Advanced Catalytic Synthesis of L-Carnitine: Overcoming Toxicity and Scalability Barriers
Introduction to Next-Generation L-Carnitine Manufacturing
The global demand for high-purity L-carnitine, a critical quaternary ammonium compound essential for lipid metabolism and widely used in nutraceutical and pharmaceutical applications, necessitates manufacturing processes that balance stereochemical precision with environmental safety. Patent CN102971285A introduces a transformative methodology for the production of L-carnitine, shifting away from traditional microbiological fermentation or racemic synthesis towards a highly efficient chiral synthetic route. This technology leverages an asymmetric [2+2] cycloaddition between ketene and specific aldehyde precursors, facilitated by advanced chiral catalysts, to generate a beta-lactone intermediate with exceptional enantiomeric excess. By addressing the historical reliance on toxic organotin reagents and complex multi-step deprotection sequences, this innovation offers a robust pathway for reliable L-carnitine supplier networks aiming to enhance product purity while adhering to stringent regulatory standards for food and feed additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of L-carnitine has been plagued by significant safety and efficiency hurdles, particularly in routes derived from chloral and ketene as described by Song et al. in 1995. These legacy processes typically require the use of hazardous trichloroacetaldehyde and, more critically, depend on stoichiometric amounts of organotin hydrides (such as n-Bu3SnH) to sequentially remove chlorine atoms from the carbon backbone. The necessity of synthesizing n-Bu3SnH in situ adds substantial operational complexity and cost, while the toxicity of tin residues poses severe challenges for downstream purification, especially when the final product is intended for human consumption. Furthermore, these conventional pathways involve multiple discrete steps to convert trichloro-intermediates into monochlorinated equivalents before the final amination, resulting in prolonged cycle times and reduced overall atom economy.
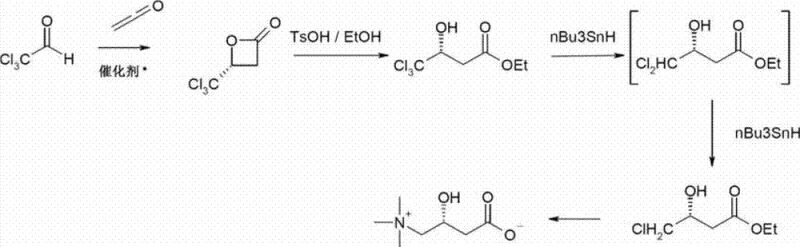
The Novel Approach
In stark contrast, the methodology disclosed in the present invention streamlines the synthesis by utilizing mono-haloacetaldehydes or trimethylammonioacetaldehydes directly in a catalytic [2+2] cycloaddition. This approach fundamentally alters the reaction landscape by generating a chiral beta-lactone precursor that requires significantly fewer transformation steps to reach the final active pharmaceutical ingredient. A pivotal breakthrough in this novel approach is the discovery that the ring-opening of the beta-lactone and the subsequent nucleophilic substitution with trimethylamine can be effectively combined into a single operational step. By carefully controlling reaction conditions, specifically the simultaneous or near-simultaneous addition of metal hydroxides and trimethylamine, the process suppresses competitive side reactions such as elimination to hydroxycrotonic acid, thereby achieving high selectivity for the desired L-carnitine structure without the need for toxic tin-based reductants.
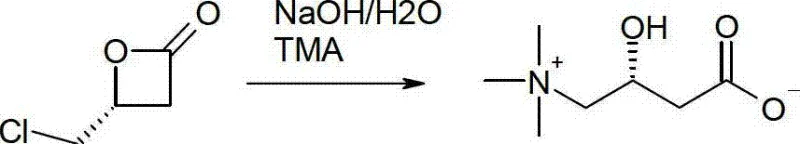
Mechanistic Insights into Asymmetric [2+2] Cycloaddition
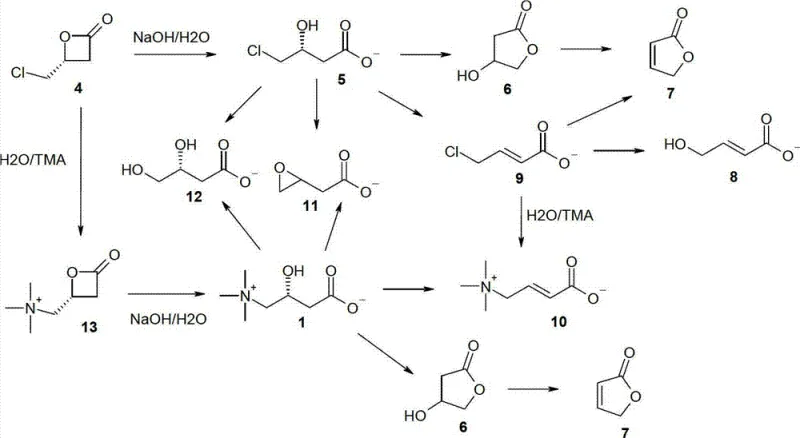
The core of this technological advancement lies in the deployment of sophisticated Lewis acid-Lewis base bifunctional metal catalysts that orchestrate the stereochemical outcome of the cycloaddition. These catalysts, which may include cinchona alkaloid derivatives like (trimethylsilyl)quinine paired with lithium perchlorate, or transition metal complexes based on cobalt and aluminum, function by simultaneously activating the electrophilic aldehyde and organizing the nucleophilic ketene within a chiral environment. The bifunctional nature ensures that the chirality is imparted through the ligand architecture rather than solely relying on the metal center, allowing for precise control over the formation of the 4-substituted oxetan-2-one ring. This mechanistic precision is crucial for ensuring that the resulting beta-lactone possesses the correct (R)-configuration, which is the prerequisite for obtaining biologically active L-carnitine upon ring opening, thus eliminating the need for costly and wasteful resolution of racemic mixtures.
Furthermore, the patent provides a detailed mechanistic understanding of the ring-opening phase, which was previously considered prone to failure due to competing degradation pathways. The inventors identified that under standard basic hydrolysis conditions, the beta-lactone intermediate could undergo elimination to form unsaturated acids or cyclize into unwanted lactones. However, by optimizing the stoichiometry of the base (using a low surplus of 1.1 to 1.6 equivalents of NaOH) and managing the concentration of trimethylamine, the reaction trajectory is shifted decisively towards nucleophilic substitution. This suppression of side products like crotonobetaine and hydroxycrotonic acid is achieved through kinetic control, where the rate of ring opening and amination outpaces the rate of elimination, ensuring that the final impurity profile meets the rigorous specifications required for high-purity nutraceutical ingredients.
How to Synthesize L-Carnitine Efficiently
The implementation of this synthesis route involves a sequence of highly controlled chemical transformations designed to maximize yield and enantiomeric purity while minimizing waste generation. The process begins with the preparation of the chiral catalyst system, often formed in situ by mixing a chiral ligand with a metal salt, followed by the slow introduction of ketene gas into a cooled solution of the haloacetaldehyde precursor. Once the chiral beta-lactone is formed, it can be isolated or telescoped directly into the hydrolysis step, where precise temperature management between 0°C and 25°C is critical to prevent thermal degradation. The detailed standardized synthesis steps, including specific solvent choices like dichloromethane or acetonitrile and quenching protocols, are outlined below to guide process development teams in replicating these results.
- Perform enantioselective [2+2] cycloaddition of ketene and haloacetaldehyde using a chiral Lewis acid-Lewis base bifunctional catalyst.
- Isolate the chiral beta-lactone intermediate or proceed directly to the next step without purification.
- Execute a controlled one-pot ring-opening reaction using aqueous NaOH and trimethylamine (TMA) to yield high-purity L-carnitine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this catalytic route offers substantial advantages by decoupling production from the volatile supply chains associated with specialized organotin reagents and activated chloral. The ability to utilize readily available mono-haloacetaldehydes and simple ketene gas significantly reduces raw material costs and mitigates the risk of supply disruptions caused by the limited availability of exotic starting materials. Moreover, the elimination of heavy metal catalysts and toxic tin byproducts simplifies the wastewater treatment process, leading to significant cost reduction in L-carnitine manufacturing related to environmental compliance and waste disposal. The streamlined nature of the synthesis, particularly the potential for one-pot conversion of the intermediate, reduces the number of unit operations, thereby lowering capital expenditure requirements for reactor capacity and shortening the overall production lead time.
- Cost Reduction in Manufacturing: The removal of expensive and toxic organotin reagents eliminates the need for complex heavy metal scavenging steps and specialized disposal protocols, directly lowering the cost of goods sold. Additionally, the use of earth-abundant metals like aluminum or cobalt in the catalyst system, or even metal-free organocatalysts in some embodiments, avoids the price volatility associated with precious metals like platinum or palladium, ensuring more stable long-term pricing for the final API intermediate.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as chloroacetaldehyde and trimethylamine, the process ensures a robust supply chain that is less susceptible to geopolitical constraints or single-source bottlenecks. The flexibility to operate at ambient pressure in many embodiments further enhances reliability by allowing the use of standard glass-lined or stainless steel reactors without the need for high-pressure autoclaves, facilitating easier technology transfer across different manufacturing sites globally.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that can be safely managed in large-scale batches. The reduction in hazardous waste generation, specifically the absence of tin sludge, aligns with modern green chemistry principles and simplifies regulatory filings for nutraceutical applications, ensuring uninterrupted market access for products destined for sensitive food and feed sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the feasibility and optimization of this catalytic pathway, derived directly from the experimental data and embodiments provided in the patent documentation. These insights are intended to assist R&D and process engineering teams in evaluating the suitability of this technology for their specific production requirements and quality targets.
Q: How does this process eliminate toxic organotin reagents?
A: Unlike previous methods requiring n-Bu3SnH for dechlorination, this patent utilizes a direct [2+2] cycloaddition of mono-haloacetaldehydes, bypassing the need for toxic tin-based reduction steps entirely.
Q: What catalysts are suitable for industrial scale-up?
A: The patent highlights cinchona alkaloid derivatives (like TMS-quinine) combined with lithium salts, as well as cobalt and aluminum bifunctional complexes, which offer high enantioselectivity and potential for recycling.
Q: Can the ring-opening step be performed in a single batch?
A: Yes, surprisingly, the patent demonstrates that simultaneous addition of NaOH and TMA allows for a one-pot conversion of the beta-lactone to L-carnitine, suppressing side reactions like hydroxycrotonic acid formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that deliver both economic and environmental value to our partners. Our CDMO division possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of L-carnitine produced via this advanced catalytic method meets the highest global standards for pharmaceutical and nutraceutical applications.
We invite you to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain to achieve a Customized Cost-Saving Analysis. By leveraging our expertise in asymmetric catalysis and process intensification, we can help you secure a stable supply of high-quality intermediates while optimizing your overall manufacturing footprint. Please contact us to request specific COA data and route feasibility assessments tailored to your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
