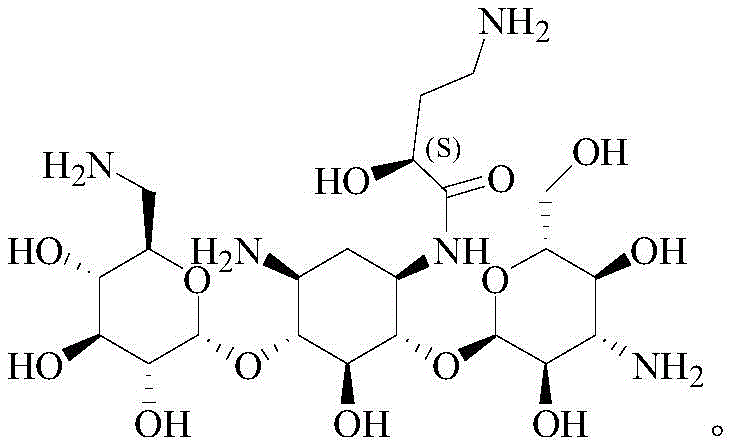
Scalable Amikacin Production via Zinc-Mediated Chelation for Global Pharmaceutical Supply Chains
Introduction to Next-Generation Amikacin Synthesis
The global demand for high-purity aminoglycoside antibiotics continues to drive innovation in process chemistry, particularly for critical drugs like Amikacin. A significant technological breakthrough in this sector is detailed in patent CN111138505B, which discloses a novel preparation method that fundamentally shifts away from hazardous silanization techniques toward a more sustainable metal chelation strategy. This patent outlines a robust synthetic route starting from Kanamycin A, utilizing acetate chelation to achieve superior regioselectivity without the need for corrosive silylating agents. For pharmaceutical manufacturers and procurement leaders, this represents a pivotal opportunity to optimize supply chains by adopting a process that is not only environmentally friendlier but also operationally simpler. By leveraging the coordination chemistry of metal ions such as zinc, the method effectively masks specific reactive sites on the Kanamycin A scaffold, enabling precise functionalization at the 1-position amino group. This technical advancement addresses long-standing pain points in antibiotic intermediate manufacturing, including equipment corrosion, hazardous waste generation, and complex purification workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Amikacin has relied heavily on silanization protection strategies, broadly categorized into persilylation and partial silylation routes. As illustrated in the prior art reaction schemes, these methods typically involve treating Kanamycin A with trimethylchlorosilane (TMSCl) to protect multiple hydroxyl and amino groups simultaneously. 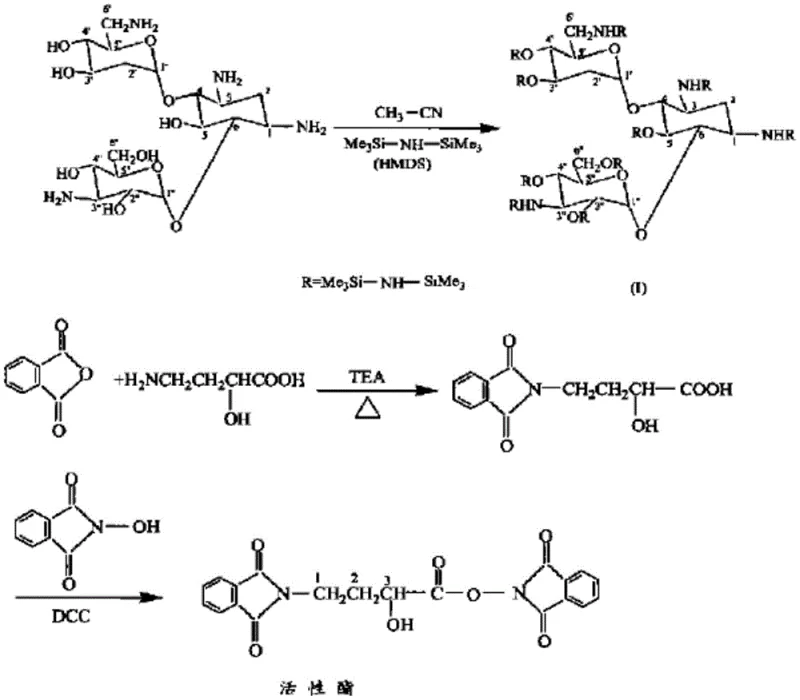 This approach suffers from severe inherent drawbacks that impact both operational safety and cost efficiency. Firstly, TMSCl is highly susceptible to hydrolysis, releasing hydrogen chloride gas which is corrosive to reactor vessels and piping, necessitating expensive corrosion-resistant materials and rigorous maintenance protocols. Secondly, the persilylation route requires the protection of all seven hydroxyl groups and four amino groups, leading to prolonged reaction times and energy-intensive post-treatment steps such as high-temperature, high-vacuum distillation to remove excess silyl reagents. Furthermore, certain variations of this route utilize N-hydroxyphthalimide (NOP) as a coupling auxiliary, which is classified as a high-sensitization substance, posing significant occupational health risks to plant personnel. The selectivity of these silanization methods is also suboptimal, often resulting in a mixture of 1-amino and 3-amino acylation byproducts in a ratio of approximately 1:2 to 1:3, which drastically reduces overall yield and complicates downstream purification.
This approach suffers from severe inherent drawbacks that impact both operational safety and cost efficiency. Firstly, TMSCl is highly susceptible to hydrolysis, releasing hydrogen chloride gas which is corrosive to reactor vessels and piping, necessitating expensive corrosion-resistant materials and rigorous maintenance protocols. Secondly, the persilylation route requires the protection of all seven hydroxyl groups and four amino groups, leading to prolonged reaction times and energy-intensive post-treatment steps such as high-temperature, high-vacuum distillation to remove excess silyl reagents. Furthermore, certain variations of this route utilize N-hydroxyphthalimide (NOP) as a coupling auxiliary, which is classified as a high-sensitization substance, posing significant occupational health risks to plant personnel. The selectivity of these silanization methods is also suboptimal, often resulting in a mixture of 1-amino and 3-amino acylation byproducts in a ratio of approximately 1:2 to 1:3, which drastically reduces overall yield and complicates downstream purification.
The Novel Approach
In stark contrast, the methodology described in patent CN111138505B introduces a sophisticated chelation-based protection strategy that circumvents the pitfalls of silicon chemistry. Instead of bulky silyl groups, this novel approach employs metal ions, specifically from acetates like zinc acetate, to form transient coordination complexes with the Kanamycin A molecule. 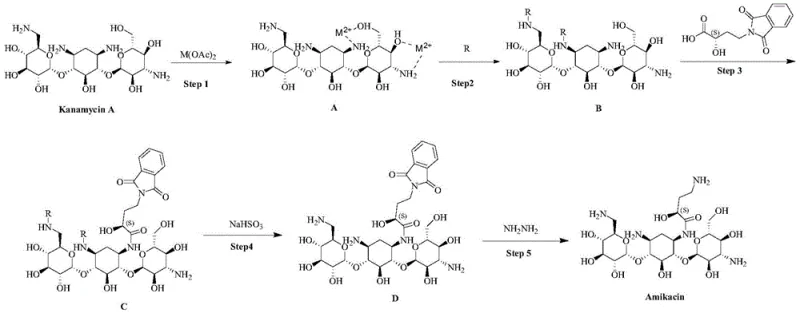 The process begins with the chelation of the 1-position and 3'-position amino groups (note: patent text clarifies 1 and 3 position amino groups are coordinated with adjacent hydroxyls), effectively reducing their nucleophilicity through electronic modulation rather than steric blocking. Subsequently, protecting groups such as p-nitrocarbobenzoxy (PNZ) are introduced selectively to the remaining active amino positions (3 and 6'). This orthogonal protection scheme allows for the highly selective condensation of the 1-position amino group with the side chain precursor, gamma-phthalimide-alpha-hydroxybutyric acid (PHBA). The result is a streamlined workflow that eliminates the need for corrosive chlorosilanes and sensitizing reagents, operating under milder conditions that are far more conducive to large-scale commercial production. The final steps involve straightforward deprotection and hydrazinolysis, yielding Amikacin with exceptional purity levels exceeding 99.0%.
The process begins with the chelation of the 1-position and 3'-position amino groups (note: patent text clarifies 1 and 3 position amino groups are coordinated with adjacent hydroxyls), effectively reducing their nucleophilicity through electronic modulation rather than steric blocking. Subsequently, protecting groups such as p-nitrocarbobenzoxy (PNZ) are introduced selectively to the remaining active amino positions (3 and 6'). This orthogonal protection scheme allows for the highly selective condensation of the 1-position amino group with the side chain precursor, gamma-phthalimide-alpha-hydroxybutyric acid (PHBA). The result is a streamlined workflow that eliminates the need for corrosive chlorosilanes and sensitizing reagents, operating under milder conditions that are far more conducive to large-scale commercial production. The final steps involve straightforward deprotection and hydrazinolysis, yielding Amikacin with exceptional purity levels exceeding 99.0%.
Mechanistic Insights into Zinc-Mediated Chelation Protection
The core innovation of this synthesis lies in the precise exploitation of coordination chemistry to control regioselectivity, a critical factor for R&D directors focused on impurity profiles. In the native Kanamycin A structure, there are four amino groups with varying reactivities; typically, the 6'-amino is the most reactive, followed by the 1 and 3 amino groups, with the 3'-amino being the least reactive due to steric shielding. Direct acylation would predominantly target the 6'-position, leading to the wrong isomer. The patented method solves this by introducing divalent metal ions ($M^{2+}$), such as $Zn^{2+}$, which act as temporary masking agents. 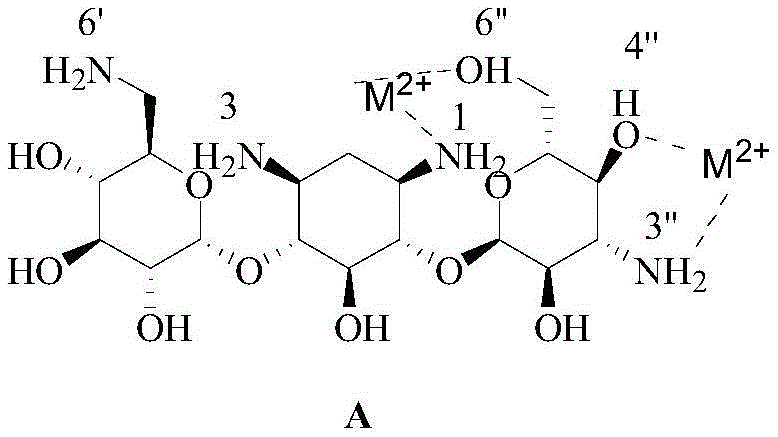 These metal ions form stable five-membered or six-membered chelate rings by coordinating with electron-donating groups—specifically pairing the 1-amino with the 6-hydroxyl, and the 3-amino with the 4-hydroxyl. This coordination withdraws electron density from the nitrogen atoms, significantly dampening their nucleophilic character and rendering them inert to acylation. Meanwhile, the 3 and 6' amino groups are sterically or electronically prevented from forming similar stable chelates in this specific configuration, leaving them exposed and reactive. This allows for the selective introduction of PNZ protecting groups at these positions. Once the 3 and 6' positions are capped, the metal chelate can be manipulated or the reaction conditions adjusted to allow the 1-amino group to react with the activated ester of PHBA. This mechanism ensures that the acylation occurs almost exclusively at the desired 1-position, minimizing the formation of the troublesome 3-amino isomer and other regio-isomeric impurities that plague traditional silanization routes.
These metal ions form stable five-membered or six-membered chelate rings by coordinating with electron-donating groups—specifically pairing the 1-amino with the 6-hydroxyl, and the 3-amino with the 4-hydroxyl. This coordination withdraws electron density from the nitrogen atoms, significantly dampening their nucleophilic character and rendering them inert to acylation. Meanwhile, the 3 and 6' amino groups are sterically or electronically prevented from forming similar stable chelates in this specific configuration, leaving them exposed and reactive. This allows for the selective introduction of PNZ protecting groups at these positions. Once the 3 and 6' positions are capped, the metal chelate can be manipulated or the reaction conditions adjusted to allow the 1-amino group to react with the activated ester of PHBA. This mechanism ensures that the acylation occurs almost exclusively at the desired 1-position, minimizing the formation of the troublesome 3-amino isomer and other regio-isomeric impurities that plague traditional silanization routes.
From an impurity control perspective, this mechanistic precision translates directly to a cleaner crude product. Traditional methods often require extensive column chromatography to separate the 1-acyl and 3-acyl isomers due to their similar polarities. By suppressing the reactivity of the 3-amino group via chelation during the critical bond-forming step, the new process inherently limits the generation of these hard-to-remove byproducts. The use of zinc acetate, a benign and inexpensive salt, further ensures that no toxic heavy metal residues persist in the final API, simplifying the regulatory clearance process. The subsequent deprotection steps using sodium bisulfite and sodium hydroxide are well-understood unit operations that efficiently remove the PNZ groups without degrading the sensitive glycosidic linkages of the aminoglycoside core. This level of mechanistic control provides a robust platform for consistent batch-to-batch quality, a key metric for any reliable antibiotic intermediate supplier.
How to Synthesize Amikacin Efficiently
The synthesis of Amikacin via this chelation route is designed for operational simplicity and scalability, making it an ideal candidate for technology transfer. The process initiates with the dissolution of Kanamycin A in a lower alcohol solvent, such as methanol, followed by the addition of zinc acetate to form the chelated Intermediate A. This step proceeds at ambient temperatures, avoiding the need for cryogenic cooling or excessive heating. Following chelation, the selective protection of the 3 and 6' amino groups is achieved using PNZ-HOSu in a dichloromethane system. The subsequent condensation with PHBA utilizes standard peptide coupling reagents like DCC and HOBT in DMF, a familiar protocol for most organic synthesis facilities. The final deprotection and hydrazinolysis steps utilize aqueous workups and recrystallization or resin adsorption for purification. For detailed standardized operating procedures, stoichiometry, and specific reaction parameters, please refer to the technical guide below.
- Chelation Protection: React Kanamycin A with zinc acetate in methanol to coordinate metal ions with 1,3-amino and adjacent hydroxyl groups, forming Intermediate A.
- Selective Amino Protection: Introduce PNZ protecting groups to the exposed 3 and 6' amino positions of Intermediate A to yield Intermediate B.
- Side Chain Condensation: React Intermediate B with gamma-phthalimide-alpha-hydroxybutyric acid (PHBA) using DCC/HOBT to acylate the 1-amino position, forming Intermediate C.
- Deprotection and Hydrazinolysis: Remove PNZ groups using NaOH/NaHSO3 to get Intermediate D, followed by hydrazine hydrate treatment to cleave the phthalimide group and yield pure Amikacin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from silanization to metal chelation offers tangible strategic benefits beyond mere chemical elegance. The elimination of trimethylchlorosilane removes a major bottleneck related to equipment integrity and safety compliance. Silane reagents require specialized storage and handling due to their moisture sensitivity and corrosive byproducts, often necessitating lined reactors and scrubber systems that increase capital expenditure. By replacing these with stable metal acetates, the new process significantly reduces the risk of unplanned downtime caused by equipment corrosion and lowers the barrier for contract manufacturing organizations (CMOs) to adopt the technology. Furthermore, the avoidance of N-hydroxyphthalimide (NOP) mitigates liability associated with handling sensitizing agents, contributing to a safer workplace and potentially lower insurance premiums. The simplified workup procedures, which replace high-vacuum distillations with filtration and extraction, lead to substantial reductions in energy consumption and cycle time, enhancing overall throughput.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the simplification of the reaction workflow and the reduction in hazardous waste disposal costs. By eliminating the need for high-vacuum distillation to remove excess silanes, the process drastically cuts energy usage and extends the lifespan of processing equipment. The reagents employed, such as zinc acetate and standard carbodiimides, are commodity chemicals with stable pricing and widespread availability, insulating the supply chain from the volatility often seen with specialized silylating agents. Additionally, the higher regioselectivity reduces the load on purification columns, decreasing the consumption of silica gel and solvents, which are significant cost drivers in API manufacturing. These factors combine to deliver a more cost-efficient production model without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for essential antibiotics, and this method strengthens resilience by relying on robust, non-proprietary raw materials. The reliance on zinc acetate and common organic solvents like methanol and ethanol ensures that production is not vulnerable to shortages of niche fluorinated or silylated reagents. The milder reaction conditions also reduce the risk of batch failures due to thermal runaways or moisture ingress, which are common failure modes in silanization chemistry. This reliability allows for more accurate forecasting and inventory planning, ensuring that downstream formulation teams receive a steady stream of high-quality intermediates. The process is inherently scalable, meaning that capacity can be ramped up quickly to meet surges in demand without requiring extensive re-engineering of the plant infrastructure.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this chelation route aligns perfectly with modern green chemistry principles. The absence of corrosive hydrogen chloride emissions simplifies废气 treatment requirements, and the aqueous waste streams are easier to treat compared to those containing organosilicon byproducts. The process generates less hazardous solid waste, particularly by avoiding the large quantities of urea byproducts often associated with carbodiimide couplings in complex silanized matrices. This reduced environmental footprint facilitates easier permitting for new production lines and ensures compliance with increasingly stringent global environmental regulations. The ability to produce high-purity Amikacin with a cleaner profile supports sustainability goals and enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chelation-based Amikacin synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating process validation and for procurement specialists assessing vendor capabilities. The answers highlight the specific advantages in selectivity, safety, and scalability that define this next-generation manufacturing approach.
Q: How does the metal chelation method improve selectivity compared to traditional silanization?
A: Traditional silanization relies on steric hindrance which often yields a mixture of 1-amino and 3-amino acylation products (ratio ~1:2-3). The metal chelation method utilizes Zn2+ ions to form stable coordination bonds specifically with the 1-amino/6-hydroxyl and 3-amino/4-hydroxyl pairs. This electronically deactivates these positions, leaving the 3 and 6' amino groups available for initial protection, thereby forcing the subsequent acylation to occur exclusively at the 1-amino position with high regioselectivity.
Q: What are the safety advantages of this process over the persilylation route?
A: The conventional persilylation route requires trimethylchlorosilane (TMSCl), which hydrolyzes to form corrosive hydrogen chloride, damaging equipment and posing health risks. It also often employs N-hydroxyphthalimide (NOP), a known sensitizer. The patented chelation method eliminates TMSCl and NOP entirely, utilizing safer reagents like zinc acetate and standard carbodiimides, significantly reducing occupational hazards and equipment maintenance costs.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It operates under mild conditions (room temperature to 50°C), avoids high-vacuum distillation required for removing excess silanes, and simplifies post-treatment to filtration and extraction. The process achieves a final product purity of ≥99.0%, minimizing the burden on downstream purification columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amikacin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the zinc-mediated chelation method is critical for maintaining competitiveness in the global antibiotic market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific unit operations required for this process, from precise chelation control to efficient resin-based purification, guaranteeing stringent purity specifications for every batch. We understand that consistency is key in the pharmaceutical supply chain, and our rigorous QC labs employ advanced analytical methods to verify the absence of regio-isomeric impurities and residual metals, delivering a product that meets the highest international pharmacopoeia standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their Amikacin supply needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener, more efficient route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Together, we can secure a sustainable and cost-effective supply of this vital antibiotic intermediate.
