Advanced Asymmetric Cyclopropanation for High-Purity Pyrethroid Intermediates and Pharmaceutical Building Blocks
The synthesis of optically active cyclopropane compounds represents a critical frontier in the manufacturing of high-value agrochemicals and pharmaceutical intermediates, particularly for the production of pyrethroid insecticides and complex drug scaffolds. Patent CN1852887A introduces a transformative methodology that addresses long-standing challenges in asymmetric catalysis, specifically regarding the balance between cost-efficiency and stereochemical control. This innovation centers on the utilization of a novel asymmetric copper complex derived from optically active cycloalkylidenebisoxazoline compounds. By leveraging this specialized ligand architecture, the process enables the efficient conversion of prochiral olefins and diazoacetic esters into high-purity trans-cyclopropane derivatives with exceptional enantiomeric excess. For R&D directors and procurement strategists, this technology offers a pathway to optimize impurity profiles while simultaneously reducing reliance on exotic, high-cost reagents, thereby securing a more robust and economically viable supply chain for essential chemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric cyclopropanation of olefins has been plagued by a significant trade-off between reagent cost and stereoselectivity. Conventional methods utilizing standard bisoxazoline ligands often struggle to achieve sufficient (+)-trans isomer selectivity when employing industrially advantageous lower alkyl diazoacetates, such as ethyl diazoacetate. To compensate for this lack of selectivity, prior art methodologies frequently necessitate the use of sterically demanding and prohibitively expensive diazoacetic esters, such as (2,6-di-tert-butyl-4-methylphenyl)diaz oacetate. This reliance on bulky reagents not only inflates raw material costs but also complicates downstream processing due to the difficulty in removing large aromatic byproducts. Furthermore, the modest trans/cis ratios achieved with traditional catalysts often require extensive and yield-eroding purification steps, such as repeated recrystallization or preparative chromatography, which are impractical for large-scale commercial manufacturing. These inefficiencies create a bottleneck for producers aiming to deliver cost reduction in agrochemical intermediate manufacturing without compromising on the stringent purity specifications required by global regulatory bodies.
The Novel Approach
The methodology disclosed in CN1852887A fundamentally disrupts this paradigm by introducing a rigidified ligand system that enforces superior stereocontrol even with simple, commodity-grade reagents. The core innovation lies in the use of optically active cycloalkylidenebisoxazoline compounds, where the oxazoline rings are bridged by a cycloalkylidene group, typically a cyclopropane ring. This structural rigidity creates a well-defined chiral environment around the copper center, effectively shielding one face of the reactive carbene intermediate and directing the olefin approach with high precision. Consequently, the process achieves remarkable trans-selectivity and enantiomeric purity (e.g., 97% e.e. for the trans isomer in specific embodiments) using ethyl diazoacetate, a reagent that is vastly more economical and accessible than its bulky counterparts. This shift allows manufacturers to bypass the economic penalties associated with specialized reagents, facilitating a streamlined synthesis route that is both chemically elegant and commercially scalable for the production of high-purity OLED material precursors or pharmaceutical intermediates.
Mechanistic Insights into Asymmetric Copper-Catalyzed Cyclopropanation
The efficacy of this catalytic system is rooted in the precise geometric constraints imposed by the cycloalkylidenebisoxazoline ligand upon the copper center. When the optically active ligand, represented by Formula (1), complexes with a copper salt such as copper(I) trifluoromethanesulfonate, it forms a cationic or neutral species that acts as a highly selective Lewis acid. The cyclopropane bridge in the ligand backbone restricts the rotational freedom of the oxazoline rings, locking them into a specific conformation that maximizes steric differentiation between the quadrants of the metal center. Upon reaction with the diazoacetic ester, the copper complex generates a metallacarbenoid species. Due to the rigid chiral pocket, the approach of the prochiral olefin substrate is strictly governed, favoring the formation of the thermodynamically stable trans-isomer while suppressing the cis-pathway. This mechanistic fidelity ensures that the resulting cyclopropane ring is formed with the desired absolute configuration, minimizing the formation of diastereomeric impurities that are notoriously difficult to separate.
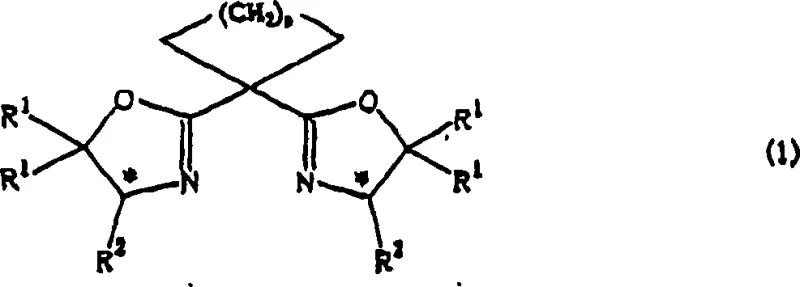
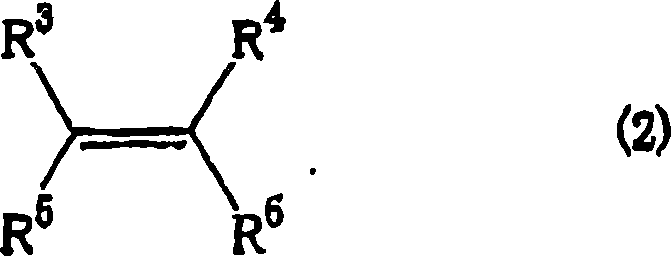
Furthermore, the versatility of this catalytic system is evidenced by its tolerance to a wide array of functional groups on the olefin substrate. As illustrated by the general structure of the prochiral olefin in Formula (2), the reaction accommodates substituents ranging from simple alkyl groups to more complex functionalities like halogens, ethers, and esters. This broad substrate scope is critical for R&D teams designing synthetic routes for diverse molecular targets. The mechanism proceeds through a concerted or stepwise carbene transfer that preserves the stereochemical integrity of the olefin while establishing two new chiral centers on the cyclopropane ring simultaneously. The ability to tune the R groups on the ligand (such as tert-butyl, benzyl, or phenyl groups) allows for further optimization of the catalyst's electronic and steric properties, enabling fine-tuning of the enantioselectivity for specific challenging substrates. This level of control is paramount for ensuring the commercial scale-up of complex polymer additives or active pharmaceutical ingredients where trace impurities can have significant biological or material consequences.
How to Synthesize Optically Active Cyclopropane Compounds Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The synthesis begins with the in situ generation of the active catalyst by mixing the chiral ligand and copper source in a dry, inert solvent such as dichloromethane or dichloroethane. Once the homogeneous catalyst solution is formed, the prochiral olefin is introduced, and the mixture is cooled to optimize selectivity, typically between -20°C and 0°C. The diazoacetate is then added slowly to control the exotherm and minimize dimerization side reactions. Following the reaction, the workup is remarkably simple, often requiring only concentration of the reaction mixture followed by standard purification techniques. For detailed operational parameters and safety guidelines regarding the handling of diazo compounds, please refer to the standardized synthesis steps provided below.
- Preparation of the Asymmetric Copper Catalyst: Mix an optically active cycloalkylidenebisoxazoline compound (Formula 1) with a copper compound (e.g., copper(I) trifluoromethanesulfonate) in a halogenated hydrocarbon solvent under inert atmosphere.
- Reaction Setup: Add the prochiral olefin substrate (Formula 2) to the catalyst solution and adjust the temperature to between -20°C and 80°C, preferably around 0°C.
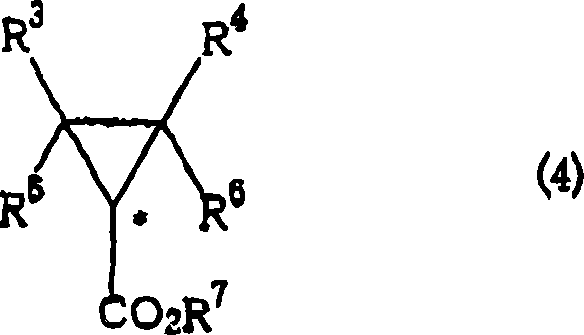
- Cyclopropanation and Workup: Slowly add the diazoacetic ester (Formula 3) to the mixture. After completion, concentrate the reaction solution and purify the resulting optically active cyclopropane compound (Formula 4) via distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalytic technology offers profound advantages that extend beyond mere chemical yield. The primary value driver is the drastic simplification of the raw material portfolio. By enabling the use of ethyl diazoacetate and simple olefins instead of specialized, bulky esters, procurement managers can leverage existing commodity supply chains, significantly mitigating the risk of supply disruption associated with niche reagents. This transition not only stabilizes the supply of high-purity agrochemical intermediates but also insulates the manufacturing process from the price volatility often seen in the market for custom-synthesized fine chemicals. Moreover, the high selectivity of the reaction reduces the burden on downstream purification units, leading to substantial cost savings in solvent consumption, energy usage, and waste disposal. The elimination of complex separation trains for cis/trans isomers translates directly into improved throughput and reduced lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The ability to utilize low-cost, industrially available diazoacetic esters like ethyl diazoacetate represents a major economic breakthrough. Traditional methods often require expensive, sterically hindered diazo reagents to achieve acceptable selectivity, which drastically inflates the Cost of Goods Sold (COGS). By switching to this novel catalyst system, manufacturers can eliminate the premium associated with these specialty reagents. Additionally, the high trans-selectivity minimizes the loss of material during purification, effectively increasing the overall process yield without the need for additional reaction cycles. This efficiency gain allows for a significant reduction in manufacturing costs, making the final API or agrochemical active more competitive in the global marketplace while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Reliance on exotic reagents creates single points of failure in the supply chain. The reagents required for this process—simple olefins, copper salts, and lower alkyl diazoacetates—are produced by multiple global vendors, ensuring a robust and redundant supply network. This diversification reduces the risk of production stoppages due to vendor-specific issues or logistical bottlenecks. For supply chain heads, this means greater predictability in delivery schedules and the ability to secure long-term contracts at favorable rates. The stability of the catalyst system also implies that inventory management can be optimized, as there is less need to hold safety stock of unstable or hard-to-source intermediates, thereby freeing up working capital and improving overall operational liquidity.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently under conditions that are easily replicated in large-scale reactors. The use of common solvents like dichloromethane or toluene simplifies solvent recovery and recycling operations, aligning with modern green chemistry principles. Furthermore, the high atom economy of the cyclopropanation reaction, combined with the reduced need for extensive chromatographic purification, results in a lower environmental footprint. This is increasingly critical as regulatory pressures on chemical manufacturing intensify globally. By adopting a process that generates less waste and consumes fewer resources, companies can ensure compliance with stringent environmental regulations while enhancing their corporate sustainability profile, a key factor for partnerships with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric cyclopropanation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What is the primary advantage of the ligand described in CN1852887A over conventional bisoxazolines?
A: The cycloalkylidenebisoxazoline ligand features a rigid cyclopropane bridge that significantly enhances stereocontrol. Unlike conventional ligands that often require bulky, expensive diazoacetates to achieve high trans-selectivity, this novel ligand allows the use of industrially favorable, low-cost lower alkyl diazoacetates (like ethyl diazoacetate) while maintaining excellent enantiomeric excess (up to 97% e.e.) and trans/cis ratios.
Q: Can this process be scaled for commercial production of pyrethroid intermediates?
A: Yes, the process is designed for industrial applicability. It utilizes readily available starting materials such as simple prochiral olefins and commodity diazoacetates. The reaction conditions are moderate (-20°C to 80°C), and the workup involves standard concentration and purification techniques, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Q: What types of substrates are compatible with this asymmetric copper complex?
A: The catalyst system demonstrates broad substrate scope, accommodating various prochiral olefins including those with halogen, alkoxy, and ester substituents. It is particularly effective for synthesizing precursors to pyrethroids, such as 2,2-dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylates and 3,3-dimethyl-2-(benzyloxymethyl)cyclopropanecarboxylates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Cyclopropane Compound Supplier
The technological advancements detailed in CN1852887A underscore the immense potential of asymmetric copper catalysis in modern chemical synthesis. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering; we adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the identity and purity of every batch. Whether you require custom synthesis of complex chiral intermediates or reliable supply of established building blocks, our technical team is prepared to support your project from gram-scale development to full commercial manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced cyclopropanation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
