Advanced Metconazole Synthesis: Overcoming Toxicity and Yield Barriers in Agrochemical Manufacturing
The global demand for high-efficacy triazole fungicides continues to surge, driven by the need for robust crop protection solutions against resistant fungal strains. At the forefront of this chemical evolution is Metconazole, a broad-spectrum systemic fungicide known for its protective and therapeutic capabilities against rusts and mildews in cereal crops. However, the historical manufacturing landscape for this critical agrochemical active has been marred by significant environmental and safety challenges. A pivotal breakthrough in this domain is documented in patent CN103664561A, which discloses a novel preparation method that fundamentally restructures the synthetic pathway. This innovation shifts the paradigm from hazardous cyanide-based chemistries to a more sustainable condensation and hydrogenation strategy. For R&D Directors and Supply Chain Heads, understanding this technological pivot is not merely an academic exercise but a strategic imperative for securing long-term supply continuity and regulatory compliance. The following analysis dissects this proprietary methodology, highlighting its potential to redefine cost structures and purity standards in the agrochemical intermediate sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Metconazole intermediates relied heavily on pathways that are increasingly untenable in modern regulatory environments. One prominent prior art route, illustrated below, initiates with dimethyl adipate and necessitates multiple methylation steps using monobromomethane, a compound notorious for its high toxicity and volatility.
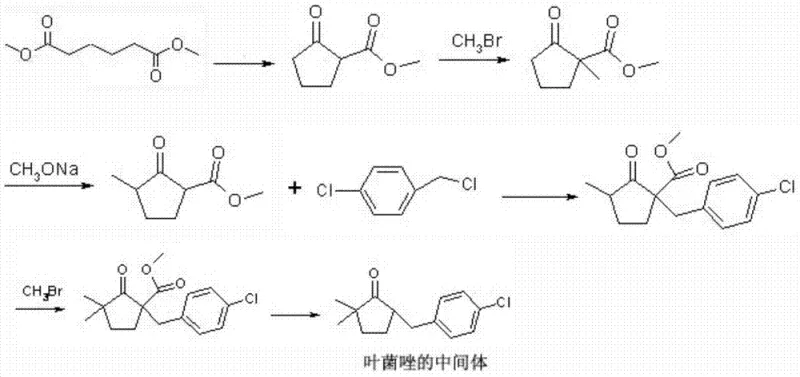
Furthermore, alternative legacy methods utilize isopropyl cyanide and sodium cyanide as starting materials, introducing severe safety liabilities regarding worker exposure and waste disposal. These conventional approaches often suffer from low overall yields due to difficult hydrolysis and decarboxylation steps where steric hindrance impedes reaction efficiency. The accumulation of impurities in these multi-step sequences necessitates energy-intensive purification processes, thereby inflating the cost of goods sold and complicating the supply chain with hazardous material logistics. For procurement managers, reliance on suppliers utilizing these outdated technologies poses a significant risk of production stoppages due to tightening environmental regulations.
The Novel Approach
In stark contrast, the methodology outlined in CN103664561A introduces a streamlined sequence that bypasses these critical bottlenecks. By selecting 2-methoxycarbonyl cyclopentanone and p-chlorobenzaldehyde as primary feedstocks, the new route leverages a Claisen-Schmidt type condensation followed by a controlled decarboxylation. This strategic selection of raw materials ensures that the carbon skeleton is constructed more efficiently, minimizing the number of unit operations required to reach the key ketone intermediate. The elimination of highly toxic cyanide reagents not only enhances the safety profile of the manufacturing facility but also drastically simplifies the wastewater treatment protocols. This approach represents a significant leap forward in cost reduction in agrochemical manufacturing, as it aligns synthetic efficiency with environmental stewardship, creating a resilient production model capable of withstanding regulatory scrutiny.
Mechanistic Insights into Condensation and Catalytic Hydrogenation
The core of this technological advancement lies in the precise orchestration of reaction conditions that maximize selectivity while minimizing byproduct formation. The initial phase involves the reaction of 2-methoxycarbonyl cyclopentanone with p-chlorobenzaldehyde in a methanol solution containing sodium methylate. This base-catalyzed condensation proceeds at moderate temperatures, typically between 20°C and 35°C, to form an unsaturated beta-keto ester intermediate. The subsequent decarboxylation step, conducted in an aqueous hydrobromic acid solution at elevated temperatures, cleanly removes the ester functionality to yield the alpha,beta-unsaturated ketone. This sequence is critical because it establishes the necessary carbon framework without introducing extraneous functional groups that would require later removal.
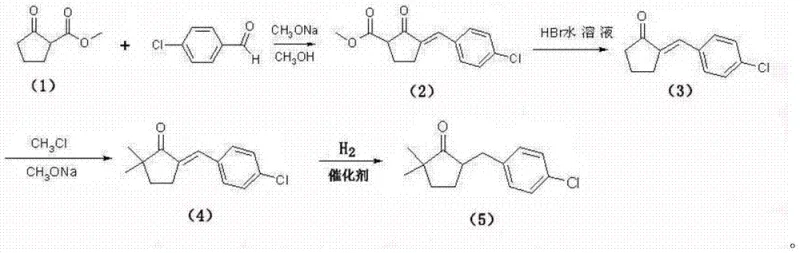
Following the formation of the unsaturated ketone, the process employs a methylation step using methyl chloride under basic conditions to introduce the gem-dimethyl group, followed by a catalytic hydrogenation. The hydrogenation step is particularly noteworthy for its use of a palladium-carbon catalyst under controlled pressure, typically around 1.0MPa to 1.2MPa. This reduction selectively saturates the carbon-carbon double bond without affecting the carbonyl group or the chloro-substituent on the aromatic ring. Such chemoselectivity is paramount for maintaining high purity levels, as over-reduction or dehalogenation would generate difficult-to-separate impurities. For R&D teams, this mechanistic clarity offers a robust platform for process optimization, ensuring that the high-purity agrochemical intermediate specifications are consistently met across different batch scales.
How to Synthesize Metconazole Intermediate Efficiently
Implementing this novel synthesis route requires strict adherence to the optimized parameters defined in the patent literature to ensure reproducibility and safety. The process begins with the preparation of the condensation product, followed by acid-mediated decarboxylation and subsequent methylation. The final transformation into the saturated ketone intermediate is achieved through catalytic hydrogenation, which serves as the gateway to the final fungicide assembly. Each step is designed to be telescoped where possible, reducing solvent usage and handling time. While the general workflow is straightforward, the specific control of temperature, pressure, and reagent stoichiometry is vital for achieving the reported yields and purity profiles. Detailed standard operating procedures for each unit operation are essential for technology transfer.
- Condense 2-methoxycarbonyl cyclopentanone with p-chlorobenzaldehyde using sodium methylate in methanol to form the unsaturated intermediate.
- Perform decarboxylation in aqueous HBr solution followed by methylation with methyl chloride under basic conditions.
- Execute catalytic hydrogenation using Pd/C to saturate the double bond, yielding the key ketone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the shift to this novel synthetic pathway offers compelling economic and logistical advantages. The primary value driver is the substantial simplification of the supply chain regarding raw material sourcing. By eliminating the dependency on tightly controlled and hazardous reagents like sodium cyanide and methyl bromide, manufacturers can reduce the administrative burden and costs associated with hazardous material storage and transport. This inherently leads to a more agile supply chain capable of responding quickly to market demands without the friction of special permits and safety audits. Furthermore, the improved yield profile means that less raw material is wasted per kilogram of finished product, directly enhancing material efficiency and reducing the variable cost of production.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents significantly lowers the direct material costs associated with the synthesis. Additionally, the simplified workup procedures, which avoid complex hydrolysis steps prone to failure, reduce the consumption of solvents and energy utilities. The higher overall yield reported in the patent data implies that less feedstock is required to produce the same amount of active ingredient, creating a multiplier effect on cost savings. These factors combine to offer a more competitive pricing structure for the final agrochemical intermediate, allowing downstream formulators to maintain margins even in volatile market conditions.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals like p-chlorobenzaldehyde and cyclopentanone derivatives ensures a stable supply base that is less susceptible to the disruptions often seen with specialty toxic reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic metallurgy, further reduces the risk of unplanned maintenance downtime. For supply chain heads, this translates to reducing lead time for high-purity agrochemical intermediates and ensuring consistent delivery schedules to formulation plants globally. The ability to scale this process from pilot to commercial tonnage without fundamental changes to the chemistry provides a secure foundation for long-term procurement contracts.
- Scalability and Environmental Compliance: The reduction in three-waste discharge (wastewater, waste gas, and waste solid) is a critical advantage in an era of stringent environmental enforcement. The process generates fewer hazardous byproducts, simplifying effluent treatment and lowering the cost of environmental compliance. This green chemistry profile not only mitigates regulatory risk but also enhances the corporate sustainability credentials of the supply chain. The commercial scale-up of complex agrochemical intermediates is facilitated by the use of standard unit operations such as filtration, distillation, and hydrogenation, which are well-understood and easily replicated in multipurpose chemical facilities worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They serve to clarify the operational benefits and safety improvements inherent in this new methodology. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks or for procurement specialists assessing supplier capabilities.
Q: How does this new method improve safety compared to traditional cyanide routes?
A: The novel route eliminates the use of highly toxic sodium cyanide and isopropyl cyanide found in prior art, significantly reducing hazardous waste handling requirements and improving operator safety profiles during industrial scale-up.
Q: What is the expected purity of the Metconazole produced via this pathway?
A: According to patent data CN103664561A, the final product content can reach 95% or higher, with a total yield exceeding 65%, ensuring high-quality standards suitable for regulatory compliance in agrochemical formulations.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like p-chlorobenzaldehyde and avoids complex purification steps, making it highly adaptable for commercial scale-up with reduced three-waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metconazole Supplier
The transition to safer and more efficient synthetic routes for critical agrochemicals like Metconazole requires a partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the intersection of innovation and reliability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of intermediate meets the exacting standards required for global pesticide registration. We understand that consistency is key in the agrochemical industry, and our processes are designed to deliver that consistency batch after batch.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our advanced synthesis capabilities, you can secure a competitive edge in the market while adhering to the highest standards of safety and environmental responsibility.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
