Scalable Manufacturing of Bifunctional Monomers via Solid-Acid Catalysis for Global Markets
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing complex functional monomers, and patent CN101497569A presents a significant breakthrough in this domain. This patent discloses a novel method for synthesizing allyl alcohol polyoxyethylene ether methacrylate, a versatile bifunctional monomer that serves as a critical building block in advanced material science. The molecule is unique because it possesses two distinct polymerizable double bonds: an allyl group and a methacrylate group, connected by a polyoxyethylene chain. This structural duality allows chemists to fine-tune the hydrophilic-lipophilic balance (HLB) and reactivity of the resulting polymers, making it indispensable for applications ranging from silicone oil modification to high-performance coatings and optical lens manufacturing. The innovation lies not just in the product itself, but in the sophisticated catalytic system employed to create it, moving away from harsh traditional methods toward a greener, solid-acid catalyzed process.
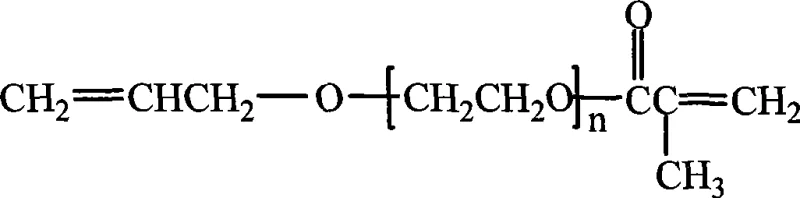
Traditionally, the esterification reactions required to produce such complex ethers have relied heavily on homogeneous liquid acid catalysts, such as concentrated sulfuric acid or p-toluenesulfonic acid. While these conventional catalysts are effective at driving the reaction equilibrium, they introduce severe downstream processing challenges that impact both operational expenditure and equipment longevity. The highly corrosive nature of liquid mineral acids necessitates the use of expensive corrosion-resistant reactors, often lined with glass or specialized alloys, significantly increasing capital expenditure for manufacturers. Furthermore, the post-reaction workup is notoriously difficult; the acid must be neutralized with bases, generating large volumes of saline wastewater that require costly treatment before disposal. This neutralization step also introduces inorganic salt impurities into the organic phase, which can be difficult to remove completely and may adversely affect the clarity and stability of the final polymer products, particularly in sensitive applications like optical materials or electronic coatings.
In stark contrast, the novel approach detailed in the patent utilizes activated carbon-supported phosphotungstic acid as a heterogeneous catalyst, fundamentally altering the process economics and environmental footprint. This solid acid catalyst combines the high acidity and catalytic activity of phosphotungstic acid with the mechanical stability and ease of separation provided by the activated carbon support. Because the catalyst is a solid, it does not dissolve in the reaction mixture, eliminating the need for neutralization and the subsequent generation of salt waste. The reaction can be conducted in standard stainless steel equipment without fear of corrosion, drastically reducing maintenance costs and extending the lifespan of production assets. Additionally, the patent highlights the ability to recover and reuse the catalyst simply by filtration after the reaction is complete, which not only reduces the consumption of raw materials but also ensures a consistent catalytic performance batch after batch, leading to a more stable and predictable manufacturing process.
Mechanistic Insights into Heterogeneous Esterification and Polymerization Control
The core of this synthesis lies in the precise control of the esterification mechanism while simultaneously preventing unwanted side reactions. The reaction involves the condensation of allyl alcohol polyoxyethylene ether with methacrylic acid, a process that is thermodynamically limited by the production of water. To drive the equilibrium toward the desired ester product, the process employs an azeotropic distillation technique using a water-carrying agent, typically a mixture of toluene and cyclohexane. As the reaction proceeds at temperatures between 100°C and 120°C, the water formed is continuously removed from the system as an azeotrope with the solvent, shifting the equilibrium according to Le Chatelier's principle. The activated carbon-supported phosphotungstic acid acts as a proton donor, activating the carbonyl group of the methacrylic acid to facilitate nucleophilic attack by the hydroxyl group of the polyether, all while remaining physically distinct from the liquid phase.
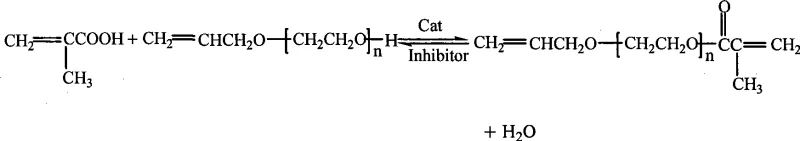
A critical aspect of the mechanistic design is the management of the molecule's high reactivity. The target product contains two vinyl groups that are susceptible to radical polymerization, especially under the thermal stress of the esterification conditions. If left unchecked, this would lead to the formation of oligomers or crosslinked gels, ruining the batch and fouling the reactor. To counteract this, the patent specifies the use of potent polymerization inhibitors such as phenothiazine or hydroquinone, added in precise amounts ranging from 0.025% to 0.2% of the total reactant weight. These inhibitors function by scavenging free radicals that might initiate premature polymerization, effectively stabilizing the monomer during the synthesis. Furthermore, the continuous introduction of nitrogen gas serves a dual purpose: it creates an inert atmosphere that excludes oxygen (a potential initiator) and assists in stripping out the water azeotrope, thereby enhancing the reaction rate and ensuring the integrity of the double bonds is preserved until the final product is isolated.
How to Synthesize Allyl Alcohol Polyoxyethylene Ether Methacrylate Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and physical parameters outlined in the patent to achieve the reported high esterification rates of over 98%. The process begins with charging the reactor with the polyether and methacrylic acid in a molar ratio of roughly 1:1 to 1:1.5, along with the solid catalyst and inhibitor. The choice of the water-carrying solvent system is crucial, with a toluene-to-cyclohexane volume ratio adjustable between 20:80 and 80:20 to optimize the boiling point and water removal efficiency. Once the mixture is heated to the optimal range of 100-120°C under a steady stream of nitrogen, the reaction typically proceeds to completion within 5 to 7 hours. The endpoint is determined practically by monitoring the water separator; when no more water is collected, the reaction is deemed complete. Following the reaction, the workup is remarkably straightforward compared to traditional methods, involving simple filtration to remove the catalyst, a polishing step with diatomite to ensure clarity, and finally vacuum distillation to remove the solvent and isolate the pure monomer.
- Charge the reactor with allyl alcohol polyoxyethylene ether, methacrylic acid, activated carbon-supported phosphotungstic acid catalyst, and a polymerization inhibitor such as hydroquinone.
- Add a water-carrying agent composed of toluene and cyclohexane, then heat the mixture to 100-120°C while continuously introducing nitrogen gas to facilitate azeotropic distillation.
- Upon completion of the esterification reaction, filter to recover the solid catalyst, treat the filtrate with diatomite, and perform vacuum distillation to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this solid-acid catalyzed process represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of corrosive liquid acids translates directly into substantial cost savings by extending the service life of reactor vessels and piping, thereby reducing the frequency of expensive equipment replacements and unplanned downtime for maintenance. Moreover, the simplified purification process, which removes the need for washing, neutralization, and drying steps associated with liquid acid catalysts, significantly shortens the overall production cycle time. This efficiency gain allows manufacturing facilities to increase their throughput capacity without additional capital investment, ensuring a more robust and responsive supply chain capable of meeting fluctuating market demands for high-performance polymer additives and crosslinkers.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the reusability of the heterogeneous catalyst and the drastic simplification of the downstream processing. Since the activated carbon-supported phosphotungstic acid can be filtered and reused multiple times, the recurring cost of catalyst consumption is minimized. Additionally, avoiding the generation of saline wastewater eliminates the regulatory and operational costs associated with effluent treatment, while the absence of inorganic salt impurities reduces the energy and time required for final product purification, leading to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply continuity. The mild reaction conditions and the stability of the solid catalyst reduce the risk of batch failures due to runaway reactions or equipment corrosion leaks. Furthermore, the raw materials required, such as methacrylic acid and allyl alcohol polyoxyethylene ether, are commodity chemicals with well-established global supply chains. By utilizing a process that is less sensitive to trace moisture or minor variations in feedstock quality compared to sensitive organometallic catalysts, manufacturers can maintain consistent production schedules even when facing minor supply chain disruptions.
- Scalability and Environmental Compliance: From a sustainability and scaling perspective, this method aligns perfectly with modern green chemistry principles. The process generates minimal waste, as the primary byproduct is water which is cleanly separated, and the catalyst is non-leaching and recyclable. This reduces the environmental burden on the facility and simplifies compliance with increasingly stringent environmental regulations regarding hazardous waste disposal. The scalability is further enhanced by the fact that the reaction kinetics are not limited by mass transfer issues common in biphasic liquid systems, allowing for seamless translation from pilot plant to multi-ton commercial production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized monomer. Understanding these details is crucial for R&D teams evaluating this material for new formulations and for procurement specialists assessing supplier capabilities. The answers are derived directly from the technical specifications and experimental data provided in the underlying patent literature, ensuring accuracy and relevance for industrial applications.
Q: What are the advantages of using activated carbon-supported phosphotungstic acid over traditional catalysts?
A: Unlike traditional liquid acids like sulfuric acid which cause severe equipment corrosion and require complex neutralization steps, this solid acid catalyst is non-corrosive, easily separated by simple filtration, and can be recovered and reused multiple times without significant loss of activity.
Q: Why is a polymerization inhibitor necessary during the esterification process?
A: Since the target molecule contains two polymerizable double bonds (allyl and methacrylate groups), there is a risk of premature polymerization at elevated temperatures. Inhibitors like phenothiazine or hydroquinone prevent this side reaction, ensuring high monomer purity and yield.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process operates under mild conditions (100-120°C) and utilizes standard esterification equipment with azeotropic water removal. The simplicity of the workup procedure, involving only filtration and distillation, makes it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Alcohol Polyoxyethylene Ether Methacrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity bifunctional monomers play in the development of next-generation polymers and coatings. Our technical team has extensively analyzed advanced synthesis routes like the one described in patent CN101497569A to ensure we offer products that meet the most rigorous performance standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver consistent quality. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that verify every batch for acid value, color, and monomer content, ensuring that the material you receive is perfectly suited for sensitive applications like optical lenses or electronic encapsulants.
We invite you to collaborate with us to optimize your supply chain for these essential chemical intermediates. Whether you require a Customized Cost-Saving Analysis for your specific volume requirements or need to discuss the technical feasibility of integrating this monomer into your existing formulations, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can add value to your business.
