Advanced Nano-Catalytic Synthesis of Rufinamide for Scalable API Manufacturing
Advanced Nano-Catalytic Synthesis of Rufinamide for Scalable API Manufacturing
The pharmaceutical landscape for antiepileptic treatments continues to evolve, driven by the demand for more efficient and safer manufacturing processes for critical active pharmaceutical ingredients (APIs). Patent CN103539750A introduces a transformative synthetic methodology for Rufinamide, a vital medication used in the adjunctive treatment of Lennox-Gastaut syndrome. This intellectual property outlines a robust three-step sequence that fundamentally reengineers the production workflow, shifting away from hazardous thermal conditions toward a streamlined, room-temperature protocol. By leveraging phase-transfer catalysis and novel nano-materials, this approach addresses long-standing bottlenecks in yield, safety, and operational complexity. For global supply chain stakeholders, this represents a pivotal shift toward more sustainable and cost-effective API production capabilities.
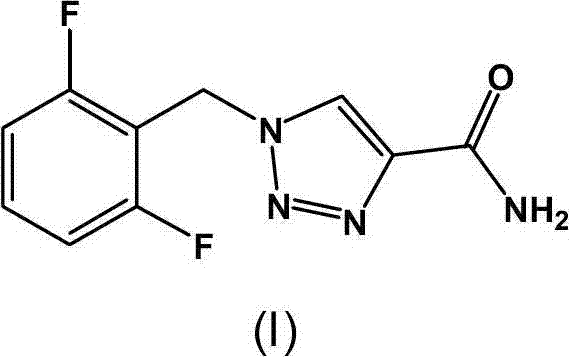
The core innovation lies in the strategic replacement of traditional homogeneous copper catalysts with a specialized nano-cuprous oxide system. This modification not only accelerates the critical ring-closing step but also simplifies the downstream purification landscape. As a leading entity in fine chemical synthesis, understanding the nuances of such patented routes is essential for maintaining a competitive edge in the reliable pharmaceutical intermediates supplier market. The following analysis dissects the technical merits of this process, highlighting its potential for cost reduction in API manufacturing and its implications for large-scale commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rufinamide has been plagued by significant operational hazards and inefficiencies that complicate large-scale production. Traditional protocols typically rely on the reaction of 2,6-difluorobenzyl halides with sodium azide in aqueous media, a process that necessitates elevated temperatures to drive the substitution to completion. This thermal requirement introduces substantial safety risks, given the potentially explosive nature of organic azides when subjected to heat and concentration. Furthermore, the subsequent isolation of the azide intermediate often involves cumbersome extraction procedures and vacuum distillation, which not only consume excessive energy but also expose personnel to hazardous materials. In the critical triazole formation step, conventional methods utilizing copper sulfate and sodium ascorbate frequently suffer from sluggish kinetics, often requiring overnight reaction periods to achieve acceptable conversion. These prolonged timelines create bottlenecks in reactor utilization, effectively capping production capacity and inflating the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology disclosed in CN103539750A offers a sophisticated solution that mitigates these risks through intelligent process design. The new route employs a biphasic system utilizing water and acetonitrile, facilitated by tetrabutylammonium chloride as a phase-transfer catalyst. This setup enables the azidation of 2,6-difluorobenzyl halides to proceed smoothly at ambient temperatures ranging from 15°C to 30°C, completely eliminating the need for external heating. The elimination of thermal stress on the azide intermediate dramatically enhances process safety profiles. Moreover, the process is designed to be telescoped; the organic layer containing the azide can be directly transferred to the next reaction vessel without isolation. This "one-pot" philosophy removes the need for distillation and extensive workup, thereby reducing solvent consumption and waste generation. The transition to a nano-cuprous oxide catalyst further revolutionizes the cycloaddition step, slashing reaction times from over 12 hours to merely 2 to 5 hours while maintaining high catalytic activity.
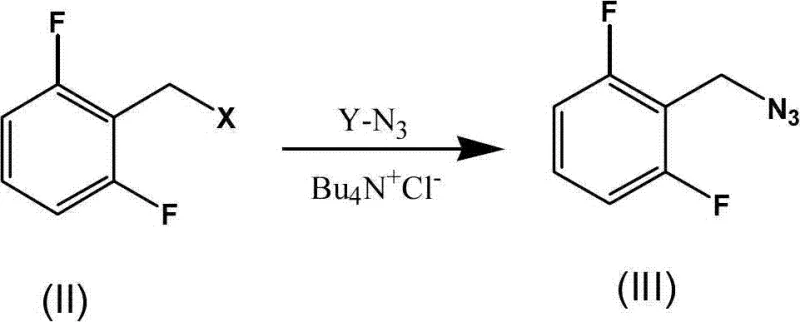
Mechanistic Insights into Nano-Cu2O Catalyzed Click Chemistry
The heart of this synthetic advancement is the deployment of nano-cuprous oxide (Cu2O) as a heterogeneous catalyst for the Huisgen 1,3-dipolar cycloaddition. Unlike traditional soluble copper salts that require stoichiometric amounts of reducing agents like sodium ascorbate to maintain the active Cu(I) oxidation state, the pre-formed nano-Cu2O particles provide a stable and highly active surface for the reaction. The preparation of this catalyst involves the reduction of copper acetate by sodium borohydride in the presence of polyvinylpyrrolidone (PVP), which acts as a capping agent to prevent nanoparticle aggregation. This results in a black solid with a high surface-area-to-volume ratio, facilitating rapid interaction between the organic azide and methyl propiolate. The mechanism likely proceeds through the coordination of the alkyne to the copper surface, activating it towards nucleophilic attack by the azide, followed by ring closure to form the 1,4-disubstituted 1,2,3-triazole scaffold. This heterogeneous nature simplifies catalyst removal, potentially allowing for filtration rather than complex chromatographic purification.
From an impurity control perspective, this catalytic system offers distinct advantages over homogeneous alternatives. The use of nano-Cu2O minimizes the leaching of copper ions into the final product, a critical quality attribute for pharmaceutical APIs where heavy metal residues are strictly regulated. The high selectivity of the nano-catalyst ensures that the formation of the desired 1,4-isomer is favored, suppressing the generation of regioisomeric byproducts that could complicate downstream crystallization. Additionally, the mild reaction conditions prevent the decomposition of the sensitive azide functionality, which is a common source of impurity formation in thermal processes. By maintaining the reaction at room temperature, the kinetic energy available for side reactions is minimized, leading to a cleaner crude profile. This inherent purity reduces the burden on the final purification steps, allowing for simpler recrystallization protocols to achieve high-purity Rufinamide suitable for clinical use.
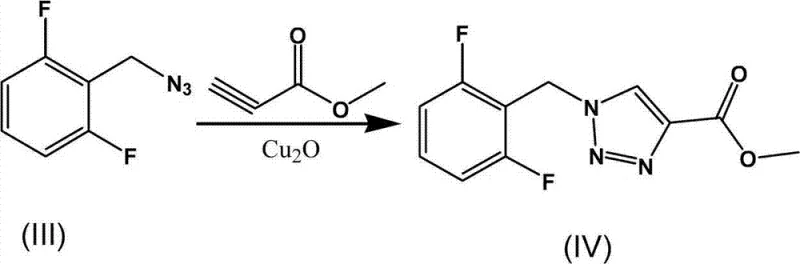
How to Synthesize Rufinamide Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the nano-catalytic system. The process begins with the careful preparation of the azide intermediate under phase-transfer conditions, ensuring complete conversion before introducing the alkyne. The subsequent addition of methyl propiolate and the nano-copper catalyst must be managed under an inert atmosphere to prevent oxidation of the catalyst surface. Monitoring via thin-layer chromatography (TLC) is recommended to determine the exact endpoint, typically achieved within 3 hours under optimal conditions. The final ammonolysis step converts the ester to the primary amide, completing the API structure. For detailed operational parameters and specific molar ratios, refer to the standardized guide below.
- Perform phase-transfer azidation of 2,6-difluorobenzyl halide using sodium azide and tetrabutylammonium chloride in a water-acetonitrile system at room temperature.
- React the resulting azide intermediate with methyl propiolate using nano-cuprous oxide (Cu2O) as a heterogeneous catalyst to form the triazole ester.
- Convert the triazole ester to Rufinamide via ammonolysis using ammonium hydroxide, followed by crystallization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend beyond simple chemistry. The shift from a multi-day batch process to a streamlined, room-temperature workflow fundamentally alters the economics of production. By removing the need for heating during the azidation step and significantly shortening the cycloaddition timeline, facility throughput can be increased without additional capital expenditure on reactor volume. The ability to telescope steps without isolating the energetic azide intermediate reduces the number of unit operations, which directly correlates to lower labor costs and reduced equipment occupancy time. Furthermore, the simplified workup procedures minimize solvent usage and waste disposal costs, aligning with modern green chemistry mandates and reducing the environmental compliance burden. These factors collectively contribute to a more resilient and cost-efficient supply chain for this critical antiepileptic medication.
- Cost Reduction in Manufacturing: The elimination of energy-intensive heating and vacuum distillation steps results in substantial utility savings. Additionally, the high catalytic efficiency of the nano-Cu2O system reduces the quantity of catalyst required and shortens the reaction cycle, allowing for more batches to be produced per week. The avoidance of complex purification techniques for the intermediate further lowers the cost of goods by reducing solvent and consumable expenses.
- Enhanced Supply Chain Reliability: The robustness of the room-temperature process reduces the risk of batch failures due to thermal runaway or exothermic events. The use of readily available starting materials and the stability of the nano-catalyst ensure consistent supply continuity. By simplifying the synthesis into fewer, more reliable steps, manufacturers can better predict lead times and maintain steady inventory levels for downstream formulation partners.
- Scalability and Environmental Compliance: The heterogeneous nature of the nano-catalyst facilitates easier scale-up, as heat transfer limitations associated with exothermic click reactions are mitigated by the mild conditions. The reduction in solvent volume and the removal of hazardous distillation steps significantly lower the facility's environmental footprint. This alignment with sustainability goals makes the process attractive for regulatory approval and long-term manufacturing licenses in strict jurisdictions.
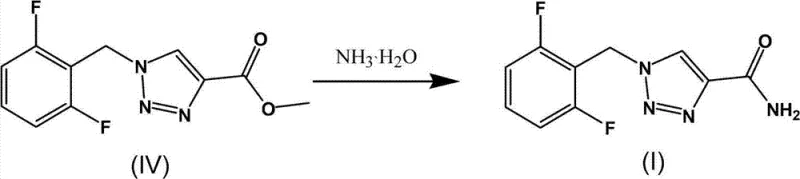
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or contract manufacturing agreements.
Q: What are the safety advantages of the new Rufinamide synthesis route?
A: The novel process eliminates the need for heating during the azide formation step, conducting the reaction at room temperature (15-30°C). This significantly reduces the thermal risk associated with organic azides and removes the need for complex distillation steps.
Q: How does the nano-cuprous oxide catalyst improve efficiency?
A: Compared to traditional copper sulfate/ascorbate systems which require overnight reaction times, the nano-Cu2O catalyst accelerates the cycloaddition step to just 2-5 hours at room temperature, drastically improving catalytic efficiency and throughput.
Q: Is intermediate isolation required between the azidation and click chemistry steps?
A: No, the process allows for a telescoped operation where the acetonitrile layer containing the azide intermediate can be directly used in the subsequent triazole formation, avoiding energy-intensive extraction and vacuum distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rufinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing pathways for life-saving medications like Rufinamide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this nano-catalytic process are fully realized in an industrial setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international pharmacopeial standards. Our commitment to process excellence allows us to deliver high-purity Rufinamide with consistent quality, supporting our partners in meeting their global regulatory obligations.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced synthetic route, we can offer competitive pricing structures that reflect the reduced operational costs of the new methodology. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and economic efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
