Industrial Scale-Up of 6-Methyl-3-Aminopyridazine via Efficient Ammoniation and Recycling
The pharmaceutical and fine chemical industries continuously seek robust methodologies for synthesizing heterocyclic building blocks, particularly those containing the pyridazine scaffold which is prevalent in bioactive molecules. Patent CN103058933A introduces a significant advancement in the industrial preparation of 6-methyl-3-aminopyridazine, a critical intermediate for various therapeutic agents. This technology addresses long-standing challenges in scalability and cost-efficiency by utilizing 6-methyl-3-chloropyridazine as the primary substrate for a direct ammoniation reaction. Unlike traditional routes that suffer from complex multi-step sequences and hazardous conditions, this novel approach streamlines the synthesis into a manageable high-pressure autoclave process. The method not only simplifies the operational workflow but also integrates a sophisticated by-product recovery system that transforms potential waste into valuable feedstock. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminopyridazines has been plagued by severe operational constraints and safety hazards associated with harsh reaction conditions. Conventional literature often describes pathways initiating from 6-methyl-pyridazin-3-one, which requires chlorination followed by ammoniation under extreme thermal stress. As illustrated in the reaction schemes below, traditional ammoniation steps frequently necessitate temperatures reaching 175°C and correspondingly high pressures, imposing rigorous demands on reactor metallurgy and safety systems. These elevated conditions not only accelerate equipment degradation and increase maintenance costs but also elevate the risk profile of the manufacturing process due to the potential for runaway reactions. Furthermore, the reliance on stoichiometric amounts of chlorinating agents like phosphorus oxychloride in early stages generates substantial acidic waste streams, complicating environmental compliance and disposal logistics. The cumulative effect of these factors results in a fragmented supply chain with higher production costs and inconsistent batch quality.
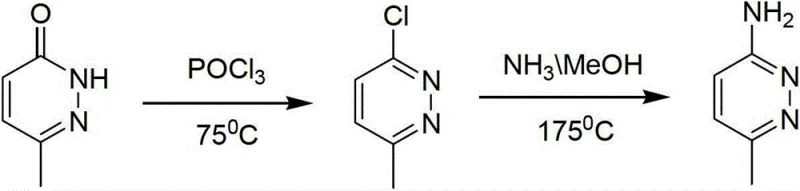
The Novel Approach
The methodology disclosed in CN103058933A fundamentally reengineers the production landscape by optimizing the ammoniation step to operate under significantly milder parameters. By reacting 6-methyl-3-chloropyridazine with concentrated ammonia water at a controlled temperature range of 120°C to 150°C and a moderate pressure of 0.2 to 0.4 MPa, the process drastically reduces the thermal load on the reaction vessel. This moderation in conditions allows for the use of standard industrial autoclaves rather than specialized high-pressure equipment, thereby lowering capital expenditure barriers for scale-up. Moreover, the process design inherently accounts for the formation of the hydrolysis by-product, 6-methyl-pyridazin-3-one, treating it not as waste but as a recyclable asset. This strategic shift eliminates the need for excessive chlorinating reagents in the main synthetic line, as the by-product can be looped back to regenerate the starting chloro-compound. Consequently, the overall atom economy is improved, and the generation of hazardous waste is minimized, aligning the production process with modern green chemistry principles.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a nucleophilic aromatic substitution (SnAr) where the chlorine atom at the 3-position of the pyridazine ring is displaced by an amino group from the ammonia source. The electron-deficient nature of the pyridazine ring, augmented by the electronegative nitrogen atoms, activates the carbon-chlorine bond towards nucleophilic attack. In this specific protocol, the use of concentrated ammonia water (25%-28%) provides a high concentration of the nucleophile, driving the equilibrium towards the desired amine product despite the competing hydrolysis reaction that forms the ketone by-product. The reaction kinetics are carefully balanced by the temperature window of 120°C-150°C; temperatures below this range may result in incomplete conversion, while exceeding them could favor excessive hydrolysis or decomposition. Understanding this mechanistic balance is crucial for R&D teams aiming to replicate the process, as slight deviations in ammonia concentration or thermal profiles can significantly impact the ratio of amine to ketone formed.
Impurity control is meticulously managed through a pH-dependent crystallization strategy that exploits the differing solubility profiles of the product and the by-product. Following the reaction, the mixture is cooled and neutralized to a pH of 7-8 using dilute hydrochloric acid, a condition under which the target 6-methyl-3-aminopyridazine precipitates as a solid while the by-product remains largely in solution. This selective precipitation is a critical purification step that avoids the need for complex chromatographic separations or extensive solvent extractions. The mother liquor, enriched with the 6-methyl-pyridazin-3-one by-product, is subsequently subjected to further pH adjustment to 5.0-5.5 to isolate the ketone. This precise manipulation of acidity ensures that the final product achieves a high purity of 98%, effectively removing trace impurities and unreacted starting materials. Such robust control over the impurity profile is essential for pharmaceutical applications where strict regulatory limits on genotoxic impurities and heavy metals must be maintained.
How to Synthesize 6-Methyl-3-Aminopyridazine Efficiently
The implementation of this industrial synthesis route requires strict adherence to the specified reaction parameters to ensure optimal yield and safety. The process begins with the charging of 6-methyl-3-chloropyridazine and concentrated ammonia into a pressure-rated reactor, followed by a controlled heating ramp to the target temperature zone. Maintaining the pressure between 0.2 and 0.4 MPa is vital to keep the ammonia in the liquid phase and ensure sufficient contact with the substrate. Post-reaction workup involves a sequential pH adjustment protocol that first isolates the amine product and then recovers the ketone by-product from the filtrate. This dual-isolation strategy maximizes material efficiency and simplifies the downstream processing train. For detailed operational specifics regarding stirring rates, cooling gradients, and filtration techniques, please refer to the standardized synthesis guide below.
- React 6-methyl-3-chloropyridazine with concentrated ammonia water (25%-28%) in an autoclave at 120°C-150°C and 0.2-0.4MPa for 5-12 hours.
- Cool the reaction mixture, adjust pH to 7-8 with dilute hydrochloric acid, and allow to stand for 48 hours to precipitate the product.
- Filter the product, then concentrate the filtrate and adjust pH to 5.0-5.5 to recover the by-product 6-methyl-pyridazin-3-one for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility in the fine chemical sector. The ability to recycle the hydrolysis by-product back into the starting material creates a closed-loop system that significantly reduces the net consumption of raw materials. This internal recycling mechanism acts as a buffer against fluctuations in the market price of precursors, providing greater cost predictability for long-term contracts. Furthermore, the elimination of harsh chlorination steps in the main production line reduces the dependency on hazardous reagents like phosphorus oxychloride, thereby lowering the costs associated with safety management, specialized storage, and waste disposal. These operational efficiencies translate into a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Cost Reduction in Manufacturing: The integration of a by-product recycling loop fundamentally alters the cost structure of the synthesis by converting what was previously waste into a valuable feedstock. By recovering 6-methyl-pyridazin-3-one and converting it back to 6-methyl-3-chloropyridazine, the effective yield of the overall process is substantially increased, reducing the amount of fresh starting material required per kilogram of final product. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure minimizes the usage of organic solvents and auxiliary chemicals. These cumulative savings allow for a drastic reduction in the variable cost of goods sold, enhancing the margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route contributes to a more resilient supply chain by minimizing the risk of production stoppages due to equipment failure or safety incidents. Operating at lower pressures and temperatures reduces the mechanical stress on reactors and valves, extending their service life and decreasing the frequency of unplanned maintenance shutdowns. Moreover, the process relies on readily available and stable reagents such as concentrated ammonia and hydrochloric acid, avoiding the supply bottlenecks often associated with specialized or highly regulated catalysts. This reliability ensures consistent delivery schedules and helps pharmaceutical partners maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, having been demonstrated effectively from gram-scale experiments to multi-kilogram pilot batches without loss of efficiency. The reduction in hazardous waste generation, particularly the minimization of acidic effluents and chlorinated by-products, simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. Compliance with increasingly stringent environmental regulations is easier to achieve when the process generates fewer "three wastes" (waste gas, waste water, waste residue). This environmental stewardship not only mitigates regulatory risk but also aligns with the sustainability goals of major multinational corporations, making the supplier a more attractive partner for green procurement initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific preparation method. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a reliable basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific supply chain requirements.
Q: How does this process improve upon conventional ammoniation methods?
A: Unlike conventional methods requiring extreme temperatures up to 175°C and high pressure, this patented process operates at milder conditions (120°C-150°C, 0.2-0.4MPa), significantly reducing equipment stress and energy consumption while maintaining high purity.
Q: What is the strategy for handling reaction by-products?
A: The process features a closed-loop recycling system where the hydrolysis by-product, 6-methyl-pyridazin-3-one, is isolated via pH adjustment and subsequently converted back into the starting material, 6-methyl-3-chloropyridazine, minimizing waste and raw material costs.
Q: What purity levels can be achieved with this industrial method?
A: Through optimized crystallization and pH control during workup, the final product 6-methyl-3-aminopyridazine consistently achieves a purity of 98%, meeting stringent requirements for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methyl-3-Aminopyridazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceutical entities. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering 6-methyl-3-aminopyridazine with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement complex recycling loops and optimize reaction conditions allows us to offer a product that meets the exacting standards of the global pharmaceutical industry while maintaining cost competitiveness.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this recycled-feedstock methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the quality and reliability of our supply before committing to large-scale orders. Let us collaborate to build a more efficient and sustainable supply chain for your critical heterocyclic building blocks.
