Advanced Synthesis of Bipolar Aryl Acridine Phosphine Spiro Compounds for High-Efficiency OLEDs
Advanced Synthesis of Bipolar Aryl Acridine Phosphine Spiro Compounds for High-Efficiency OLEDs
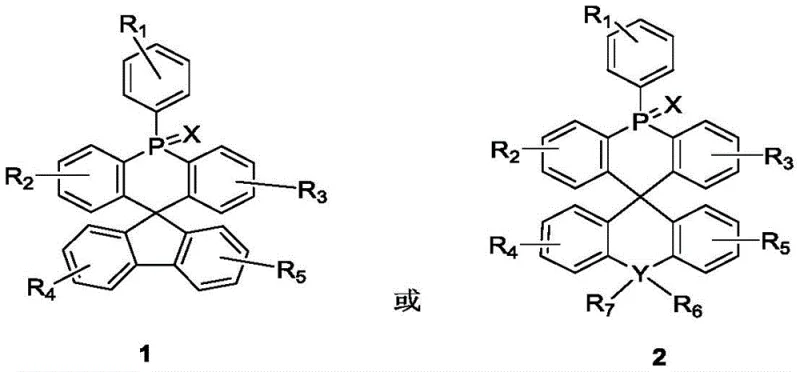
The rapid evolution of the organic light-emitting diode (OLED) industry demands host materials that can simultaneously offer high thermal stability, efficient charge transport, and suppressed concentration quenching. Patent CN110845537A introduces a groundbreaking class of aryl acridine phosphine spiro bipolar compounds that address these critical challenges through a novel molecular architecture. By connecting a 5-arylacridine phosphine unit with fluorene or related derivatives via an sp3 hybridized carbon atom, this technology creates a rigid, three-dimensional structure that inherently prevents pi-stacking. This structural innovation is pivotal for next-generation display and lighting applications, offering a reliable OLED material supplier pathway for manufacturers seeking to overcome the efficiency roll-off common in traditional host systems.
The development of high-performance phosphorescent OLEDs (PhOLEDs) has historically been hindered by the limitations of conventional host materials. Traditional unipolar hosts, such as CBP or mCP, often suffer from low glass transition temperatures or mismatched triplet energy levels that lead to energy back-transfer and reduced device longevity. Furthermore, the planar nature of many existing aromatic hosts facilitates intermolecular aggregation at high doping concentrations, causing severe concentration quenching and triplet-triplet annihilation. These issues necessitate complex device engineering to manage exciton distribution, often increasing manufacturing costs and reducing yield. The search for a robust solution requires a fundamental shift towards bipolar materials that can balance hole and electron transport while maintaining a stable amorphous morphology under operational stress.
In contrast to these legacy systems, the novel approach detailed in the patent utilizes a spiro-configured backbone to physically separate conjugated systems, thereby inhibiting aggregation without sacrificing charge mobility. The synthesis strategy employs a versatile modular design where the phosphorus center can be functionalized with oxygen, sulfur, or selenium. This tunability allows for precise adjustment of the HOMO and LUMO energy levels to match various guest emitters, from blue to red phosphors. The resulting compounds exhibit superior thermal decomposition temperatures and maintain high fluorescence quantum efficiency, making them ideal candidates for commercial scale-up of complex organic semiconductors in high-end display panels.
Mechanistic Insights into Acid-Catalyzed Spiro Cyclization and Chalcogen Oxidation
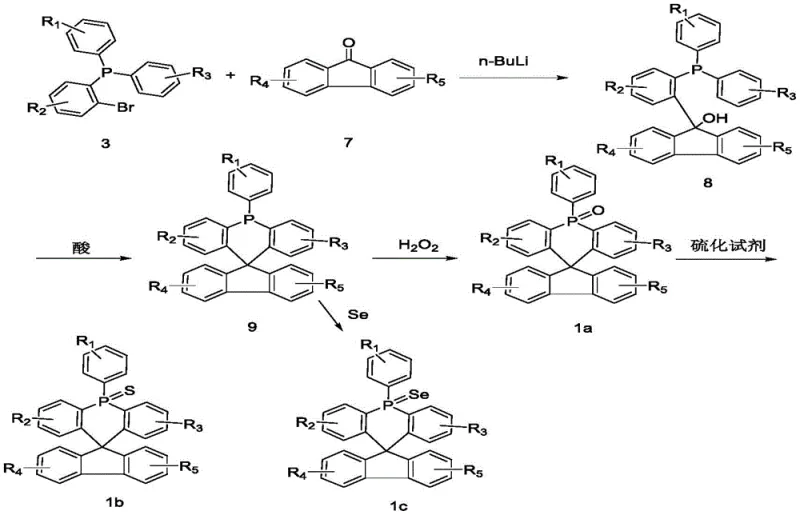
The core of this synthetic methodology lies in the efficient construction of the spiro junction via an acid-catalyzed intramolecular Friedel-Crafts-type cyclization. The process begins with the generation of a nucleophilic organolithium species from a 2-bromotriarylphosphine precursor, which attacks a ketone substrate like fluorenone to form a tertiary alcohol intermediate. Upon exposure to strong protic acids such as concentrated sulfuric acid or methanesulfonic acid, the hydroxyl group is protonated and eliminated as water, generating a highly reactive carbocation. This cationic center is immediately trapped by the ortho-position of the adjacent phenyl ring attached to the phosphorus atom, closing the ring to form the rigid spiro-acridine framework. This mechanism ensures high regioselectivity and minimizes the formation of linear oligomeric byproducts that often plague similar condensation reactions.
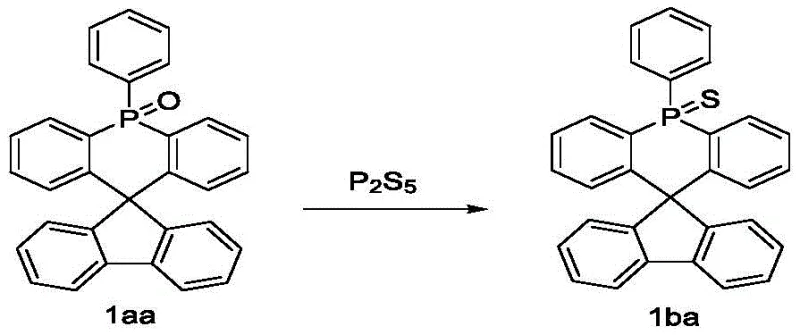
Following the formation of the spiro-phosphine core, the electronic properties are fine-tuned through a controlled oxidation sequence. The trivalent phosphorus atom is initially oxidized to the pentavalent phosphine oxide state using hydrogen peroxide in acetic acid, a mild yet effective reagent system that avoids over-oxidation of the sensitive aromatic rings. For applications requiring deeper HOMO levels or enhanced electron transport, the oxygen atom can be subsequently substituted with sulfur or selenium using reagents like Lawesson's reagent or elemental sulfur. This post-functionalization step leverages the sigma-star/pi-star hyperconjugation effects of the P=S or P=Se bonds to stabilize the LUMO level, providing a powerful tool for cost reduction in electronic chemical manufacturing by allowing a single intermediate to be diversified into multiple product grades.
How to Synthesize Aryl Acridine Phosphine Spiro Compounds Efficiently
The synthesis of these high-value OLED intermediates follows a streamlined four-stage protocol designed for reproducibility and scalability. The process initiates with low-temperature lithiation to ensure clean metal-halogen exchange, followed by nucleophilic addition and acid-mediated ring closure to establish the core skeleton. Subsequent oxidation and chalcogen exchange steps allow for the final tuning of electrochemical properties. Detailed standard operating procedures regarding stoichiometry, solvent choices, and workup protocols are essential for maintaining the high purity required for electronic applications.
- Perform halogen-lithium exchange on 2-bromotriarylphosphine using n-BuLi at low temperatures (-10 to -120°C) followed by nucleophilic addition to a ketone substrate.
- Execute an intramolecular ring-closing reaction in the presence of a strong acid catalyst (e.g., sulfuric acid or methanesulfonic acid) at 20-120°C to form the spiro core.
- Oxidize the phosphine center using hydrogen peroxide in acetic acid, followed by optional sulfurization or selenization to tune the electronic properties of the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers significant strategic benefits over traditional multi-step cross-coupling methods often used for similar host materials. The reliance on readily available bulk chemicals such as bromotriarylphosphines and fluorenones reduces dependency on exotic or single-source precursors, thereby enhancing supply chain reliability and mitigating the risk of raw material shortages. Furthermore, the reaction conditions utilize common industrial solvents like tetrahydrofuran, toluene, and acetic acid, which simplifies solvent recovery and waste management processes. This compatibility with standard chemical infrastructure facilitates the commercial scale-up of complex organic semiconductors without requiring specialized high-pressure or cryogenic equipment beyond standard low-temperature reactors.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts such as palladium or iridium in the key bond-forming steps, relying instead on cost-effective lithium and acid reagents. This removal of precious metals not only lowers the direct material cost but also drastically simplifies the downstream purification process by avoiding the need for rigorous heavy metal scavenging steps. The high yields reported in the cyclization and oxidation stages further contribute to substantial cost savings by maximizing the output per batch and minimizing the volume of waste solvent generated per kilogram of product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commodity chemicals with established global supply chains, ensuring consistent availability and price stability. Unlike custom-synthesized intermediates that may face long lead times, the precursors used here can be sourced from multiple vendors, reducing the risk of production bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry environments for the cyclization step, ensures high batch-to-batch consistency, which is critical for reducing lead time for high-purity OLED material batches destined for mass production lines.
- Scalability and Environmental Compliance: The process is inherently scalable as it avoids hazardous reagents like gaseous chlorine or highly toxic cyanides, aligning with modern green chemistry principles. The use of aqueous workups and standard column chromatography or recrystallization for purification allows for straightforward adaptation to multi-kilogram or ton-scale production. Additionally, the byproducts generated, primarily salts and water, are easier to treat compared to heavy metal waste streams, supporting environmental compliance and reducing the overall ecological footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of these spiro-compounds in device fabrication. The answers are derived directly from the experimental data and theoretical calculations presented in the patent documentation, ensuring accuracy for R&D teams evaluating these materials for integration into their current device stacks.
Q: What are the advantages of the spiro structure in these OLED materials?
A: The spiro structure provides a rigid, three-dimensional geometry that prevents molecular aggregation and concentration quenching. This results in high thermal stability, a stable amorphous state, and effectively separated HOMO-LUMO orbitals for improved charge transport.
Q: How can the energy levels of these compounds be tuned?
A: The frontier molecular orbital energy levels can be precisely regulated by modifying the heteroatom X bonded to the phosphorus atom. Replacing oxygen with sulfur or selenium alters the electron-donating capability and hyperconjugation effects, shifting the HOMO and LUMO levels.
Q: Are these materials suitable for blue phosphorescent OLEDs?
A: Yes, these compounds possess high triplet energy levels due to their limited pi-conjugation and orthogonal structural units. This makes them excellent host materials for confining triplet excitons in blue, green, and red phosphorescent devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acridine Phosphine Spiro Compounds Supplier
As the demand for high-efficiency OLED displays continues to surge, the ability to source advanced host materials with consistent quality is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific low-temperature lithiation and acid-catalyzed cyclization steps required for these spiro compounds, ensuring that every batch meets stringent purity specifications. With rigorous QC labs employing HPLC, GC-MS, and NMR analysis, we guarantee that our materials are free from trace impurities that could compromise device lifetime or efficiency.
We invite procurement leaders and technical directors to collaborate with us on optimizing their material supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our aryl acridine phosphine spiro compounds can enhance your next-generation OLED products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
