Advanced Fluorinated Cobalt Salts for Superior Rubber-Metal Adhesion and Thermal Stability
Introduction to Advanced Fluorinated Cobalt Adhesion Promoters
The development of high-performance rubber additives is critical for the longevity and safety of radial tires and industrial rubber products, particularly in demanding environments where metal-rubber adhesion is paramount. Patent CN103183845B introduces a groundbreaking methodology for synthesizing fluorine-containing cobalt neodecanoate and cobalt boroacylate mixed salts, representing a significant evolution from traditional cobalt-based adhesion promoters. This technology leverages the unique electronic properties of fluorine atoms to modify the coordination environment around the cobalt center, resulting in a product with superior thermal stability and enhanced reactivity during the vulcanization process. By integrating fluorinated organic acids such as trifluoroacetic acid and pentafluoropropionic acid into the molecular structure, the resulting cobalt salts exhibit markedly improved resistance to aging and weathering compared to their non-fluorinated counterparts. For R&D directors and technical leaders in the polymer industry, this patent offers a compelling pathway to next-generation rubber compounds that can withstand higher operating temperatures and harsher chemical exposures without compromising interfacial bonding strength.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing processes for cobalt neodecanoate and related adhesion promoters have long relied on solvent-based acid-base reactions, typically utilizing volatile organic compounds like toluene or xylene as reaction media. These conventional methods suffer from inherent inefficiencies, including the need for extensive solvent recovery systems, high energy consumption for distillation, and significant environmental liabilities associated with VOC emissions. Furthermore, standard cobalt salts often lack the necessary thermal robustness for modern high-performance rubber applications, leading to premature degradation of the adhesive interface under extreme heat or oxidative stress. The reliance on simple carboxylic acids without functional modification limits the tunability of the cobalt complex, often resulting in products with inconsistent cobalt content and suboptimal dispersion within the rubber matrix. These factors collectively contribute to higher production costs and variable quality in the final rubber-metal composite products, creating a persistent bottleneck for manufacturers seeking to optimize supply chain reliability and product consistency.
The Novel Approach
The innovative process described in the patent data circumvents these historical challenges by employing a solvent-free thermal synthesis route that directly reacts cobalt hydroxide with a precise blend of fluorinated and non-fluorinated carboxylic acids. This approach not only eliminates the safety hazards and costs associated with flammable solvents but also allows for precise control over the ligand composition surrounding the cobalt ion through a sequential addition strategy. The introduction of fluorine atoms fundamentally alters the physicochemical properties of the salt, imparting greater electrophilicity to the carboxyl groups and enhancing the overall stability of the molecule against thermal decomposition.  As illustrated in the reaction pathway, the subsequent boroacylation step further boosts the reactivity of the cobalt species, ensuring rapid and effective migration to the rubber-metal interface during curing. This streamlined, one-pot style methodology significantly simplifies the production workflow, reducing unit operations and minimizing waste generation while delivering a product with demonstrably higher cobalt content and superior functional performance.
As illustrated in the reaction pathway, the subsequent boroacylation step further boosts the reactivity of the cobalt species, ensuring rapid and effective migration to the rubber-metal interface during curing. This streamlined, one-pot style methodology significantly simplifies the production workflow, reducing unit operations and minimizing waste generation while delivering a product with demonstrably higher cobalt content and superior functional performance.
Mechanistic Insights into Fluorine-Modified Cobalt Coordination Chemistry
The core scientific advancement in this technology lies in the strategic manipulation of the cobalt coordination sphere through the incorporation of highly electronegative fluorine substituents on the carboxylate ligands. When fluorinated acids such as trifluoroacetic acid react with cobalt hydroxide, the resulting cobalt-fluoro-carboxylate bonds exhibit distinct electronic characteristics compared to standard alkyl-carboxylates, primarily due to the strong electron-withdrawing nature of the fluorine atoms. 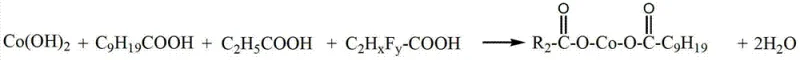 This electronic effect increases the acidity of the parent carboxylic acid and stabilizes the negative charge on the carboxylate anion, which in turn influences the lability of the cobalt-ligand bond during the rubber vulcanization cycle. The mechanism involves an initial acid-base neutralization where water is continuously removed under vacuum to drive the equilibrium towards salt formation, ensuring high conversion rates without the need for azeotropic distillation agents. The presence of fluorine also enhances the lipophilicity of the cobalt complex, facilitating better compatibility with the hydrophobic rubber matrix and promoting more uniform distribution throughout the compound. For technical teams, understanding this mechanistic nuance is vital, as it explains the observed improvements in heat resistance and the ability of the additive to maintain adhesion strength even after prolonged exposure to elevated temperatures.
This electronic effect increases the acidity of the parent carboxylic acid and stabilizes the negative charge on the carboxylate anion, which in turn influences the lability of the cobalt-ligand bond during the rubber vulcanization cycle. The mechanism involves an initial acid-base neutralization where water is continuously removed under vacuum to drive the equilibrium towards salt formation, ensuring high conversion rates without the need for azeotropic distillation agents. The presence of fluorine also enhances the lipophilicity of the cobalt complex, facilitating better compatibility with the hydrophobic rubber matrix and promoting more uniform distribution throughout the compound. For technical teams, understanding this mechanistic nuance is vital, as it explains the observed improvements in heat resistance and the ability of the additive to maintain adhesion strength even after prolonged exposure to elevated temperatures.
Following the initial salt formation, the process employs a sophisticated ligand exchange or metathesis reaction to fine-tune the final product composition and remove potentially corrosive short-chain acids. 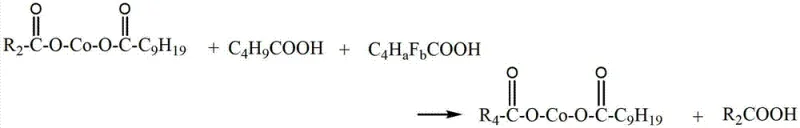 In this critical step, higher molecular weight acids like pivalic acid and fluorinated valeric acid are introduced to displace a portion of the more volatile propionic acid and its fluorinated analogues. This exchange is thermodynamically driven by the removal of the lower boiling point acids under reduced pressure and elevated temperatures ranging from 160°C to 190°C. The retention of specific fluorinated valeric acid moieties ensures that the beneficial effects of fluorination are preserved in the final long-chain structure, while the displacement of excessive short-chain acids mitigates the risk of corrosion to the steel wire cords in the final application. This dual-stage synthesis allows for a highly customized impurity profile, effectively balancing reactivity with stability, and ensuring that the final cobalt boron mixed salt meets stringent specifications for both cobalt content and free acid levels.
In this critical step, higher molecular weight acids like pivalic acid and fluorinated valeric acid are introduced to displace a portion of the more volatile propionic acid and its fluorinated analogues. This exchange is thermodynamically driven by the removal of the lower boiling point acids under reduced pressure and elevated temperatures ranging from 160°C to 190°C. The retention of specific fluorinated valeric acid moieties ensures that the beneficial effects of fluorination are preserved in the final long-chain structure, while the displacement of excessive short-chain acids mitigates the risk of corrosion to the steel wire cords in the final application. This dual-stage synthesis allows for a highly customized impurity profile, effectively balancing reactivity with stability, and ensuring that the final cobalt boron mixed salt meets stringent specifications for both cobalt content and free acid levels.
How to Synthesize Fluorine-Containing Cobalt Neodecanoate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced adhesion promoters, emphasizing precise temperature control and sequential reagent addition to achieve the desired molecular architecture. The process begins with the careful blending of cobalt hydroxide and a specific ratio of propionic, fluorinated propionic, and neodecanoic acids, followed by a controlled heating phase to initiate the acid-base reaction and remove generated water. Subsequent steps involve the strategic addition of pivalic and fluorinated valeric acids to induce ligand exchange, followed by a high-temperature boroacylation reaction with butyl borate to finalize the active species. This structured approach ensures reproducibility and scalability, making it an ideal candidate for transfer from laboratory benchtop to large-scale commercial manufacturing facilities.
- Mix cobalt hydroxide with a blend of propionic acid, fluorinated propionic acid, and neodecanoic acid, then heat to 150-170°C to drive off water and form the initial cobalt salt mixture.
- Add pivalic acid and fluorinated valeric acid to the reaction system, raising the temperature to 160-190°C to facilitate ligand exchange and displace volatile short-chain acids.
- React the resulting fluorinated cobalt neodecanoate with butyl borate at 180-200°C for boroacylation, followed by neutralization with activated calcium carbonate to stabilize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fluorinated cobalt salt technology offers substantial strategic benefits, primarily driven by the elimination of hazardous solvents and the simplification of the manufacturing process. Traditional solvent-based routes require significant capital investment in explosion-proof equipment, solvent storage tanks, and complex recovery distillation columns, all of which add to the operational expenditure and extend the lead time for production batches. By shifting to a solvent-free thermal process, manufacturers can drastically reduce utility costs associated with solvent heating and condensation, while simultaneously lowering the regulatory burden related to VOC emissions and hazardous waste disposal. This streamlined operation translates directly into a more resilient supply chain, as the production process becomes less susceptible to disruptions caused by solvent shortages or environmental compliance audits. Furthermore, the use of readily available bulk chemicals like cobalt hydroxide and common carboxylic acids, supplemented by specialized fluorinated acids, ensures a stable raw material base that supports consistent long-term supply.
- Cost Reduction in Manufacturing: The transition to a solvent-free synthesis route fundamentally alters the cost structure of producing high-performance cobalt adhesion promoters by removing the need for expensive organic solvents and their associated recovery infrastructure. Without the requirement to purchase, store, and recycle large volumes of toluene or xylene, the direct material costs are significantly lowered, and the energy intensity of the process is reduced due to the absence of solvent distillation steps. Additionally, the simplified workflow reduces labor hours and maintenance requirements for complex solvent handling systems, leading to substantial overall cost savings in the manufacturing of these specialty rubber additives. These efficiencies allow for a more competitive pricing structure for high-purity fluorinated cobalt salts, providing value to downstream rubber compounders without sacrificing performance quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by minimizing dependency on volatile solvent markets and reducing the complexity of logistics involved in handling flammable liquids. The process relies on stable solid and liquid feedstocks that are easier to transport and store, thereby reducing the risk of shipment delays or regulatory hold-ups at ports and borders. Moreover, the higher thermal stability and longer shelf-life of the fluorinated product mean that inventory can be held for extended periods without degradation, offering greater flexibility in managing stock levels and responding to fluctuating market demand. This reliability is crucial for automotive and industrial tire manufacturers who require just-in-time delivery of critical adhesion promoters to maintain their own production schedules.
- Scalability and Environmental Compliance: Scaling this technology from pilot batches to full commercial production is straightforward due to the absence of solvent-related safety constraints and the use of standard reactor equipment capable of high-temperature operation. The process generates minimal hazardous waste, primarily consisting of water and displaced short-chain acids which can be managed or recycled, aligning perfectly with increasingly stringent global environmental regulations. This eco-friendly profile not only future-proofs the manufacturing facility against tightening emission standards but also enhances the brand reputation of the supplier as a sustainable partner in the green chemistry initiative. The ability to scale up while maintaining strict control over product purity and cobalt content ensures that the supply of these advanced additives can grow in tandem with the expanding demand for high-performance radial tires.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this fluorinated cobalt salt technology, derived directly from the patent specifications and process details. These insights are intended to clarify the operational advantages and performance characteristics for stakeholders evaluating this material for their rubber compounding formulations.
Q: How does fluorine incorporation improve cobalt salt performance in rubber compounds?
A: The introduction of fluorine atoms creates strong C-F bonds which significantly enhance the thermal stability and weather resistance of the cobalt salt. This structural modification increases the electrophilicity of the carboxyl group, improving the reactivity of the cobalt complex during the vulcanization process and leading to stronger rubber-to-metal interfacial adhesion.
Q: What are the safety advantages of this solvent-free synthesis method compared to traditional processes?
A: Traditional methods often rely on volatile organic solvents like toluene or xylene, which pose significant fire hazards and require complex recovery systems. This patented process operates via direct thermal acid-base reaction and vacuum dehydration, eliminating the need for flammable solvents and drastically reducing VOC emissions and associated safety risks in the manufacturing plant.
Q: Why is activated calcium carbonate added in the final stage of production?
A: Activated calcium carbonate serves as a stabilizing agent to buffer the pH of the final mixture. Since the synthesis involves strong fluorinated acids which can leave the product overly acidic, the calcium carbonate neutralizes excess acidity, preventing potential corrosion of steel wire cords in the final rubber application and ensuring the long-term stability of the adhesion promoter.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Cobalt Neodecanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced adhesion promoters in the production of durable and high-performance rubber goods, and we are uniquely positioned to support your transition to this next-generation technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex thermal synthesis of fluorinated cobalt salts is executed with precision and consistency. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for automotive and industrial applications, guaranteeing optimal rubber-metal bonding and thermal stability. Our commitment to quality assurance means that you receive a product that not only matches the patent specifications but is optimized for seamless integration into your existing compounding processes.
We invite you to collaborate with us to explore how this innovative fluorinated cobalt technology can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply capabilities can support your long-term strategic goals in the competitive rubber additives market.
