Advanced Non-Metallic Catalytic Route for Scalable Production of Bioactive Triazole Scaffolds
Introduction to Next-Generation Triazole Synthesis
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, safer, and more economically viable synthetic pathways. Patent CN111925335B introduces a groundbreaking preparation method for 5-amino-1,2,3-triazole compounds, a structural motif ubiquitous in high-value pharmaceutical agents ranging from anticancer drugs to antiviral medications. This innovation addresses critical bottlenecks in the production of these essential scaffolds by replacing hazardous reagents and costly catalysts with a benign, base-mediated system. The biological significance of this core structure cannot be overstated, as evidenced by its presence in potent carboxylamidotriazoles and potassium channel activators that are currently pivotal in treating inflammatory kidney diseases and viral infections. By shifting the paradigm from transition-metal dependence to simple alkaline catalysis, this technology offers a robust platform for the sustainable manufacturing of complex nitrogen-containing heterocycles.
Furthermore, the strategic value of this patent lies in its ability to streamline the supply chain for active pharmaceutical ingredients (APIs) that rely on triazole backbones. Traditional synthesis routes have long been plagued by safety concerns regarding explosive intermediates and the environmental burden of heavy metal waste. The disclosed method utilizes readily available carbodiimides and diazo compounds, reacting them under mild conditions to achieve high yields without the need for rigorous anhydrous or anaerobic environments. This operational simplicity translates directly into reduced capital expenditure for reactor specifications and lower operating costs for energy consumption. For global chemical manufacturers, adopting this methodology represents a proactive step towards aligning with increasingly stringent environmental regulations while maintaining the high purity standards required by top-tier pharmaceutical clients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
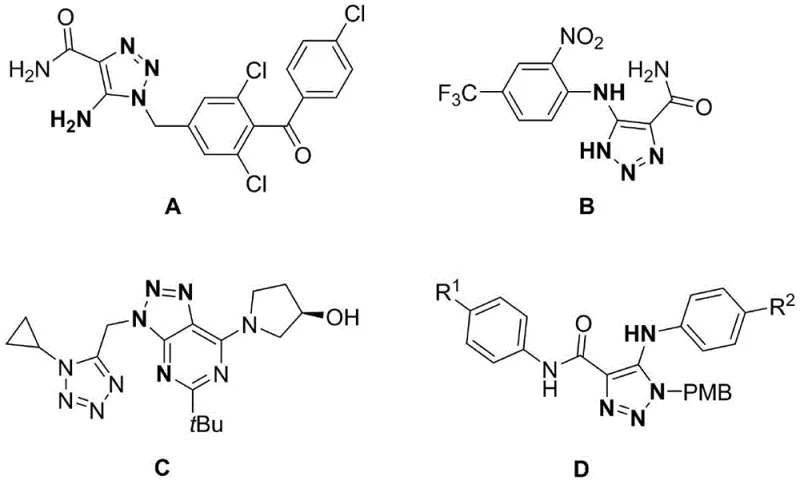
Historically, the construction of the 1,2,3-triazole ring has relied heavily on transition-metal-catalyzed azide-alkyne cycloadditions, a process that presents substantial challenges for large-scale industrial application. As illustrated in prior art, these conventional pathways typically necessitate the use of precious metal catalysts such as rhodium, ruthenium, or iridium, which are not only prohibitively expensive but also subject to volatile market pricing and supply constraints. Beyond the economic implications, the reliance on these heavy metals introduces severe complications in downstream processing, as residual metal levels must be reduced to parts-per-million concentrations to satisfy regulatory guidelines for human therapeutics. Additionally, the traditional reliance on organic azides poses a significant safety hazard due to their inherent instability and potential for explosive decomposition, requiring specialized handling protocols and explosion-proof facilities that drive up infrastructure costs.
Moreover, the reaction conditions associated with these legacy methods are often unforgiving, demanding strict exclusion of moisture and oxygen to prevent catalyst deactivation or side reactions. This sensitivity necessitates the use of high-purity solvents and inert gas blanketing, further escalating the operational complexity and cost of goods sold. The atom economy of these processes is frequently suboptimal, generating stoichiometric amounts of metal-containing waste that requires costly disposal procedures. For procurement managers and supply chain directors, these factors combine to create a fragile supply network where production delays can occur due to catalyst shortages or safety incidents. The cumulative effect of these limitations is a manufacturing process that is difficult to scale, environmentally burdensome, and economically inefficient compared to modern alternatives.
The Novel Approach
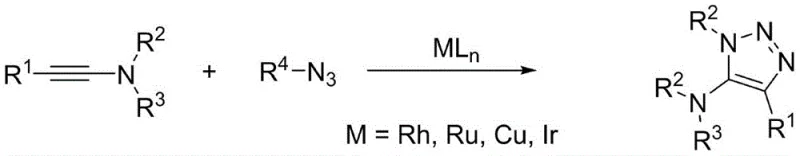
In stark contrast to the complexities of metal catalysis, the novel approach detailed in CN111925335B leverages a simple yet highly effective base-promoted cyclization between carbodiimides and diazo compounds. This metal-free strategy fundamentally alters the reaction landscape by eliminating the need for expensive transition metals entirely, thereby removing the associated costs of catalyst procurement and heavy metal scavenging steps. The reaction proceeds smoothly in common organic solvents such as acetonitrile under ambient or mildly heated conditions, demonstrating remarkable tolerance to a wide array of functional groups including halogens, esters, and ketones. This robustness ensures that diverse substrates can be processed using a unified set of parameters, significantly simplifying process development and reducing the time required to bring new derivatives to market.
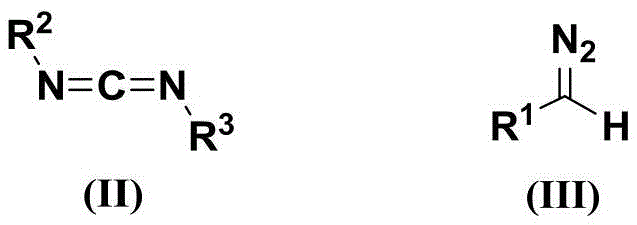
From a safety and compliance perspective, this methodology offers a decisive advantage by circumventing the use of unstable organic azides, replacing them with more manageable diazo precursors that react efficiently in the presence of inexpensive inorganic bases like potassium hydroxide. The absence of strict anhydrous requirements means that standard industrial reactors can be utilized without the need for specialized drying trains or inert atmosphere gloveboxes. This accessibility lowers the barrier to entry for contract manufacturing organizations and allows for seamless scale-up from laboratory benchtop to multi-ton commercial production. The result is a streamlined synthetic route that delivers high-purity 5-amino-1,2,3-triazole intermediates with superior atom economy and a drastically reduced environmental footprint, positioning it as the preferred choice for sustainable pharmaceutical manufacturing.
Mechanistic Insights into Base-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the nucleophilic addition of the diazo species to the electrophilic carbon of the carbodiimide, initiated by the deprotonation of the diazo compound by the base. Unlike metal-carbene pathways that involve complex coordination spheres and oxidative addition steps, this base-mediated mechanism proceeds through a straightforward anionic intermediate that undergoes rapid intramolecular cyclization to form the triazole ring. This pathway avoids the formation of metal-carbene complexes that can lead to unpredictable side reactions such as dimerization or insertion into C-H bonds, thereby enhancing the selectivity for the desired 5-amino substitution pattern. The kinetic profile of the reaction is favorable, typically reaching completion within 2 to 24 hours at temperatures ranging from 0°C to 100°C, which allows for precise control over reaction progress and minimizes the formation of thermal degradation byproducts.
Impurity control is inherently superior in this metal-free system because the primary sources of contamination—residual heavy metals and azide decomposition products—are absent from the reaction matrix. The crude product mixture primarily contains unreacted starting materials and simple salts derived from the base, which are easily removed during the aqueous workup or subsequent purification steps like column chromatography or recrystallization. This clean impurity profile is critical for R&D directors who must validate the safety of drug substances, as it simplifies the analytical characterization and reduces the risk of genotoxic impurities associated with metal residues. Furthermore, the high functional group tolerance ensures that sensitive moieties on the aryl or alkyl substituents remain intact throughout the synthesis, preserving the structural integrity required for downstream biological activity. This predictability makes the process highly reliable for generating diverse chemical libraries needed for structure-activity relationship studies.
How to Synthesize 5-Amino-1,2,3-Triazole Efficiently
Implementing this synthesis in a practical setting involves a straightforward sequence of operations that leverages standard laboratory and plant equipment. The process begins with the preparation of a reaction mixture containing the base and the carbodiimide substrate in a suitable solvent, followed by the controlled addition of the diazo compound. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by adding a base such as potassium hydroxide and a carbodiimide compound into an organic solvent like acetonitrile at room temperature.
- Introduce the diazo compound to the mixture and maintain stirring under mild conditions (0°C to 100°C) for 2 to 24 hours to facilitate nucleophilic addition and cyclization.
- Upon completion, concentrate the reaction system under reduced pressure and purify the crude residue via column chromatography or recrystallization to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis protocol offers transformative benefits that extend far beyond simple chemical efficiency. By eliminating the dependency on precious metal catalysts like rhodium and iridium, manufacturers can insulate their production costs from the extreme volatility characteristic of the precious metals market. This shift not only stabilizes the cost of goods sold but also removes the logistical burden of sourcing and qualifying high-purity metal catalysts from limited global suppliers. Additionally, the removal of heavy metals from the process flow negates the need for expensive scavenging resins and specialized filtration units, resulting in substantial capital savings and a reduction in consumable inventory requirements.
- Cost Reduction in Manufacturing: The economic impact of switching to a base-catalyzed system is profound, as it replaces high-cost catalytic systems with inexpensive, commodity-grade inorganic bases such as potassium hydroxide or cesium carbonate. This substitution drastically lowers the raw material cost per kilogram of the final product, allowing for more competitive pricing strategies in the global marketplace. Furthermore, the simplified purification process reduces solvent consumption and waste disposal fees, contributing to a leaner and more cost-effective manufacturing operation overall. The avoidance of complex metal removal steps also shortens the batch cycle time, increasing overall plant throughput and asset utilization without requiring additional capital investment.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by utilizing widely available starting materials like carbodiimides and diazo compounds, which are produced by numerous chemical vendors globally. Unlike specialized transition metal catalysts that may face supply disruptions or long lead times, these reagents can be sourced reliably in bulk quantities, ensuring continuous production schedules. The robustness of the reaction conditions also means that production is less susceptible to delays caused by environmental controls or equipment failures related to inert atmosphere maintenance. This reliability is crucial for meeting the just-in-time delivery expectations of major pharmaceutical clients and maintaining strong supplier relationships.
- Scalability and Environmental Compliance: The scalability of this process is exceptional due to its mild thermal profile and lack of exothermic hazards associated with azide chemistry. Facilities can scale production from pilot batches to commercial tons with minimal engineering changes, facilitating rapid response to market demand surges. From an environmental standpoint, the elimination of toxic heavy metals and explosive azides aligns perfectly with green chemistry principles and corporate sustainability goals. This compliance reduces the regulatory burden associated with hazardous waste permits and emissions reporting, making the facility more attractive to environmentally conscious investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential adopters.
Q: Why is this metal-free synthesis preferred over traditional transition metal catalysis?
A: Traditional methods often rely on expensive and toxic transition metals like rhodium or ruthenium, which require strict removal processes to meet pharmaceutical purity standards. This novel base-catalyzed approach eliminates heavy metal contamination risks entirely, simplifying downstream purification and reducing regulatory burdens.
Q: Does this method avoid the use of hazardous azide reagents?
A: Yes, unlike conventional [3+2] cycloadditions that utilize potentially explosive organic azides, this process employs stable carbodiimides and diazo compounds. This significantly enhances operational safety and reduces the need for specialized explosion-proof infrastructure in the manufacturing facility.
Q: What is the substrate scope for R1, R2, and R3 groups in this reaction?
A: The methodology demonstrates high functional group tolerance, accommodating various aryl and alkyl substituents including halogens, esters, and ketones. This versatility allows for the efficient generation of diverse chemical libraries for drug discovery without needing to optimize conditions for each specific derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the pharmaceutical intermediate sector. Our team of expert chemists has thoroughly evaluated the technology described in CN111925335B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this metal-free route, leveraging our rigorous QC labs and stringent purity specifications to deliver 5-amino-1,2,3-triazole derivatives that meet the highest global quality standards. Our commitment to process excellence ensures that every batch is optimized for yield and purity, minimizing impurities and maximizing value for our partners.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can accelerate your project timelines and reduce overall development costs.
