficiently at room temperature (25°C) in the presence of triethylamine, which acts as a base to scavenge the leaving group. From an impurity control perspective, the mildness of the final coupling step is particularly advantageous, as it prevents the isomerization of the oxime double bond and the degradation of the thiazole ring, thereby ensuring a high-purity profile for the final active pharmaceutical ingredient (API) intermediate.
The final step involves the formation of an amide bond between the piperazine nitrogen and the activated ester of the aminothiazole oxime. This reaction proceeds efficiently at room temperature (25°C) in the presence of triethylamine, which acts as a base to scavenge the leaving group. From an impurity control perspective, the mildness of the final coupling step is particularly advantageous, as it prevents the isomerization of the oxime double bond and the degradation of the thiazole ring, thereby ensuring a high-purity profile for the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize Piperazine Bridged Naphthalimide Aminothiazole Oximes Efficiently
The synthesis of these high-value intermediates is designed for operational simplicity, making it accessible for both laboratory-scale optimization and industrial-scale production. The process begins with the preparation of Intermediate II, where precise stoichiometric control (1:1.2 molar ratio) ensures complete consumption of the valuable anhydride. Following isolation, the intermediate is subjected to reflux with excess piperazine to drive the substitution to completion. The final coupling requires careful monitoring via thin-layer chromatography to prevent over-reaction, although the inherent stability of the reactants provides a wide processing window. For detailed operational parameters, safety guidelines, and workup procedures, please refer to the standardized synthesis protocol provided below.
- Preparation of Intermediate II: React 4-bromo-1,8-naphthalic anhydride with various amino compounds in ethanol at 80°C.
- Preparation of Intermediate III: React Intermediate II with anhydrous piperazine in ethylene glycol monomethyl ether at 120°C.
- Final Coupling: React Intermediate III with 2-(2-amino-4-thiazolyl)-2-(methoxyimino) acetic acid thiobenzothiazole ester using triethylamine in ethanol at 25°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, the adoption of this synthetic route offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. The primary benefit stems from the utilization of commodity chemicals as starting materials; 4-bromo-1,8-naphthalic anhydride and piperazine are produced in vast quantities globally, ensuring a stable and resilient supply base that is immune to the volatility often seen with exotic reagents. This reliance on bulk chemicals drastically reduces the risk of supply disruptions and allows for more accurate long-term forecasting of raw material costs. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which are a significant cost driver in many API manufacturing processes.
- Cost Reduction in Manufacturing: The streamlined three-step sequence inherently lowers the cost of production by reducing the number of unit operations, labor hours, and solvent volumes required. By avoiding cryogenic conditions or high-pressure reactors, the process can be executed in standard glass-lined or stainless-steel equipment found in most multipurpose chemical plants, eliminating the need for capital-intensive infrastructure upgrades. The high yields reported in the patent examples, often exceeding 80% for the initial steps, contribute to a superior mass balance, meaning less raw material is wasted per kilogram of final product. This efficiency translates into substantial cost savings that can be passed down the supply chain, making the final therapeutic more affordable.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding costly production delays. Since the reaction conditions are mild and the reagents are stable, the process is less susceptible to variations in ambient temperature or humidity, enhancing reproducibility across different manufacturing sites. This reliability allows supply chain heads to reduce safety stock levels and implement just-in-time inventory strategies, freeing up working capital. Additionally, the short synthesis timeline means that lead times from order to delivery can be significantly compressed compared to competitors using more convoluted routes.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples without loss of efficiency. The use of ethanol as a primary solvent is particularly beneficial from an environmental, health, and safety (EHS) perspective, as it is a green solvent with low toxicity and easy recyclability. This simplifies waste treatment protocols and reduces the burden on effluent treatment plants, helping manufacturers meet increasingly stringent environmental regulations. The ability to scale from 100 kgs to 100 MT annual production without fundamental changes to the chemistry makes this an ideal candidate for commercial expansion as demand for new antimicrobials grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these piperazine bridged naphthalimide derivatives. Understanding these aspects is crucial for stakeholders evaluating the feasibility of incorporating these intermediates into their product portfolios. The answers are derived directly from the technical specifications and experimental data provided in the source patent documentation.
Q: What is the antimicrobial spectrum of these piperazine bridged compounds?
A: The compounds exhibit broad-spectrum activity against Gram-positive bacteria including MRSA, Gram-negative bacteria such as E. coli and Pseudomonas aeruginosa, and fungi like Candida albicans.
Q: How does the synthesis route impact production costs?
A: The three-step route utilizes cheap and readily available raw materials like 4-bromo-1,8-naphthalic anhydride and operates under mild conditions, significantly reducing energy consumption and purification costs.
Q: Are these intermediates scalable for commercial API production?
A: Yes, the synthesis avoids expensive transition metal catalysts and uses standard solvents like ethanol, making it highly suitable for scale-up from kilogram to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Bridged Naphthalimide Aminothiazole Oxime Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality antimicrobial intermediates in the fight against drug-resistant pathogens. Our team of expert chemists has thoroughly analyzed the synthetic route described in Patent CN112480105B and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or preparing for market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of piperazine bridged naphthalimide aminothiazole oxime we deliver meets the highest international standards.
We invite you to collaborate with us to leverage this innovative chemistry for your next-generation anti-infective programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized manufacturing processes can reduce your overall COGS. Please contact us today to request specific COA data for our available analogues and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the development of life-saving medicines and address the global challenge of antimicrobial resistance.
