Optimizing ABT-263 Production: A Technical Analysis of Novel Synthetic Routes for Commercial Scale-up
The development of potent Bcl-2 family protein antagonists has revolutionized oncology treatment strategies, with compound ABT-263 standing out as a critical candidate for inducing apoptosis in tumor cells. As the pharmaceutical industry seeks more efficient pathways to access these complex molecules, Patent CN101220008B presents a groundbreaking synthetic methodology that addresses longstanding bottlenecks in production. This patent details a robust three-fragment coupling strategy that not only simplifies the operational complexity but also drastically enhances the environmental profile of the synthesis. By shifting away from hazardous photochemical conditions and difficult gaseous reagents, this new approach offers a viable pathway for reliable API intermediate supplier networks aiming to secure stable supply chains for next-generation cancer therapeutics. The structural complexity of ABT-263, characterized by its distinct sulfonamide and cyclohexene moieties, demands precise chemical control, which this invention successfully delivers through optimized reaction conditions.
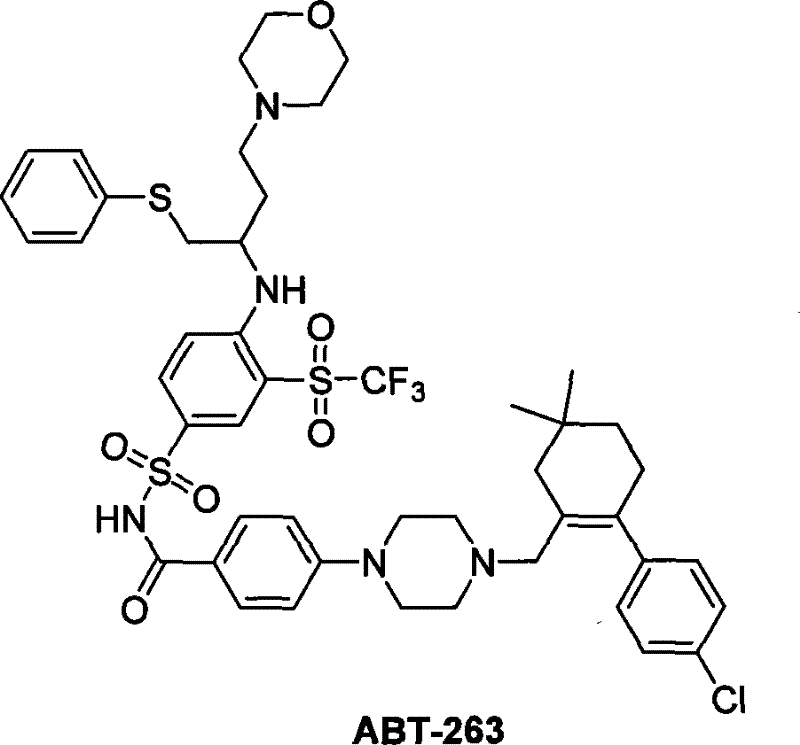
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in US2007027135, relied heavily on the use of trifluoromethyl iodide, a gaseous reagent that poses significant handling challenges and requires specialized photochemical catalysis equipment. These conventional routes were plagued by low operational efficiency and the necessity for stoichiometric amounts of heavy metal oxidants, leading to substantial environmental pollution and high waste disposal costs. Furthermore, the oxidation steps in traditional syntheses often suffered from poor yields, reportedly as low as 17%, which severely impacted the economic feasibility of large-scale manufacturing. The reliance on such harsh and inefficient conditions created a barrier to entry for many manufacturers, limiting the availability of high-purity intermediates and driving up the cost of goods for downstream drug developers. Additionally, the multi-step nature of previous routes increased the risk of impurity accumulation, complicating the purification processes required to meet stringent pharmaceutical standards.
The Novel Approach
The innovative strategy outlined in Patent CN101220008B fundamentally reengineers the synthesis by introducing a catalytic oxidation system that utilizes chromium trioxide in catalytic quantities rather than stoichiometric amounts. This modification alone boosts the oxidation yield dramatically from 17% to approximately 90%, representing a massive gain in atom economy and resource utilization. Moreover, the integration of a Mannich reaction for constructing the cyclohexene fragment effectively shortens the synthetic sequence, eliminating unnecessary intermediate isolation steps and reducing overall processing time. By replacing the problematic photochemical steps with standard thermal reactions and utilizing readily available starting materials, this method significantly lowers the technical barrier for production. The result is a streamlined process that is not only more cost-effective but also aligns better with modern green chemistry principles, making it an attractive option for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Catalytic Oxidation and Fragment Coupling
The core of this synthetic advancement lies in the meticulous design of three distinct fragments that are converged in the final stages. Fragment 2, the sulfonamide core, is synthesized through a refined oxidation pathway where 2-fluorotrifluoromethylphenylsulfide is converted to the corresponding sulfone using a catalytic amount of chromium trioxide combined with periodic acid. This mechanistic shift avoids the generation of excessive heavy metal waste, a common issue in traditional sulfone preparations. The subsequent chlorosulfonation and amination steps proceed under mild conditions to yield the key sulfonamide intermediate, ensuring high purity and minimal byproduct formation. This careful control over the oxidation state is critical for maintaining the integrity of the fluorinated aromatic ring, which is essential for the biological activity of the final molecule.
Simultaneously, Fragment 3 is constructed via a highly efficient Mannich reaction involving paraformaldehyde, 4,4-dimethylcyclohexanone, and a piperazine derivative. This one-pot condensation creates the necessary carbon-nitrogen backbone with excellent regioselectivity. The resulting intermediate then undergoes a Grignard reaction with 4-chlorobromobenzene to install the chlorophenyl group, followed by hydrolysis to reveal the carboxylic acid functionality. The final assembly involves the coupling of Fragment 2 and Fragment 3 using EDCI and DMAP in dichloromethane, a standard yet highly effective amide bond formation protocol. This convergent approach allows for parallel synthesis of fragments, optimizing production timelines and facilitating easier quality control of individual components before final assembly.
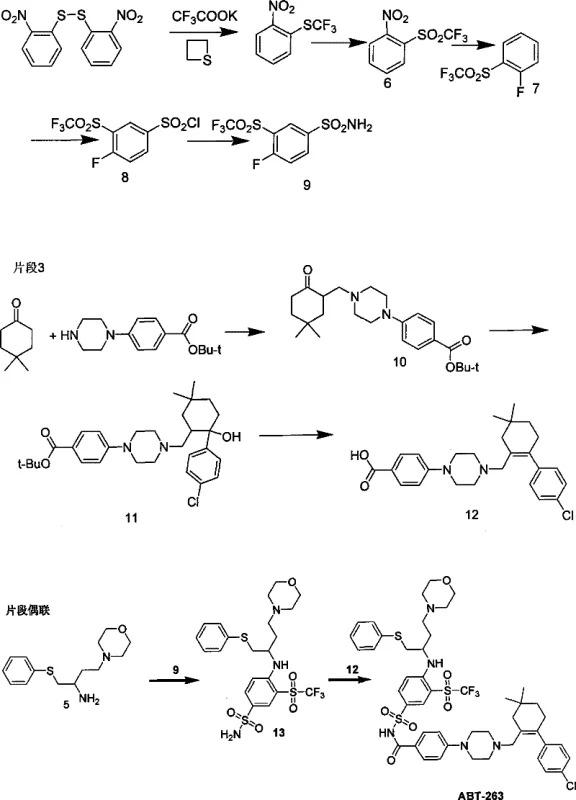
How to Synthesize ABT-263 Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly during the oxidation and coupling phases. The process begins with the preparation of the amine-containing Fragment 1 from protected aspartic acid derivatives, followed by the independent synthesis of the sulfonamide and cyclohexene fragments. The detailed standardized synthetic steps see the guide below, which outlines the specific reagents, temperatures, and workup procedures necessary to achieve the reported high yields. Adhering to these protocols ensures that the critical stereochemical and functional group integrity is maintained throughout the sequence, ultimately delivering the target molecule with the purity required for clinical applications.
- Synthesize Fragment 1 via reductive amination of protected aspartic acid derivatives to form the amine core.
- Prepare Fragment 2 (Sulfonamide) using improved catalytic chromium trioxide oxidation to achieve high yields without heavy metal waste.
- Construct Fragment 3 via Mannich reaction and Grignard addition, followed by final amide coupling with EDCI/DMAP to yield ABT-263.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of specialized photochemical equipment and gaseous reagents like trifluoromethyl iodide removes significant capital expenditure requirements and safety hazards associated with traditional methods. This simplification of the process infrastructure allows for more flexible manufacturing setups and reduces the dependency on niche suppliers for hazardous materials. Furthermore, the dramatic increase in yield during the oxidation step translates directly into reduced raw material consumption, lowering the overall cost of goods sold without compromising on quality. These factors combined create a more resilient supply chain capable of meeting fluctuating market demands for oncology intermediates.
- Cost Reduction in Manufacturing: The transition to a catalytic oxidation system eliminates the need for expensive stoichiometric oxidants and reduces waste disposal costs associated with heavy metal residues. By shortening the synthetic route through the Mannich reaction, the process consumes less solvent and energy per kilogram of product, leading to substantial cost savings in utility and material overheads. The use of common coupling agents like EDCI and DMAP further ensures that reagent costs remain predictable and manageable within standard pharmaceutical budgets.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as aspartic acid derivatives and common ketones mitigates the risk of supply disruptions often caused by specialty reagents. The robustness of the reaction conditions, which operate at standard temperatures and pressures, ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts. This reliability is essential for downstream partners who require uninterrupted access to high-quality intermediates for their own drug development pipelines.
- Scalability and Environmental Compliance: The avoidance of heavy metal pollution and the reduction of three wastes (waste water, waste gas, waste residue) align the process with increasingly strict environmental regulations globally. The simplified workflow facilitates easier scale-up from laboratory to commercial production volumes, as the unit operations involved are standard in the fine chemical industry. This scalability ensures that the manufacturing capacity can be expanded rapidly to support clinical trials and eventual commercial launch without requiring extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their own production needs. The answers provided are derived directly from the technical specifications and experimental data presented in the patent documentation.
Q: How does the new oxidation method improve yield compared to prior art?
A: The patented method replaces traditional stoichiometric oxidation with a catalytic chromium trioxide system, increasing yields from approximately 17% to over 90% while minimizing environmental impact.
Q: What are the key advantages of the Mannich reaction in this synthesis?
A: Utilizing the Mannich reaction significantly shortens the synthetic route for the cyclohexene fragment, reducing the number of isolation steps and improving overall process efficiency.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the avoidance of difficult-to-handle gaseous reagents like trifluoromethyl iodide and the use of standard coupling agents like EDCI make this route highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ABT-263 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex oncology targets like ABT-263. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in implementing catalytic oxidation and convergent synthesis strategies allows us to offer competitive pricing without sacrificing the quality essential for clinical success.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall development timeline.
