Scalable Synthesis of Asymmetric Triarylmethane Derivatives Using Recyclable Bismuth Catalysts
Scalable Synthesis of Asymmetric Triarylmethane Derivatives Using Recyclable Bismuth Catalysts
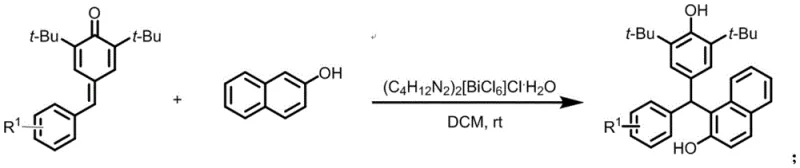
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN110483252B, which discloses a novel method for synthesizing asymmetric triarylmethane derivatives. This technology leverages a unique bismuth-based complex, specifically (C4H12N2)2[BiCl6]Cl·H2O, to catalyze the 1,6-conjugate addition reaction between p-methylene benzoquinone (p-QMs) derivatives and 2-hydroxynaphthol. Unlike traditional methods that often rely on harsh conditions or expensive, non-recyclable catalysts, this innovation operates at room temperature in dichloromethane, offering a streamlined pathway to complex molecular scaffolds essential for the pharmaceutical and functional materials industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of asymmetric triarylmethane skeletons has relied heavily on classical Friedel-Crafts alkylations or metal-catalyzed coupling reactions. While effective in certain contexts, these conventional approaches suffer from significant drawbacks that hinder their utility in modern, large-scale manufacturing. Traditional Lewis acid catalysts such as BF3·Et2O often require strictly anhydrous conditions and low temperatures to control selectivity, leading to high energy consumption and complex operational protocols. Furthermore, many organocatalysts like N-heterocyclic carbenes or phospholines, while selective, are often expensive to synthesize and cannot be easily recovered from the reaction mixture. This inability to recycle catalysts results in substantial waste generation and inflated production costs, creating a bottleneck for the reliable supply of high-purity pharmaceutical intermediates. Additionally, the use of toxic solvents or the generation of hazardous byproducts in these older methods poses serious environmental compliance challenges for chemical manufacturers aiming for sustainable operations.
The Novel Approach
The methodology presented in patent CN110483252B represents a paradigm shift towards sustainable and efficient synthesis. By utilizing the bismuth complex (C4H12N2)2[BiCl6]Cl·H2O, the reaction achieves high conversion rates under remarkably mild conditions. The process involves a direct 1,6-conjugate addition where the electrophilic p-methylene benzoquinone reacts with the nucleophilic 2-hydroxynaphthol. As illustrated in the general reaction scheme below, this transformation proceeds smoothly at room temperature (25-35°C) in dichloromethane, eliminating the need for cryogenic cooling or high-pressure equipment. The robustness of this catalytic system allows for a wide substrate scope, accommodating various substituents on the aromatic rings without compromising yield. Crucially, the solid nature of the catalyst facilitates its recovery via simple filtration, enabling multiple reuse cycles which drastically reduces the overall catalyst cost per kilogram of product.
Mechanistic Insights into Bismuth-Catalyzed 1,6-Conjugate Addition
The efficacy of this synthetic route lies in the unique electronic and structural properties of the bismuth catalyst. Bismuth, as a heavy Group 15 element, exhibits significant Lewis acidity due to the presence of empty orbitals that can coordinate with electron-rich species. In this reaction mechanism, the bismuth center likely coordinates with the carbonyl oxygen or the conjugated system of the p-methylene benzoquinone, thereby enhancing its electrophilicity. This activation lowers the energy barrier for the nucleophilic attack by the 2-hydroxynaphthol at the exocyclic methylene carbon. The specific structure of the catalyst, featuring a hexachlorobismuthate anion stabilized by protonated piperazine cations, provides a balanced environment that promotes reactivity while maintaining stability in the reaction medium. This precise coordination chemistry ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired triarylmethane product over potential side products.
![Chemical structure of the bismuth catalyst (C4H12N2)2[BiCl6]Cl·H2O](/insights/img/asymmetric-triarylmethane-bismuth-catalysis-pharma-supplier-20260303181322-02.webp)
Impurity control is another critical aspect where this mechanistic understanding translates to practical value. The mild reaction conditions (room temperature) inherently suppress thermal degradation pathways and polymerization side reactions that are common with highly reactive quinone methides under harsher conditions. Furthermore, the specific steric and electronic environment provided by the catalyst helps to minimize non-selective background reactions. By avoiding strong Brønsted acids or aggressive Lewis acids that might promote rearrangement or decomposition, the process yields a cleaner crude product profile. This simplifies downstream purification, as fewer byproducts mean less intensive chromatography or recrystallization steps are required to meet the stringent purity specifications demanded by the pharmaceutical industry. The result is a process that not only delivers high yields but also ensures a consistent and manageable impurity profile.
How to Synthesize Asymmetric Triarylmethane Derivatives Efficiently
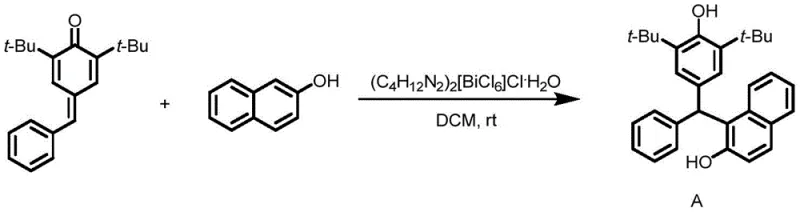
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, reflecting the patent's emphasis on operational simplicity. The general protocol involves charging a reaction vessel with the p-methylene benzoquinone derivative and 2-hydroxynaphthol in a molar ratio of approximately 1:2 to 1:3. Dichloromethane is added as the solvent, followed by the introduction of the bismuth catalyst at a loading of 10 mol%. The mixture is then stirred at ambient temperature, with reaction progress monitored by TLC until the starting material is consumed. For instance, the synthesis of compound A (Example 1) demonstrates the efficiency of this method, achieving a 92% yield with high purity after standard workup and column chromatography. This specific example highlights how the steric bulk of the tert-butyl groups on the quinone methide does not hinder the reaction, showcasing the catalyst's tolerance for sterically demanding substrates.
- Prepare the reaction mixture by combining p-methylene benzoquinone derivatives and 2-hydroxynaphthol in dichloromethane solvent.
- Add the bismuth catalyst (C4H12N2)2[BiCl6]Cl·H2O at a loading of 10 mol% relative to the quinone methide substrate.
- Stir the reaction at room temperature (25-35°C) until completion, then filter to recover the catalyst and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bismuth-catalyzed technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for specialized low-temperature reactors or high-pressure vessels, capital investment requirements for new production lines are significantly lowered. Moreover, the ability to operate at room temperature translates to substantial energy savings, as there is no need for continuous heating or cooling utilities during the reaction phase. This energy efficiency contributes to a lower carbon footprint, aligning with the increasing global demand for green chemistry practices in the supply chain of fine chemicals and active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the recyclability of the catalyst. In traditional processes where catalysts are single-use, the cost of noble metals or complex organocatalysts can constitute a significant portion of the raw material bill. Here, the bismuth catalyst can be recovered by simple filtration, washed, and reused, effectively amortizing its cost over multiple batches. Additionally, the high yields reported (often exceeding 90%) minimize raw material waste, ensuring that expensive starting materials like substituted benzaldehydes and naphthols are converted efficiently into the final product. The use of common solvents like dichloromethane, which are readily available and easily recycled, further optimizes the cost structure compared to processes requiring exotic or difficult-to-remove solvents.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable pharmaceutical intermediate supplier. This synthesis route enhances reliability by utilizing robust, commercially available starting materials and a catalyst that does not rely on critically scarce precious metals like palladium or platinum. The wide substrate scope demonstrated in the patent means that a single production platform can be adapted to manufacture a diverse library of triarylmethane derivatives simply by changing the aldehyde precursor. This flexibility allows manufacturers to respond rapidly to changing market demands or custom synthesis requests without needing to retool entire production lines. The simplicity of the workup procedure also reduces the turnaround time between batches, increasing overall throughput and ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to multi-ton production often reveals hidden complexities, but this method is inherently scalable due to its mild conditions and lack of exothermic hazards. The absence of highly reactive or pyrophoric reagents simplifies safety management and regulatory compliance. From an environmental perspective, the reduction in waste generation—both from the recyclable catalyst and the high atom economy of the addition reaction—facilitates easier waste treatment and disposal. This aligns with stringent environmental regulations in major chemical manufacturing hubs, reducing the risk of production shutdowns due to compliance issues and ensuring a sustainable long-term supply of high-purity organic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bismuth-catalyzed synthesis. These insights are derived directly from the experimental data and technical disclosures within patent CN110483252B, providing a clear picture of the technology's capabilities and limitations for potential partners and licensees.
Q: Can the bismuth catalyst be recycled in this synthesis process?
A: Yes, the patent explicitly states that the catalyst (C4H12N2)2[BiCl6]Cl·H2O can be recovered by filtration after the reaction, washed, dried, and reused multiple times without significant loss of activity, which significantly lowers production costs.
Q: What are the typical reaction conditions for this 1,6-conjugate addition?
A: The reaction proceeds efficiently at room temperature (25-35°C) in dichloromethane (DCM). It requires a molar ratio of approximately 1:2 between the p-methylene benzoquinone derivative and 2-hydroxynaphthol, with a catalyst loading of 10 mol%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial applicability due to its simple operation, short reaction time, use of common solvents like DCM, and the ability to recycle the catalyst, making it environmentally friendly and cost-effective for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Triarylmethane Supplier
The technological advancements described in patent CN110483252B underscore the immense potential of bismuth catalysis in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative routes in delivering high-quality intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of asymmetric triarylmethane derivatives meets the exacting standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize these green synthesis routes for maximum efficiency and cost-effectiveness.
We invite you to explore how this advanced synthesis technology can benefit your supply chain. Whether you require custom synthesis of specific triarylmethane analogs or large-scale production of key intermediates, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition from development to commercial supply.
