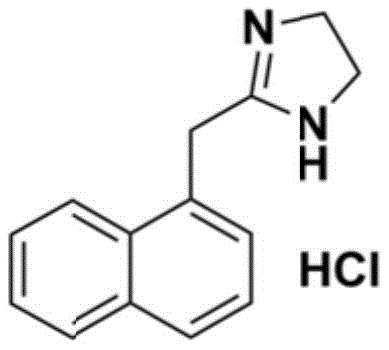
Advanced Catalytic Synthesis of Naphazoline Hydrochloride for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for vasoactive agents that balance high purity with manufacturing efficiency. A significant advancement in this domain is detailed in patent CN110041261B, which discloses a novel preparation method for Naphazoline Hydrochloride, a critical active pharmaceutical ingredient used globally for relieving nasal congestion. This specific technical disclosure outlines a sulfur-catalyzed cyclization strategy that fundamentally alters the production landscape for this imidazoline derivative. By leveraging specific catalysts such as carbon disulfide, sodium sulfide, or thiourea, the process achieves exceptional yield and purity metrics while simplifying the operational workflow. For R&D Directors and Procurement Managers evaluating reliable pharmaceutical intermediates suppliers, this methodology represents a pivotal shift away from hazardous and operationally rigid legacy processes. The ability to produce high-purity Naphazoline Hydrochloride with superior powder properties directly impacts the downstream formulation efficiency, making this patent data a cornerstone for strategic sourcing decisions in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
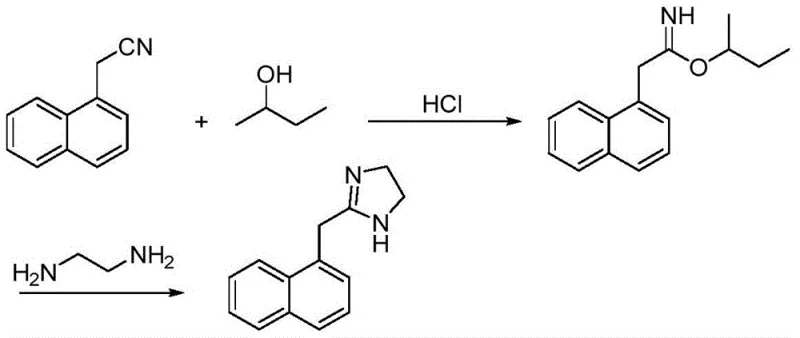
Historically, the industrial synthesis of Naphazoline Hydrochloride has been plagued by significant technical bottlenecks that hinder cost-effective manufacturing and supply chain stability. Prior art, such as the method disclosed in Japanese patent JP06345737, relies on the formation of an imidate intermediate using naphthaleneacetonitrile and alcohols under strictly anhydrous conditions. This requirement for absolute moisture control imposes severe constraints on reactor preparation and raw material quality, drastically increasing operational complexity and the risk of batch failure. Furthermore, alternative routes utilizing thioacetamide often necessitate the use of toxic solvents like chloroform and complex post-treatment procedures involving ice quenching and petroleum ether grinding. These legacy methods not only pose substantial environmental and safety hazards but also result in products with inconsistent powder morphology, characterized by high angles of repose and poor flowability. Such deficiencies complicate downstream tableting processes and often require additional milling steps, thereby inflating the overall cost of goods sold and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
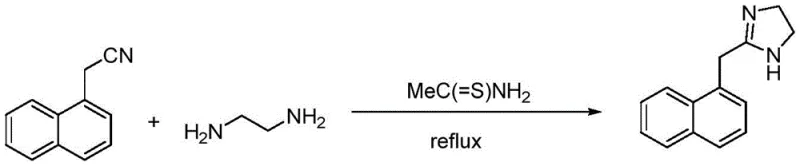
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent data introduces a streamlined sulfur-catalyzed pathway that eliminates the need for strict anhydrous environments and hazardous chlorinated solvents. By utilizing naphthaleneacetonitrile and ethylenediamine in the presence of catalytic amounts of sulfur-containing compounds, the reaction proceeds efficiently at moderate temperatures ranging from 90-120°C. This methodological shift allows for the direct formation of the naphazoline free base, which is subsequently converted to the hydrochloride salt through a controlled salification process. The elimination of toxic solvents and the relaxation of moisture constraints significantly reduce the environmental footprint and operational risk associated with production. Moreover, the process incorporates a optimized recrystallization step that ensures the final product exhibits excellent physical properties, including low bulk density and a loose particle structure. This breakthrough offers a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing by simplifying unit operations and enhancing overall process safety.
Mechanistic Insights into Sulfur-Catalyzed Cyclization
The core innovation of this synthesis lies in the mechanistic role of the sulfur-containing catalyst in facilitating the cyclization of the nitrile group with ethylenediamine. In the absence of a catalyst, the nucleophilic attack of the amine on the nitrile carbon is often sluggish or requires extreme conditions that promote side reactions. The introduction of catalysts such as CS2, Na2S, or thiourea activates the nitrile functionality, likely through the formation of a transient thioimidate or similar activated intermediate that is more susceptible to nucleophilic attack by the diamine. This activation lowers the energy barrier for the ring-closing step, allowing the reaction to proceed to completion within a relatively short timeframe of 1.5 to 3 hours at temperatures between 90-120°C. The specific molar ratio of the catalyst to the naphthylacetonitrile, optimized between 0.05 and 0.12:1, is critical for maximizing conversion while minimizing the formation of sulfur-containing byproducts that could complicate purification. Understanding this catalytic cycle is essential for R&D teams aiming to replicate or scale this process, as it highlights the delicate balance between catalyst loading and reaction kinetics required to achieve the reported high yields.
Beyond the primary cyclization, the control of impurities and crystal growth is meticulously managed through the salification and recrystallization stages. The patent specifies a precise water addition protocol during the separation of the free base, where water is added at a rate of 0.8-1.5L/min to induce precipitation. This controlled addition prevents the rapid formation of amorphous solids or oiling out, which can trap impurities within the crystal lattice. Subsequently, the salification step utilizes hydrochloric acid at a concentration of 5-30% to adjust the pH to a narrow range of 1-3. This specific acidity window ensures complete salt formation without promoting acid-catalyzed degradation or the co-precipitation of colored impurities. The final recrystallization from a methanol-acetone system further refines the purity, removing trace organic impurities and ensuring the final API intermediate meets stringent pharmacopeial standards. This multi-stage purification strategy is key to achieving the reported purity levels exceeding 99.90%, which is vital for ensuring the safety and efficacy of the final drug product.
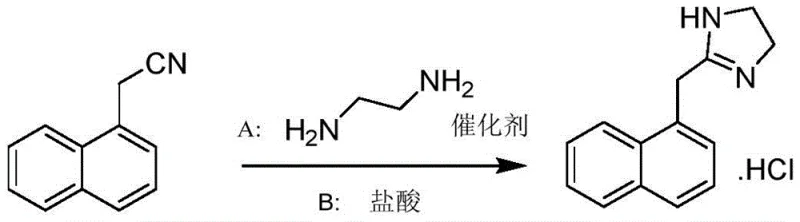
How to Synthesize Naphazoline Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding temperature, catalyst loading, and solvent ratios to ensure reproducibility and quality. The process begins with the charging of naphthaleneacetonitrile and ethylenediamine into a reaction vessel, followed by the addition of the selected sulfur catalyst. The mixture is heated to the target temperature and maintained for the specified duration to ensure complete conversion. Following the reaction, the workup involves a controlled water addition to precipitate the free base, which is then filtered and dried. The subsequent salification and recrystallization steps are equally critical, requiring precise control over pH and cooling rates to achieve the desired crystal habit. For a comprehensive understanding of the operational specifics, the detailed standardized synthesis steps are provided in the guide below, which breaks down the exact quantities and conditions for laboratory and pilot-scale execution.
- React naphthaleneacetonitrile with ethylenediamine using a sulfur-containing catalyst such as CS2, Na2S, or thiourea at 90-120°C to form the naphazoline free base.
- Dissolve the free base in an organic solvent like acetonitrile and adjust pH to 1-3 using hydrochloric acid to precipitate the crude hydrochloride salt.
- Recrystallize the crude product using a methanol-acetone solvent system with activated carbon decolorization to obtain the final high-purity fine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this sulfur-catalyzed synthesis route offers substantial strategic advantages for procurement managers and supply chain heads focused on cost efficiency and reliability. The primary benefit stems from the drastic simplification of the process workflow, which eliminates the need for specialized anhydrous reactors and the handling of hazardous chlorinated solvents. This reduction in operational complexity translates directly into lower capital expenditure requirements and reduced operating costs, as the process can be run in standard glass-lined or stainless steel reactors without the need for extensive drying protocols. Furthermore, the use of readily available and inexpensive catalysts like sodium sulfide or thiourea ensures that raw material costs remain stable and predictable, mitigating the risk of supply chain disruptions associated with specialty reagents. The ability to source reliable pharmaceutical intermediates supplier capabilities that can execute this simpler chemistry enhances supply security and allows for more flexible production scheduling.
- Cost Reduction in Manufacturing: The elimination of toxic solvents such as chloroform and the removal of energy-intensive anhydrous conditions significantly lower the variable costs associated with production. By avoiding the need for solvent recovery systems dedicated to hazardous waste and reducing the energy load for drying reactors, manufacturers can achieve substantial cost savings. Additionally, the high yield and purity achieved in fewer steps reduce the volume of waste generated per kilogram of product, further driving down disposal costs and improving the overall economic efficiency of the manufacturing process. This logical deduction of cost benefits makes the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain by minimizing the risk of batch failures due to moisture ingress or reagent instability. Since the process does not require strict anhydrous conditions, the sensitivity to environmental factors is reduced, leading to more consistent batch-to-batch quality and higher first-pass yields. This reliability ensures that delivery schedules can be met with greater confidence, reducing the lead time for high-purity pharmaceutical intermediates and allowing downstream formulators to maintain leaner inventory levels. The use of common, non-restricted reagents also simplifies procurement logistics, ensuring that raw material availability is not a bottleneck for production continuity.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with parameters such as water addition rates and temperature profiles that are easily controlled in large-scale reactors. The avoidance of hazardous solvents aligns with increasingly stringent environmental regulations, reducing the regulatory burden and the risk of compliance-related shutdowns. The improved powder properties of the final product, characterized by a low angle of repose and good flowability, also facilitate easier handling and packaging at scale, reducing downtime during filling operations. This combination of scalability and environmental friendliness positions the technology as a sustainable choice for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and technical feasibility of the sulfur-catalyzed route. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or contract manufacturing partnerships. The responses highlight the specific advantages in terms of purity, safety, and process efficiency that distinguish this method from conventional alternatives.
Q: How does the sulfur-catalyzed method improve upon traditional anhydrous synthesis routes?
A: Unlike traditional methods requiring strict anhydrous conditions and toxic solvents like chloroform, the sulfur-catalyzed approach operates under simpler conditions, eliminating the need for rigorous moisture control and hazardous solvent handling, thereby reducing operational risks and costs.
Q: What are the critical parameters for ensuring optimal powder morphology in Naphazoline Hydrochloride?
A: Critical parameters include the specific rate of water addition during the free base separation (0.8-1.5L/min) and the precise pH adjustment during salification (pH 1-3), which collectively ensure loose particle structure, low angle of repose, and excellent flowability for downstream tableting.
Q: Can this synthesis route be scaled for commercial production without compromising purity?
A: Yes, the process is designed for industrial amplification, utilizing common reagents and avoiding complex post-treatment steps like ice quenching or petroleum ether grinding, allowing for consistent production of material with purity exceeding 99.90% and high yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphazoline Hydrochloride Supplier
The technical potential of this sulfur-catalyzed synthesis route is immense, offering a pathway to high-quality Naphazoline Hydrochloride that meets the rigorous demands of the global pharmaceutical market. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to life. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch produced adheres to the highest standards of quality and consistency. We understand the critical nature of API intermediates in the drug development lifecycle and are committed to providing a supply partner that can navigate the complexities of chemical synthesis with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify the compatibility of this process with your existing quality frameworks. Let us collaborate to enhance your supply chain efficiency and secure a reliable source of high-purity intermediates for your critical medications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
