Advanced Synthesis of Quinoline 5-Carboxamides for Commercial PDE IV Inhibitor Production
Advanced Synthesis of Quinoline 5-Carboxamides for Commercial PDE IV Inhibitor Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical anti-inflammatory agents, particularly those targeting Phosphodiesterase IV (PDE IV) inhibition. Patent CN1878765A discloses a novel and highly efficient process for the preparation of specific substituted quinoline 5-carboxamides, which serve as potent PDE IV inhibitors. These compounds are pivotal in downregulating inflammatory cell activation and relaxing smooth muscle cells in the trachea and bronchi, offering significant therapeutic potential for asthma and other inflammatory conditions without the cardiovascular side effects associated with non-selective PDE inhibitors. The disclosed methodology represents a substantial advancement over previous techniques by eliminating the need for expensive transition metal catalysts and simplifying the overall reaction sequence. This technical insight report analyzes the chemical innovations within this patent and translates them into tangible commercial advantages for R&D directors, procurement managers, and supply chain heads seeking a reliable pharmaceutical intermediate supplier. By leveraging this optimized chemistry, manufacturers can achieve cost reduction in API manufacturing while ensuring the high-purity standards required for clinical and commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing quinoline carboxamides, such as those described in earlier literature, often rely on complex multi-step sequences that involve the use of necessary expensive starting materials and precious metal catalysts. A significant drawback of these conventional routes is the reliance on palladium catalysts, which are not only costly but also present substantial challenges in removal during the purification process. As is known to those skilled in the art, palladium catalyst removal is generally difficult and often requires specialized scavenging resins or extensive chromatography, which drives up production costs and extends processing time. Furthermore, traditional pathways may involve tedious steps that lower the overall throughput and introduce opportunities for impurity generation, thereby compromising the quality of the final active pharmaceutical ingredient. The economic burden of these inefficiencies is compounded by the need for stringent quality control measures to ensure residual metal levels comply with regulatory guidelines. Consequently, manufacturers utilizing these older methods face higher operational expenditures and longer lead times, which can hinder their ability to respond quickly to market demands for high-purity pharmaceutical intermediates.
The Novel Approach
In contrast, the novel approach disclosed in the patent provides a streamlined and economically viable pathway that circumvents the limitations of previous methodologies by utilizing more accessible and cost-effective reagents. This innovative process achieves the construction of the quinoline core through a condensation reaction between an aniline derivative and a trifluoro enone, followed by a cyclization step that does not require precious metal catalysis. 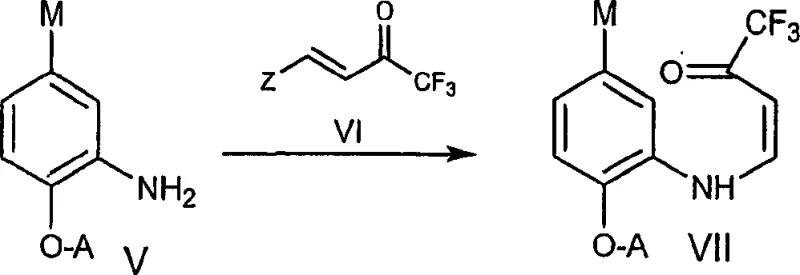 . The elimination of palladium catalysts not only reduces the direct material costs but also simplifies the downstream processing, as there is no need for complex metal removal steps. The reaction conditions are optimized to operate at lower temperatures and with higher yields, as demonstrated by the exemplary yields reported in the patent examples. This efficiency translates directly into a more sustainable manufacturing process with a reduced environmental footprint, aligning with modern green chemistry principles. For procurement teams, this means a more stable supply chain with less dependency on volatile precious metal markets, ensuring consistent availability of the critical intermediate for downstream drug production.
. The elimination of palladium catalysts not only reduces the direct material costs but also simplifies the downstream processing, as there is no need for complex metal removal steps. The reaction conditions are optimized to operate at lower temperatures and with higher yields, as demonstrated by the exemplary yields reported in the patent examples. This efficiency translates directly into a more sustainable manufacturing process with a reduced environmental footprint, aligning with modern green chemistry principles. For procurement teams, this means a more stable supply chain with less dependency on volatile precious metal markets, ensuring consistent availability of the critical intermediate for downstream drug production.
Mechanistic Insights into POCl3-Catalyzed Cyclization
The core of this synthetic innovation lies in the efficient cyclization step that constructs the quinoline ring system with high regioselectivity and yield. The process involves treating the intermediate enaminone with a suitable dehydrating agent, such as phosphorus oxychloride (POCl3), in a solvent like acetonitrile or ethyl acetate. 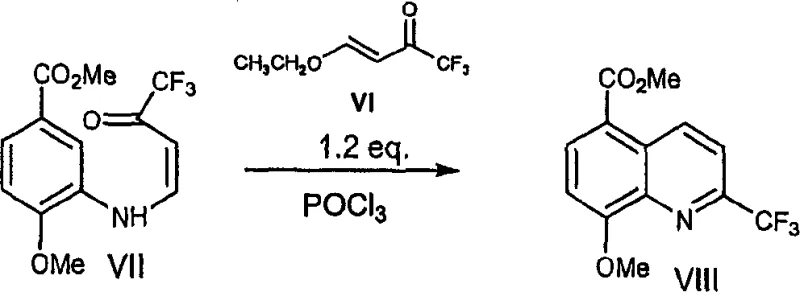 . This cyclization mechanism proceeds through the activation of the carbonyl group, facilitating an intramolecular nucleophilic attack that closes the ring to form the quinoline structure. The choice of solvent plays a critical role in this transformation, with acetonitrile proving to be particularly effective in promoting the reaction while minimizing side reactions. The use of POCl3 as a dehydrating agent is advantageous due to its high reactivity and availability, allowing the reaction to proceed to completion within a reasonable timeframe under reflux conditions. This mechanistic pathway ensures that the trifluoromethyl group is correctly positioned at the 2-position of the quinoline moiety, which is essential for the biological activity of the final PDE IV inhibitor. Understanding this mechanism allows R&D teams to fine-tune reaction parameters for optimal performance during scale-up, ensuring that the process remains robust and reproducible across different batch sizes.
. This cyclization mechanism proceeds through the activation of the carbonyl group, facilitating an intramolecular nucleophilic attack that closes the ring to form the quinoline structure. The choice of solvent plays a critical role in this transformation, with acetonitrile proving to be particularly effective in promoting the reaction while minimizing side reactions. The use of POCl3 as a dehydrating agent is advantageous due to its high reactivity and availability, allowing the reaction to proceed to completion within a reasonable timeframe under reflux conditions. This mechanistic pathway ensures that the trifluoromethyl group is correctly positioned at the 2-position of the quinoline moiety, which is essential for the biological activity of the final PDE IV inhibitor. Understanding this mechanism allows R&D teams to fine-tune reaction parameters for optimal performance during scale-up, ensuring that the process remains robust and reproducible across different batch sizes.
Impurity control is another critical aspect of this mechanism, as the formation of by-products can significantly impact the purity profile of the final drug substance. The patent details specific workup procedures, such as quenching the reaction mixture with aqueous potassium carbonate, which effectively neutralizes excess dehydrating agent and facilitates the separation of the organic product. The subsequent purification steps, including crystallization from methanol, are designed to remove any remaining impurities and isolate the desired quinoline carboxylate in high purity. This focus on impurity control is vital for meeting the stringent specifications required for pharmaceutical intermediates, as even trace impurities can affect the safety and efficacy of the final drug product. By optimizing the stoichiometry of the reagents and the temperature profile of the reaction, manufacturers can minimize the formation of undesired side products and maximize the yield of the target compound. This level of control over the chemical process provides a significant competitive advantage, ensuring that the supply of the intermediate remains consistent and reliable for long-term commercial production.
How to Synthesize Quinoline 5-Carboxamides Efficiently
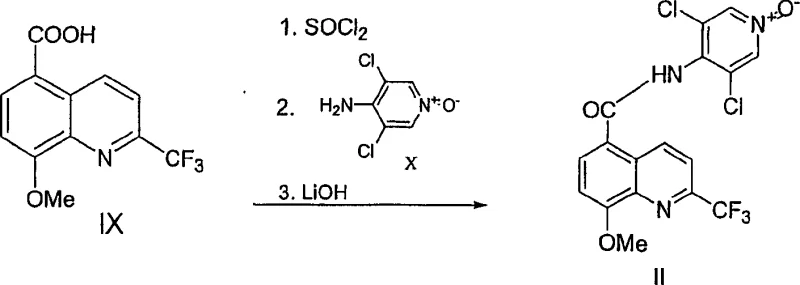
The synthesis of these valuable quinoline 5-carboxamides is achieved through a logical four-step sequence that begins with the condensation of an aniline derivative and concludes with an amide coupling reaction. The initial step involves reacting a substituted aniline with a trifluoro enone to form an enaminone intermediate, which serves as the precursor for the ring-closing reaction. Following this, the enaminone undergoes cyclization in the presence of a dehydrating agent to generate the quinoline core, which is then hydrolyzed to the corresponding carboxylic acid if the starting material was an ester. The final step involves converting the acid to an acid chloride and coupling it with an aminopyridine N-oxide to yield the target carboxamide. Detailed standardized synthesis steps see the guide below.
- Condense an aniline derivative with a trifluoro enone to form an intermediate enaminone.
- Cyclize the intermediate using a dehydrating agent such as phosphorus oxychloride to form the quinoline core.
- Hydrolyze the ester group to a carboxylic acid if necessary using aqueous base.
- Couple the resulting acid chloride with an aminopyridine N-oxide to form the final carboxamide.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic route offers profound commercial benefits that directly address the pain points of traditional supply chains, particularly regarding cost stability and material availability. By eliminating the need for expensive palladium catalysts and complex purification protocols, the process significantly reduces the overall cost of goods sold, making it an attractive option for large-scale manufacturing. The use of commercially available starting materials further enhances supply chain reliability, as there is no dependency on custom-synthesized reagents that may have long lead times or limited availability. This robustness ensures that production schedules can be maintained without interruption, even in the face of market fluctuations for raw materials. Furthermore, the simplified workflow reduces the operational complexity of the manufacturing process, allowing for faster turnaround times and increased production capacity. These factors combined create a resilient supply chain capable of meeting the demanding requirements of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant cost driver from the manufacturing budget, as palladium salts are not only expensive to purchase but also costly to dispose of in compliance with environmental regulations. Additionally, the higher yields achieved through this optimized process mean that less raw material is wasted, further driving down the cost per kilogram of the final product. The simplified purification steps reduce the consumption of solvents and energy, contributing to substantial cost savings in utility and waste management expenses. These cumulative efficiencies result in a more competitive pricing structure for the intermediate, allowing downstream drug manufacturers to optimize their own production costs. Consequently, this process enables a more sustainable economic model for the production of PDE IV inhibitors, benefiting all stakeholders in the supply chain.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals rather than specialized reagents ensures a stable and continuous supply of raw materials, mitigating the risk of production delays due to material shortages. The robustness of the reaction conditions means that the process is less sensitive to variations in raw material quality, further enhancing the reliability of the supply chain. This stability is crucial for maintaining consistent production schedules and meeting delivery commitments to customers, thereby strengthening business relationships. Moreover, the scalability of the process allows for flexible production volumes, enabling suppliers to respond quickly to changes in demand without compromising quality. This adaptability is a key factor in building a resilient supply chain that can withstand external disruptions and maintain operational continuity.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and common solvents that are easily handled in large-scale facilities. The absence of heavy metal catalysts simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations and reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer. The ability to scale from laboratory to commercial production without significant process re-engineering reduces the time and investment required to bring the product to market. This efficiency accelerates the commercialization timeline, allowing patients to benefit from new therapies sooner while maximizing the return on investment for the manufacturer.
Frequently Asked Questions (FAQ)
The following questions and answers are derived from the technical details of the patent to address common concerns regarding the implementation and benefits of this synthesis. They cover aspects of process efficiency, purity control, and scalability, providing clarity for technical decision-makers evaluating this technology.
Q: What are the primary advantages of this synthesis over prior art methods?
A: This method avoids the use of expensive palladium catalysts and tedious purification steps, utilizing more economical starting materials and achieving higher yields under milder conditions.
Q: How is high purity maintained during the commercial scale-up of complex quinoline derivatives?
A: High purity is ensured through specific solvent choices during cyclization and crystallization steps, which effectively remove impurities and by-products without requiring complex chromatography.
Q: Is this process suitable for reducing lead time for high-purity pharmaceutical intermediates?
A: Yes, the streamlined reaction sequence and the use of commercially available reagents significantly simplify the supply chain, allowing for faster production cycles and reliable delivery schedules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline 5-Carboxamides Supplier
NINGBO INNO PHARMCHEM stands as a premier partner for the commercialization of this advanced synthesis, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at translating complex laboratory routes into robust industrial processes, ensuring that the high yields and purity specifications demonstrated in the patent are maintained at scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest quality standards required for pharmaceutical applications. Our commitment to excellence ensures that our clients receive a reliable supply of high-quality intermediates that support their drug development and commercialization goals. By partnering with us, you gain access to a wealth of technical expertise and manufacturing capacity that can accelerate your project timelines and reduce your overall development risks.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient route. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Contact us today to explore how we can collaborate to bring your PDE IV inhibitor projects to successful commercialization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
