Advanced Synthesis of N'-Aryl-N,N-Dimethylformamidine for Oncology Drug Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for key heterocyclic intermediates, particularly those serving as the backbone for potent tyrosine kinase inhibitors. Patent CN103539702A introduces a transformative methodology for the preparation of N'-aryl-N,N-dimethylformamidine, a critical building block in the synthesis of antitumor agents such as Erlotinib, Lapatinib, and Gefitinib. This innovation addresses a longstanding chemical challenge: the efficient functionalization of aromatic amines bearing electron-withdrawing groups at the ortho position, which traditionally resist standard formamidylation conditions due to steric and electronic deactivation. By leveraging an in situ generated iminium salt from inexpensive commodity chemicals, this process offers a scalable, high-yield alternative that bypasses the limitations of legacy technologies. The strategic importance of this chemistry cannot be overstated, as it directly impacts the supply chain security and cost structure of multi-billion dollar oncology therapies. For procurement leaders and R&D directors alike, understanding this mechanistic shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering complex precursors with consistent quality.
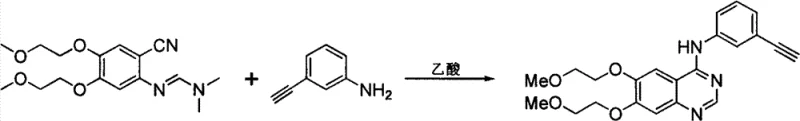
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N'-aryl-N,N-dimethylformamidine has relied heavily on three primary strategies, each fraught with significant operational and economic drawbacks that hinder large-scale commercial viability. The first conventional approach involves the reaction of aromatic primary amines with N,N-dimethylformamide dimethyl acetal (DMF-DMA) at elevated temperatures, a process that often suffers from poor atom economy and requires harsh thermal conditions that can degrade sensitive functional groups. 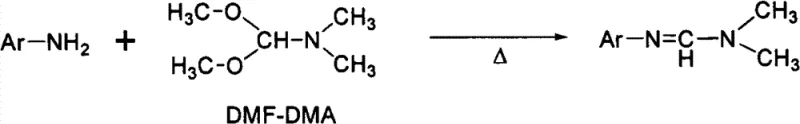 Furthermore, when the aromatic amine possesses an ortho-substituent like a cyano group, the nucleophilicity of the amino nitrogen is severely compromised by intramolecular hydrogen bonding and steric congestion, leading to negligible conversion rates. The second method utilizes Vilsmeier-Haack reagents generated from DMF and phosphorus oxychloride (POCl3), which, while effective for simple substrates, introduces severe environmental and safety liabilities due to the handling of highly corrosive and toxic phosphorus species.
Furthermore, when the aromatic amine possesses an ortho-substituent like a cyano group, the nucleophilicity of the amino nitrogen is severely compromised by intramolecular hydrogen bonding and steric congestion, leading to negligible conversion rates. The second method utilizes Vilsmeier-Haack reagents generated from DMF and phosphorus oxychloride (POCl3), which, while effective for simple substrates, introduces severe environmental and safety liabilities due to the handling of highly corrosive and toxic phosphorus species. 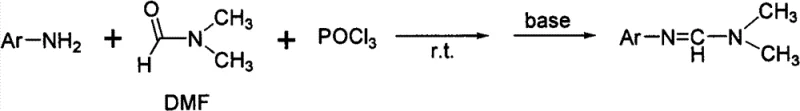 The generation of acidic phosphorus waste streams necessitates complex downstream neutralization and disposal protocols, drastically inflating the cost reduction in API manufacturing calculations. Additionally, the third method employing arylsulfonyl chlorides adds unnecessary complexity and cost by requiring specialized sulfonylating agents that are not as readily available as bulk solvents.
The generation of acidic phosphorus waste streams necessitates complex downstream neutralization and disposal protocols, drastically inflating the cost reduction in API manufacturing calculations. Additionally, the third method employing arylsulfonyl chlorides adds unnecessary complexity and cost by requiring specialized sulfonylating agents that are not as readily available as bulk solvents.
The Novel Approach
In stark contrast to these legacy techniques, the novel protocol described in CN103539702A utilizes a two-step sequence initiated by the quaternization of DMF with dimethyl sulfate to form a highly electrophilic iminium salt. This activation strategy fundamentally alters the reactivity profile of the formyl source, rendering it sufficiently potent to overcome the deactivating effects of ortho-cyano substituents on the aromatic amine. The subsequent coupling reaction proceeds under remarkably mild conditions, typically between 0°C and 60°C, in the presence of a simple alkoxide base such as sodium methoxide. This approach eliminates the need for high-temperature reflux or toxic phosphorylating agents, thereby streamlining the operational workflow and enhancing process safety. The versatility of this method is evidenced by its successful application in synthesizing diverse intermediates for Erlotinib and Gefitinib, where traditional routes often struggle with purity profiles. By shifting the paradigm from direct acetal exchange or Vilsmeier activation to iminium salt coupling, manufacturers can achieve superior impurity control and higher overall throughput, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iminium Salt-Mediated Formamidylation
The core innovation of this technology lies in the generation and utilization of the N,N-dimethyliminium cation, a potent electrophile that drives the formamidylation equilibrium forward even with sterically hindered substrates. The mechanism initiates with the alkylation of the carbonyl oxygen of DMF by dimethyl sulfate, creating a resonance-stabilized cationic species that is far more reactive towards nucleophilic attack than neutral DMF or its acetals. 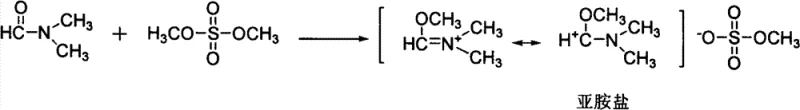 This activation step is crucial because it effectively converts a relatively stable amide into a reactive imidate equivalent without generating stoichiometric amounts of hazardous byproducts. Upon introduction of the ortho-amino benzonitrile, the amino group attacks the iminium carbon, forming a tetrahedral intermediate that subsequently collapses to release methanol and methyl sulfate anions. The presence of a base is mechanistically essential in this second stage, as it facilitates the deprotonation of the ammonium intermediate, regenerating the neutral formamidine product and preventing the reversal of the reaction.
This activation step is crucial because it effectively converts a relatively stable amide into a reactive imidate equivalent without generating stoichiometric amounts of hazardous byproducts. Upon introduction of the ortho-amino benzonitrile, the amino group attacks the iminium carbon, forming a tetrahedral intermediate that subsequently collapses to release methanol and methyl sulfate anions. The presence of a base is mechanistically essential in this second stage, as it facilitates the deprotonation of the ammonium intermediate, regenerating the neutral formamidine product and preventing the reversal of the reaction. 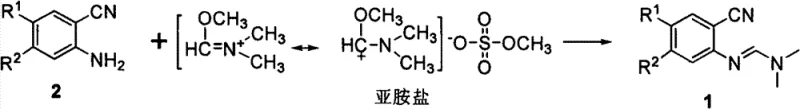 Unlike the DMF-DMA method where methanol elimination is thermally driven, this base-promoted pathway allows for precise kinetic control at lower temperatures, minimizing side reactions such as hydrolysis or polymerization. Furthermore, the specific choice of base, with sodium methoxide proving superior to hydroxides or carbonates, suggests that the basicity and nucleophilicity balance is critical for maintaining the integrity of the sensitive cyano group while ensuring complete conversion of the amine. This mechanistic clarity provides R&D teams with the confidence to adapt the process for analog synthesis, knowing that the electronic demands of the substrate are met by the high energy of the iminium electrophile.
Unlike the DMF-DMA method where methanol elimination is thermally driven, this base-promoted pathway allows for precise kinetic control at lower temperatures, minimizing side reactions such as hydrolysis or polymerization. Furthermore, the specific choice of base, with sodium methoxide proving superior to hydroxides or carbonates, suggests that the basicity and nucleophilicity balance is critical for maintaining the integrity of the sensitive cyano group while ensuring complete conversion of the amine. This mechanistic clarity provides R&D teams with the confidence to adapt the process for analog synthesis, knowing that the electronic demands of the substrate are met by the high energy of the iminium electrophile.
How to Synthesize N'-Aryl-N,N-Dimethylformamidine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the dimethyl sulfate and the timing of base addition to maximize yield and minimize exothermic risks. The process begins with the controlled mixing of DMF and dimethyl sulfate at moderate temperatures to ensure complete salt formation before introducing the amine substrate. Detailed standardized operating procedures regarding solvent selection, specifically the preference for non-polar solvents like toluene to facilitate product isolation, are critical for reproducibility. For a comprehensive guide on the exact molar ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the technical instructions below.
- React N,N-dimethylformamide (DMF) with dimethyl sulfate at 40°C to 90°C to generate the reactive iminium salt intermediate.
- In a separate vessel, dissolve the ortho-amino aromatic nitrile in an organic solvent such as toluene or ethyl acetate.
- Add the iminium salt to the amine solution in the presence of a base (e.g., sodium methoxide) at 0°C to 60°C to yield the target formamidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this novel synthesis route offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility inherent in traditional intermediate manufacturing. By replacing expensive and specialized reagents like DMF-DMA or POCl3 with commodity chemicals such as dimethyl sulfate and DMF, the raw material cost basis is significantly reduced, providing a buffer against market price fluctuations. The elimination of phosphorus-based waste streams not only lowers environmental compliance costs but also simplifies the regulatory burden associated with hazardous waste disposal, contributing to substantial cost savings in overall production. Moreover, the mild reaction conditions reduce energy consumption and equipment wear, allowing for longer campaign runs and improved asset utilization rates. For supply chain heads, the robustness of this chemistry means reduced lead time for high-purity pharmaceutical intermediates, as the process is less prone to batch failures caused by thermal degradation or reagent instability. The ability to synthesize difficult ortho-substituted intermediates in high yield ensures a steady flow of materials for downstream API production, mitigating the risk of drug shortages.
- Cost Reduction in Manufacturing: The substitution of high-cost activating agents with bulk commodity chemicals drastically lowers the bill of materials, while the simplified workup procedure reduces solvent usage and processing time. The avoidance of expensive catalysts or specialized reagents means that the marginal cost per kilogram is optimized for large-scale production without compromising quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available raw materials like dimethyl sulfate and DMF ensures that production is not bottlenecked by the scarcity of niche reagents, guaranteeing consistent availability for long-term contracts. The robustness of the reaction against substrate variations allows for flexible sourcing of amine precursors, further strengthening supply continuity.
- Scalability and Environmental Compliance: The absence of toxic phosphorus byproducts aligns with modern green chemistry principles, facilitating easier permitting and expansion of manufacturing capacity. The process is inherently safer to scale due to the moderate temperature requirements, reducing the need for specialized high-pressure or high-temperature reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process optimization and regulatory considerations. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is this new method superior for synthesizing ortho-cyano substituted formamidines?
A: Conventional methods using DMF-DMA often fail with ortho-cyano groups due to steric hindrance and hydrogen bonding that block nucleophilic attack. This novel iminium salt pathway overcomes these electronic barriers, allowing high-yield synthesis of critical intermediates for drugs like Erlotinib.
Q: Does this process eliminate the use of hazardous phosphorus oxychloride (POCl3)?
A: Yes, unlike the Vilsmeier-Haack approach which requires toxic POCl3, this method utilizes dimethyl sulfate and DMF, significantly reducing environmental hazards and simplifying waste treatment protocols.
Q: What represents the optimal base for this coupling reaction?
A: Experimental data indicates that alkoxides like sodium methoxide (NaOCH3) provide superior yields compared to hydroxides or carbonates, ensuring efficient deprotonation and driving the reaction to completion under mild conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N'-Aryl-N,N-Dimethylformamidine Supplier
As the demand for targeted oncology therapies continues to surge, the need for high-quality, cost-effective intermediates like N'-aryl-N,N-dimethylformamidine has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies such as the iminium salt coupling process to deliver superior products. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global pharmaceutical partners. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to optimize your supply chain for Erlotinib, Gefitinib, and related kinase inhibitor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this novel route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can become a strategic asset for your organization.
