Revolutionizing 2-Acylindole Production via Visible Light Photocatalysis for Global Supply Chains
Revolutionizing 2-Acylindole Production via Visible Light Photocatalysis for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients. A groundbreaking development detailed in Chinese Patent CN111217736A introduces a highly efficient method for synthesizing 2-acylindole compounds under mild photocatalytic conditions. This technology leverages visible light irradiation to drive decarboxylative addition and cyclization reactions, bypassing the severe limitations of traditional thermal methods. By utilizing readily available o-alkenyl aryl isonitriles and alpha-keto carboxylic acids, this process achieves high atom economy and operational simplicity. The ability to conduct these transformations at room temperature in an air atmosphere represents a paradigm shift in how we approach indole functionalization. For procurement and R&D leaders, this patent signals a move towards greener, more cost-effective manufacturing protocols that do not compromise on molecular complexity.
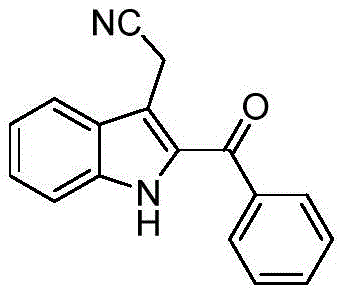
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of an acyl group at the C-2 position of the indole ring has been a formidable challenge in organic synthesis, often necessitating the use of expensive transition metal catalysts such as palladium. Traditional routes typically rely on directed C-H activation strategies which require specific guiding groups or ligands to control regioselectivity, adding significant steps to the synthetic sequence. Furthermore, these conventional methods frequently demand harsh reaction conditions, including high temperatures and the use of excess stoichiometric oxidants to regenerate the active catalytic species. Such rigorous environments not only increase energy consumption but also pose safety risks and complicate waste management protocols in large-scale facilities. The reliance on pre-functionalized substrates further restricts the scope of accessible derivatives, limiting the chemical space available for drug discovery teams. Consequently, the overall cost of goods sold (COGS) for 2-acylindole intermediates produced via these legacy methods remains prohibitively high for many commercial applications.
The Novel Approach
In stark contrast, the novel photocatalytic approach described in the patent data utilizes visible light to generate reactive acyl radicals directly from stable alpha-keto carboxylic acids. This method eliminates the need for harsh oxidants and allows the reaction to proceed under exceptionally mild conditions, specifically at room temperature and in the presence of air. The use of blue LED irradiation provides a clean energy source that drives the redox cycle of the iridium photocatalyst without generating excessive heat. This innovation drastically simplifies the operational setup, removing the necessity for specialized high-pressure reactors or inert gas manifolds. Moreover, the reaction demonstrates excellent functional group tolerance, accommodating various substituents on both the isonitrile and the acid components without significant yield erosion. This flexibility enables the rapid generation of diverse libraries of 2-acylindole derivatives, accelerating the lead optimization phase for pharmaceutical candidates.
Mechanistic Insights into Photoredox-Catalyzed Decarboxylative Cyclization
The core of this transformative chemistry lies in the ability of the excited state photocatalyst to facilitate single-electron transfer (SET) processes under mild conditions. Upon irradiation with 20W blue LEDs, the iridium complex absorbs photons and transitions to a highly reducing excited state capable of interacting with the alpha-keto acid substrate. This interaction triggers a decarboxylation event, releasing carbon dioxide and generating a nucleophilic acyl radical species with high efficiency. The stability of these acyl radicals is crucial, as they must survive long enough to engage in the subsequent addition step without undergoing premature dimerization or decomposition. The reaction medium, typically polar aprotic solvents like DMF or THF, supports the solubility of ionic intermediates and stabilizes the transition states involved in the radical cascade.
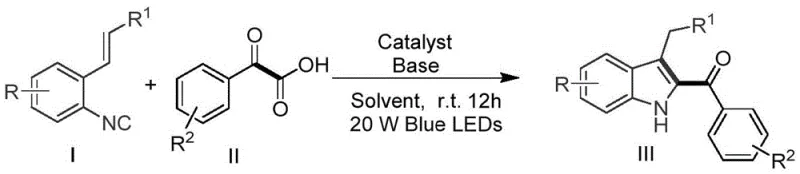
Following the generation of the acyl radical, the mechanism proceeds through a radical addition to the electron-deficient alkene moiety of the o-alkenyl aryl isonitrile. This addition creates a new carbon-carbon bond and generates an intermediate radical centered on the benzylic position. Subsequent intramolecular cyclization onto the isonitrile carbon occurs rapidly, forming the five-membered pyrrole ring characteristic of the indole core. The final aromatization step restores the aromaticity of the system, yielding the stable 2-acylindole product while regenerating the ground state photocatalyst to close the catalytic cycle. Impurity control is inherently managed by the specificity of the radical pathway, which avoids the non-selective background reactions often seen in thermal radical initiators. The use of mild bases such as potassium phosphate further ensures that acid-sensitive functional groups remain intact throughout the transformation.
How to Synthesize 2-Acylindole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the reagents and the intensity of the light source. The patent specifies a molar ratio of 1:2 between the o-alkenyl aryl isonitrile and the alpha-keto acid, ensuring that the radical precursor is in excess to drive the reaction to completion. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations. Operators should verify the wavelength output of their LED arrays to match the absorption profile of the specific iridium catalyst employed. Proper stirring is also essential to ensure uniform light penetration throughout the reaction vessel, preventing localized hot spots or incomplete conversion.
- Dissolve o-alkenyl aryl isonitrile and alpha-keto carboxylic acid in a suitable solvent such as THF or DMF with an Iridium photocatalyst and base.
- Stir the reaction mixture under 20W blue LED irradiation at room temperature in an air atmosphere for 12 hours.
- Monitor reaction progress by TLC, then perform extraction with ethyl acetate, concentration, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere technical novelty. The shift from precious metal catalysis requiring inert atmospheres to a bench-top compatible visible light process fundamentally alters the cost structure of manufacturing these intermediates. By removing the dependency on complex engineering controls for oxygen exclusion, capital expenditure for reactor infrastructure is significantly reduced. Additionally, the use of commercially available and stable starting materials mitigates supply risk, as these commodities are sourced from robust global supply chains rather than niche custom synthesis providers. The simplified workup procedure, involving standard extraction and chromatography, reduces solvent consumption and processing time, leading to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the associated ligand systems results in direct material cost savings for every batch produced. Furthermore, the ability to run reactions at room temperature removes the energy costs associated with heating large reactor volumes over extended periods. The low catalyst loading required for the iridium complex means that metal recovery costs are minimized, and the overall catalyst expense per kilogram of product is drastically lowered. Process efficiency is enhanced by the shorter reaction times compared to multi-step thermal sequences, allowing for higher throughput in existing facilities without additional capital investment.
- Enhanced Supply Chain Reliability: Sourcing alpha-keto acids and isonitriles is far more straightforward than procuring specialized organometallic reagents, ensuring consistent availability even during market fluctuations. The robustness of the reaction in air means that production schedules are less vulnerable to delays caused by gas supply issues or equipment failures related to inertion systems. This reliability translates to more predictable lead times for downstream customers who depend on these intermediates for their own API synthesis. Diversifying the supplier base becomes easier as the barrier to entry for manufacturing this chemistry is lower due to the simplified operational requirements.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but advancements in LED technology and flow chemistry make this route highly amenable to ton-scale production. The absence of toxic oxidants and heavy metal waste streams aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. Waste treatment costs are reduced because the primary byproduct is carbon dioxide, which vents safely, and the solvent systems used are standard and recyclable. This green chemistry profile enhances the marketability of the final product to environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance. Understanding these nuances is critical for process chemists evaluating the feasibility of technology transfer. We encourage technical teams to review these points before initiating pilot studies.
Q: Does this photocatalytic method require inert gas protection?
A: No, unlike many traditional transition metal-catalyzed reactions that require strict nitrogen or argon atmospheres, this patented process operates efficiently in an air atmosphere, significantly simplifying operational requirements.
Q: What are the advantages of using alpha-keto acids over traditional acylating agents?
A: Alpha-keto acids serve as stable precursors that generate acyl radicals under visible light, avoiding the need for harsh oxidants or pre-functionalized substrates often required in palladium-catalyzed C-H activation methods.
Q: Is the Iridium catalyst recoverable for large-scale production?
A: While the patent utilizes homogeneous Iridium complexes, the low catalyst loading (molar ratio of 50:1 relative to substrate) minimizes cost impact, and standard industrial scavenging techniques can be applied for metal removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this photocatalytic technology to reshape the landscape of heterocyclic intermediate production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications for complex pharmaceutical intermediates. We are committed to leveraging innovations like CN111217736A to deliver superior value to our global clientele.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific project needs. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments to support your next development milestone.
